J Korean Acad Prosthodont.
2012 Jan;50(1):44-52.
Analysis of attachment, proliferation and differentiation response of human mesenchymal stem cell to various implant surfaces coated with rhBMP-2
- Affiliations
-
- 1Department of Medical Science, Major in Dentistry, Korea University, Seoul, Korea.
- 2Department of Chemical and Biomolecular Engineering, Patent and Tradement office, Daejeon, Korea.
- 3Department of Neuroscience, Medical research institute, Ansan Hospital, Korea University, Ansan, Korea.
- 4Department of Prosthodontics, Ansan Hospital, Korea University, Seoul, Ansan, Korea. koprosth@unitel.co.kr
Abstract
- PURPOSE
In this paper we tried to evaluate the most appropriate surface for rhBMP-2 coating among 4 rough titanium surfaces.
MATERIALS AND METHODS
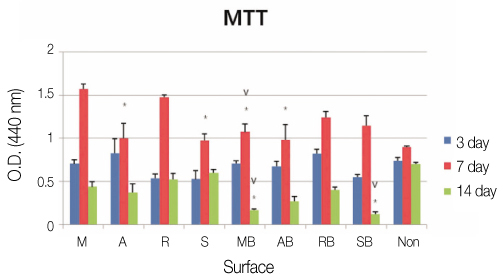
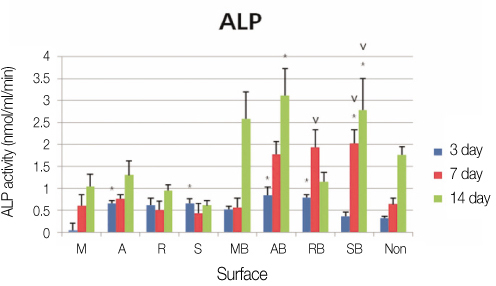
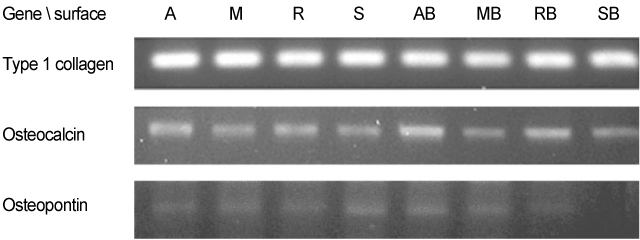
We used machined surface as a control group and anodized, RBM and SLA surfaces as test groups. We coated rhBMP-2 on the 4 surfaces and with uncoated surfaces for each case, we cultured human mesenchymal stem cells on all 8 surfaces. 24 hours after we measured the stem cell'attachment with SEM, and on 3rd, 7th, and 14th days, we checked the cell proliferation and differentiation by using MTT and ALP activity assay. And on the 7th day after the culture, we performed RT-PCR assay to determine whether the expression levels of Type I collagen, osteocalcin, osteopontin were changed.
RESULTS
We observed with SEM that 4 rhBMP-2 coated surfaces exhibited wider and tighter cell attachment and more cell process spreading than uncoated surfaces. The anodized rhBMP-2 surface caused robustest effects. In MTT assay we could not find any meaningful difference. In ALP assay there was a significant increase (P<.05) in the ALP activity of anodized rhBMP-2 coated surface compared with that of the control (3rd and 14th days) and with that of the RBM rhBMP-2 coated surface (14th day). In RT-PCR assay there was increased expressions in the anodized rhBMP-2 coated surface for osteocalcin, and osteopontin.
CONCLUSION
We found that the anodized rhBMP-2 coated surface were most prominent stem cell attachment and differentiation in compared to control and Machined rhBMP-2 coated, RBM rhBMP-2 coated surface.
Keyword
MeSH Terms
Figure
Reference
-
1. Wong M, Eulenberger J, Schenk R, Hunziker E. Effect of surface topography on the osseointegration of implant materials in trabecular bone. J Biomed Master Res. 1995. 29:1567–1576.2. Bessho K, Carnes DL, Cavin R, Chen HY, Ong JL. BMP stimulation of bone response adjacent to titanium implants in vivo. Clin Oral Implants Res. 1999. 10:212–218.3. Liu Y, Huse RO, de Groot K, Buser D, Hunziker EB. Delivery mode and efficacy of BMP-2 in association with implants. J Dent Res. 2007. 86:84–89.
Article4. Stadlinger B, Pilling E, Huhle M, Mai R, Bierbaum S, Scharnweber D, Kuhlisch E, Loukota R, Eckelt U. Evaluation of osseointegration of dental implants coated with collagen, chondroitin sulphate and BMP-4: an animal study. Int J Oral Maxillofac Surg. 2008. 37:54–59.
Article5. Schliephake H, Aref A, Scharnweber D, Bierbaum S, Roessler S, Sewing A. Effect of immobilized bone morphogenic protein 2 coating of titanium implants on peri-implant bone formation. Clin Oral Implants Res. 2005. 16:563–569.
Article6. Hartwig CH, Esenwein SA, Pfund A, Küsswetter Dagger W, Herr G. Improved osseointegration of titanium implants of different surface characteristics by the use of bone morphogenetic protein (BMP-3): an animal study performed at the metaphyseal bone bed in dogs. Z Orthop Ihre Grenzgeb. 2003. 141:705–711.
Article7. Esenwein SA, Esenwein S, Herr G, Muhr G, Küsswetter W, Hartwig CH. Osteogenetic activity of BMP-3-coated titanium specimens of different surface texture at the orthotopic implant bed of giant rabbits. Chirurg. 2001. 72:1360–1368.
Article8. Aebli N, Stich H, Schawalder P, Theis JC, Krebs J. Effects of bone morphogenetic protein-2 and hyaluronic acid on the osseointegration of hydroxyapatite-coated implants: an experimental study in sheep. J Biomed Mater Res A. 2005. 73:295–302.
Article9. Sumner DR, Turner TM, Urban RM, Turek T, Seeherman H, Wozney JM. Locally delivered rhBMP-2 enhances bone ingrowth and gap healing in a canine model. J Orthop Res. 2004. 22:58–65.
Article10. Hall J, Sorensen RG, Wozney JM, Wikesjö UM. Bone formation at rhBMP-2-coated titanium implants in the rat ectopic model. J Clin Periodontol. 2007. 34:444–451.
Article11. Choi KH, Moon KO, Kim SH, Yun JH, Jang KL, Cho KS. Purification and biological activity of recombinant human bone morphogenetic protein-2 produced by E. coli expression system. J Korean Acad Periodontol. 2008. 38:41–50.
Article12. Song SJ, Jeon O, Yang HS, Han DK, Kim BS. Effects of culture conditions on osteogenic differentiation in human mesenchymal stem cells. J Microbiol Biotechnol. 2007. 17:1113–1119.13. Chen D, Zhao M, Mundy GR. Bone morphogenetic proteins. Growth Factors. 2004. 22:233–241.
Article14. Urist MR. Bone formation by autoinduction. Science. 1965. 150:893–899.
Article15. Wozney JM, Rosen V, Celeste AJ, Mitsock LM, Whitters MJ, Kriz RW, Hewick RM, Wang EA. Novel regulators of bone formation: molecular clones and activities. Science. 1988. 242:1528–1534.
Article16. Wozney JM. Overview of bone morphogenetic proteins. Spine. 2002. 27:S2–S8.
Article17. Chen D, Harris MA, Rossini G, Dunstan CR, Dallas SL, Feng JQ, Mundy GR, Harris SE. Bone morphogenetic protein 2 (BMP-2) enhances BMP-3, BMP-4, and bone cell differentiation marker gene expression during the induction of mineralized bone matrix formation in cultures of fetal rat calvarial osteoblasts. Calcif Tissue Int. 1997. 60:283–290.
Article18. Takuwa Y, Ohse C, Wang EA, Wozney JM, Yamashita K. Bone morphogenetic protein-2 stimulates alkaline phosphatase activity and collagen synthesis in cultured osteoblastic cells, MC3T3-E1. Biochem Biophys Res Commun. 1991. 174:96–101.
Article19. Brekke JH, Toth JM. Principles of tissue engineering applied to programmable osteogenesis. J Biomed Mater Res. 1998. 43:380–398.
Article20. Lim TY, Wang W, Shi Z, Poh CK, Neoh KG. Human bone marrow-derived mesenchymal stem cells and osteoblast differentiation on titanium with surface-grafted chitosan and immobilized bone morphogenetic protein-2. J Mater Sci Mater Med. 2009. 20:1–10.
Article21. Shi Z, Neoh KG, Kang ET, Poh CK, Wang W. Surface functionalization of titanium with carboxymethyl chitosan and immobilized bone morphogenetic protein-2 for enhanced osseointegration. Biomacromolecules. 2009. 10:1603–1611.
Article22. Boyan BD, Bonewald LF, Paschalis EP, Lohmann CH, Rosser J, Cochran DL, Dean DD, Schwartz Z, Boskey AL. Osteoblastmediated mineral deposition in culture is dependent on surface microtopography. Calcif Tissue Int. 2002. 71:519–529.
Article23. Citeau A, Guicheux J, Vinatier C, Layrolle P, Nguyen TP, Pilet P, Daculsi G. In vitro biological effects of titanium rough surface obtained by calcium phosphate grid blasting. Biomaterials. 2005. 26:157–165.
Article24. Le Guehennec L, Lopez-Heredia MA, Enkel B, Weiss P, Amouriq Y, Layrolle P. Osteoblastic cell behaviour on different titanium implant surfaces. Acta Biomater. 2008. 4:535–543.
Article25. Li LH, Kong YM, Kim HW, Kim YW, Kim HE, Heo SJ, Koak JY. Improved biological performance of Ti implants due to surface modification by micro-arc oxidation. Biomaterials. 2004. 25:2867–2875.
Article26. Wall I, Donos N, Carlqvist K, Jones F, Brett P. Modified titanium surfaces promote accelerated osteogenic differentiation of mesenchymal stromal cells in vitro. Bone. 2009. 45:17–26.
Article27. Wennerberg A. On surface roughness and implant incorporation (Thesis). 1997. Göteborg, Sweden: Dept of Biomaterials, University of Göteborg.28. Balasundaram G, Yao C, Webster TJ. TiO2 nanotubes functionalized with regions of bone morphogenetic protein-2 increases osteoblast adhesion. J Biomed Mater Res A. 2008. 84:447–453.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Effect of RGD peptide coating of implant titanium surface on human mesenchymal stem cell response
- Attachment and Differentiation of Human Umbilical Cord Stem Cells on to the Tooth Root Surface with and without the Use of Fibroblast Growth Factor-An In Vitro Study
- A study of mesenchymal stem cell proliferation and surface characteristics of the titanium discs coated with MS275/PLGA by an electrospray
- The Role of Histone Acetylation in Mesenchymal Stem Cell Differentiation
- A study of growth factors, chondrogenic differentiation of mesenchymal stem cells and cell response by needle size differences in vitro