J Vet Sci.
2018 Mar;19(2):232-241. 10.4142/jvs.2018.19.2.232.
Recombinant-attenuated Salmonella Pullorum strain expressing the hemagglutinin-neuraminidase protein of Newcastle disease virus (NDV) protects chickens against NDV and Salmonella Pullorum challenge
- Affiliations
-
- 1Key Laboratory of Animal Disease and Public Health, Henan University of Science and Technology, and Key Laboratory of Live Carrier Biomaterial and Animal Disease Prevention and Control, Luoyang 471003, China. chengxch66@163.com
- KMID: 2407622
- DOI: http://doi.org/10.4142/jvs.2018.19.2.232
Abstract
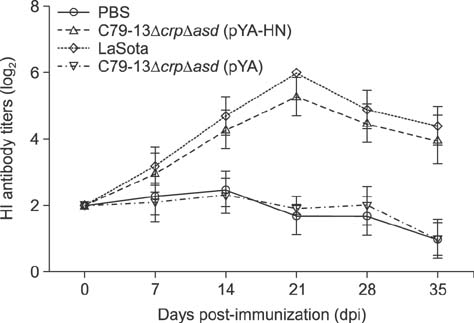
- Newcastle disease virus (NDV) and Salmonella Pullorum have significant damaging effects on the poultry industry, but no previous vaccine can protect poultry effectively. In this study, a recombinant-attenuated S. Pullorum strain secreting the NDV hemagglutinin-neuraminidase (HN) protein, C79-13ΔcrpΔasd (pYA-HN), was constructed by using the suicide plasmid pREasd-mediated bacteria homologous recombination method to form a new bivalent vaccine candidate against Newcastle disease (ND) and S. Pullorum disease (PD). The effect of this vaccine candidate was compared with those of the NDV LaSota and C79-13ΔcrpΔasd (pYA) strains. The serum hemagglutination inhibition antibody titers, serum immunoglobulin G (IgG) antibodies, secretory IgA, and stimulation index in lymphocyte proliferation were increased significantly more (p < 0.01) in chickens inoculated with C79-13ΔcrpΔasd (pYA-HN) than with C79-13ΔcrpΔasd (pYA) but were not significantly increased compared with the chickens immunized with the LaSota live vaccine (p > 0.05). Moreover, the novel strain provides 60% and 80% protective efficacy against the NDV virulent strain F48E9 and the S. Pullorum virulent strain C79-13. In summary, in this study, a recombinant-attenuated S. Pullorum strain secreting NDV HN protein was constructed. The generation of the S. Pullorum C79-13ΔcrpΔasd (pYA-HN) strain provides a foundation for the development of an effective living-vector double vaccine against ND and PD.
Keyword
MeSH Terms
Figure
Reference
-
1. Alexander DJ, Aldous EW, Fuller CM. The long view: a selective review of 40 years of Newcastle disease research. Avian Pathol. 2012; 41:329–335.
Article2. Asensi GF, de Sales NF, Dutra FF, Feijó DF, Bozza MT, Ulrich RG, Miyoshi A, de Morais K, Azevedo VA, Silva JT, Le Loir Y, Paschoalin VM. Oral immunization with Lactococcus lactis secreting attenuated recombinant staphylococcal enterotoxin B induces a protective immune response in a murine model. Microb Cell Fact. 2013; 12:32.3. Awais MM, Akhtar M, Muhammad F, ul Haq A, Anwar MI. Immunotherapeutic effects of some sugar cane (Saccharum officinarum L.) extracts against coccidiosis in industrial broiler chickens. Exp Parasitol. 2011; 128:104–110.
Article4. Awan MA, Otte MJ, James AD. The epidemiology of Newcastle disease in rural poultry: a review. Avian Pathol. 1994; 23:405–423.
Article5. Boursnell ME, Green PF, Samson AC, Campbell JI, Deuter A, Peters RW, Millar NS, Emmerson PT, Binns MM. A recombinant fowlpox virus expressing the hemagglutinin-neuraminidase gene of Newcastle disease virus (NDV) protects chickens against challenge by NDV. Virology. 1990; 178:297–300.
Article6. Briand FX, Massin P, Jestin V. Characterisation of a type 1 Avian Paramyxovirus belonging to a divergent group. Vet Microbiol. 2014; 168:25–33.
Article7. Castillo Alvarez AM, Vaquero-Vera A, Fonseca-Liñán R, Ruiz-Pérez F, Villegas-Sepúlveda N, Ortega-Pierres G. A prime-boost vaccination of mice with attenuated Salmonella expressing a 30-mer peptide from the Trichinella spiralis gp43 antigen. Vet Parasitol. 2013; 194:202–206.8. Cattoli G, Susta L, Terregino C, Brown C. Newcastle disease: a review of field recognition and current methods of laboratory detection. J Vet Diagn Invest. 2011; 23:637–656.9. Cornax I, Miller PJ, Afonso CL. Characterization of live LaSota vaccine strain-induced protection in chickens upon early challenge with a virulent Newcastle disease virus of heterologous genotype. Avian Dis. 2012; 56:464–470.
Article10. Cosset FL, Bouquet JF, Drynda A, Chebloune Y, Rey-Senelonge A, Kohen G, Nigon VM, Desmettre P, Verdier G. Newcastle disease virus (NDV) vaccine based on immunization with avian cells expressing the NDV hemagglutinin-neuraminidase glycoprotein. Virology. 1991; 185:862–866.
Article11. Fang WH, Liang XY. Expression of the newcastle disease virus fusion glycoprotein in vero cells using attenuated Salmonella typhimurium as transgenic carrier. Sheng Wu Hua Xue Yu Sheng Wu Wu Li Xue Bao (Shanghai). 2002; 34:488–493.12. Freytag LC, Clements JD. Mucosal adjuvants. Vaccine. 2005; 23:1804–1813.
Article13. Haan L, Verweij WR, Holtrop M, Brands R, van Scharrenburg GJ, Palache AM, Agsteribbe E, Wilschut J. Nasal or intramuscular immunization of mice with influenza subunit antigen and the B subunit of Escherichia coli heat-labile toxin induces IgA- or IgG-mediated protective mucosal immunity. Vaccine. 2001; 19:2898–2907.
Article14. Hird TR, Grassly NC. Systematic review of mucosal immunity induced by oral and inactivated poliovirus vaccines against virus shedding following oral poliovirus challenge. PLoS Pathog. 2012; 8:e1002599.
Article15. Holmgren J, Czerkinsky C. Mucosal immunity and vaccines. Nat Med. 2005; 11:4 Suppl. S45–S53.
Article16. Huang Y, Yang S, Hu B, Xu C, Gao D, Zhu M, Huang Q, Zhang L, Wu J, Zhang X, Khan MI. Genetic, pathogenic and antigenic diversity of Newcastle disease viruses in Shandong Province, China. Vet Microbiol. 2015; 180:237–244.
Article17. Jazayeri SD, Ideris A, Zakaria Z, Omar AR. Attenuated Salmonella typhimurium SV4089 as a potential carrier of oral DNA vaccine in chickens. J Biomed Biotechnol. 2012; 2012:264986.18. Jeon BW, Jawale CV, Kim SH, Lee JH. Attenuated Salmonella Gallinarum secreting an Escherichia coli heat-labile enterotoxin B subunit protein as an adjuvant for oral vaccination against fowl typhoid. Vet Immunol Immunopathol. 2012; 150:149–160.
Article19. Jeon BW, Nandre RM, Lee JH. Oral immunization with an attenuated Salmonella Gallinarum mutant as a fowl typhoid vaccine with a live adjuvant strain secreting the B subunit of Escherichia coli heat-labile enterotoxin. BMC Vet Res. 2013; 9:96.20. Kang HY, Srinivasan J, Curtiss R 3rd. Immune responses to recombinant pneumococcal PspA antigen delivered by live attenuated Salmonella enterica serovar Typhimurium vaccine. Infect Immun. 2002; 70:1739–1749.
Article21. Kumar S, Nayak B, Collins PL, Samal SK. Evaluation of the Newcastle disease virus F and HN proteins in protective immunity by using a recombinant avian paramyxovirus type 3 vector in chickens. J Virol. 2011; 85:6521–6534.
Article22. Lee LF, Heidari M, Zhang H, Lupiani B, Reddy SM, Fadly A. Cell culture attenuation eliminates rMd5ΔMeq-induced bursal and thymic atrophy and renders the mutant virus as an effective and safe vaccine against Marek's disease. Vaccine. 2012; 30:5151–5158.
Article23. Liu P, Wang ZQ, Liu RD, Jiang P, Long SR, Liu LN, Zhang XZ, Cheng XC, Yu C, Ren HJ, Cui J. Oral vaccination of mice with Trichinella spiralis nudix hydrolase DNA vaccine delivered by attenuated Salmonella elicited protective immunity. Exp Parasitol. 2015; 153:29–38.
Article24. Matsuda K, Chaudhari AA, Lee JH. Evaluation of safety and protection efficacy on cpxR and lon deleted mutant of Salmonella Gallinarum as a live vaccine candidate for fowl typhoid. Vaccine. 2011; 29:668–674.
Article25. Nagai Y, Klenk HD, Rott R. Proteolytic cleavage of the viral glycoproteins and its significance for the virulence of Newcastle disease virus. Virology. 1976; 72:494–508.
Article26. Nandre RM, Chaudhari AA, Matsuda K, Lee JH. Immunogenicity of a Salmonella Enteritidis mutant as vaccine candidate and its protective efficacy against salmonellosis in chickens. Vet Immunol Immunopathol. 2011; 144:299–311.
Article27. Nandre RM, Jawale CV, Lee JH. Enhanced protective immune responses against Salmonella Enteritidis infection by Salmonella secreting an Escherichia coli heat-labile enterotoxin B subunit protein. Comp Immunol Microbiol Infect Dis. 2013; 36:537–548.
Article28. Pan ZM, Huang JL, Cheng NN, Cui YC, You M, Tang LH, Zhang XM, Jiao XA, Liu XF. [Construction and immunogenicity of attenuated Salmonella typhimurium stably harbouring DNA vaccine against Newcastle disease virus]. Bing Du Xue Bao. 2008; 24:41–46. Chinese.29. Poles J, Alvarez Y, Hioe CE. Induction of intestinal immunity by mucosal vaccines as a means of controlling HIV infection. AIDS Res Hum Retroviruses. 2014; 30:1027–1040.
Article30. Rahman MM, Uyangaa E, Han YW, Kim SB, Kim JH, Choi JY, Eo SK. Oral co-administration of live attenuated Salmonella enterica serovar Typhimurium expressing chicken interferon-α and interleukin-18 enhances the alleviation of clinical signs caused by respiratory infection with avian influenza virus H9N2. Vet Microbiol. 2012; 157:448–455.
Article31. Rahman MM, Uyangaa E, Han YW, Kim SB, Kim JH, Choi JY, Yoo DJ, Hong JT, Han SB, Kim B, Kim K, Eo SK. Oral administration of live attenuated Salmonella enterica serovar Typhimurium expressing chicken interferon-α alleviates clinical signs caused by respiratory infection with avian influenza virus H9N2. Vet Microbiol. 2011; 154:140–151.
Article32. Sawant PM, Verma PC, Subudhi PK, Chaturvedi U, Singh M, Kumar R, Tiwari AK. Immunomodulation of bivalent Newcastle disease DNA vaccine induced immune response by co-delivery of chicken IFN-γ and IL-4 genes. Vet Immunol Immunopathol. 2011; 144:36–44.
Article33. Susta L, Jones ME, Cattoli G, Cardenas-Garcia S, Miller PJ, Brown CC, Afonso CL. Pathologic characterization of genotypes XIV and XVII Newcastle disease viruses and efficacy of classical vaccination on specific pathogen-free birds. Vet Pathol. 2015; 52:120–131.
Article34. Wang C, Li X, Zhang C, Wu T, Li Y, Cheng X. A eukaryotic expression plasmid carrying chicken interleukin-18 enhances the response to newcastle disease virus vaccine. Clin Vaccine Immunol. 2015; 22:56–64.
Article35. Wang L, Wang X, Bi K, Sun X, Yang J, Gu Y, Huang J, Zhan B, Zhu X. Oral vaccination with attenuated Salmonella typhimurium-delivered TsPmy DNA vaccine elicits protective immunity against Trichinella spiralis in BALB/c mice. PLoS Negl Trop Dis. 2016; 10:e0004952.36. Yang X, Zhou Y, Li J, Fu L, Ji G, Zeng F, Zhou L, Gao W, Wang H. Recombinant infectious bronchitis virus (IBV) H120 vaccine strain expressing the hemagglutinin-neuraminidase (HN) protein of Newcastle disease virus (NDV) protects chickens against IBV and NDV challenge. Arch Virol. 2016; 161:1209–1216.
Article37. Zhang Y, Zhang X, Liao X, Huang X, Cao S, Wen X, Wen Y, Wu R, Liu W. Construction of a bivalent DNA vaccine co-expressing S genes of transmissible gastroenteritis virus and porcine epidemic diarrhea virus delivered by attenuated Salmonella typhimurium. Virus Genes. 2016; 52:354–364.
Article38. Zhao Z, Xu Y, Wu B, Cheng X, Li Y, Tang X, Zhang C, Chen H. [Characterization of attenuated Salmonella C500 strain with a delta asd mutant and use as an asd+ balanced-lethal host-vector system]. Sheng Wu Gong Cheng Xue Bao. 2009; 25:29–36. Chinese.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Characterisation of genotype VII Newcastle disease virus (NDV) isolated from NDV vaccinated chickens, and the efficacy of LaSota and recombinant genotype VII vaccines against challenge with velogenic NDV
- Preparation and diagnostic utility of a hemagglutination inhibition test antigen derived from the baculovirus-expressed hemagglutinin-neuraminidase protein gene of Newcastle disease virus
- Protection of chickens from Newcastle disease with a recombinant baculovirus subunit vaccine expressing the fusion and hemagglutininneuraminidase proteins
- Newcastle disease virus vectored vaccines as bivalent or antigen delivery vaccines
- Protective efficacy of commercial inactivated Newcastle disease virus vaccines in chickens against a recent Korean epizootic strain