Endocrinol Metab.
2017 Dec;32(4):451-459. 10.3803/EnM.2017.32.4.451.
Effects of Single Vitamin D₃ Injection (200,000 Units) on Serum Fibroblast Growth Factor 23 and Sclerostin Levels in Subjects with Vitamin D Deficiency
- Affiliations
-
- 1Brain Korea 21 PLUS Project for Medical Science, Yonsei University, Seoul, Korea. lsk@yuhs.ac
- 2Division of Endocrinology and Metabolism, Department of Internal Medicine, Yantai Affiliated Hospital of Binzhou Medical University, Yantai, China.
- 3Division of Endocrinology and Endocrine Research Institute, Department of Internal Medicine, Yonsei University College of Medicine, Seoul, Korea.
- 4Division of Endocrinology and Metabolism, Department of Internal Medicine, Dongguk University Ilsan Hospital, Dongguk University College of Medicine, Goyang, Korea.
- 5Division of Endocrinology and Metabolism, Department of Internal Medicine, Ajou University School of Medicine, Suwon, Korea.
- KMID: 2406189
- DOI: http://doi.org/10.3803/EnM.2017.32.4.451
Abstract
- BACKGROUND
Vitamin D deficiency remains common in all age groups and affects skeletal and non-skeletal health. Fibroblast growth factor 23 is a bone-derived hormone that regulates phosphate and 1,25-dihydroxyvitamin D homeostasis as a counter regulatory factor. 1,25-Dihydroxyvitamin D stimulates fibroblast growth factor 23 synthesis in bone, while fibroblast growth factor 23 suppresses 1,25-dihydroxyvitamin D production in the kidney. The aim of this study was to evaluate the effects of vitamin D₃ intramuscular injection therapy on serum fibroblast growth factor 23 concentrations, and several other parameters associated with bone metabolism such as sclerostin, dickkopf-1, and parathyroid hormone.
METHODS
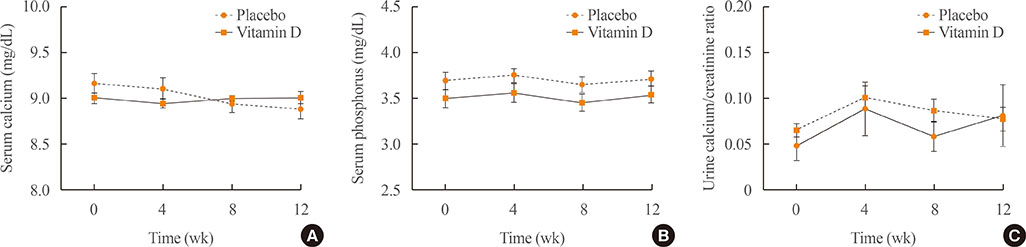
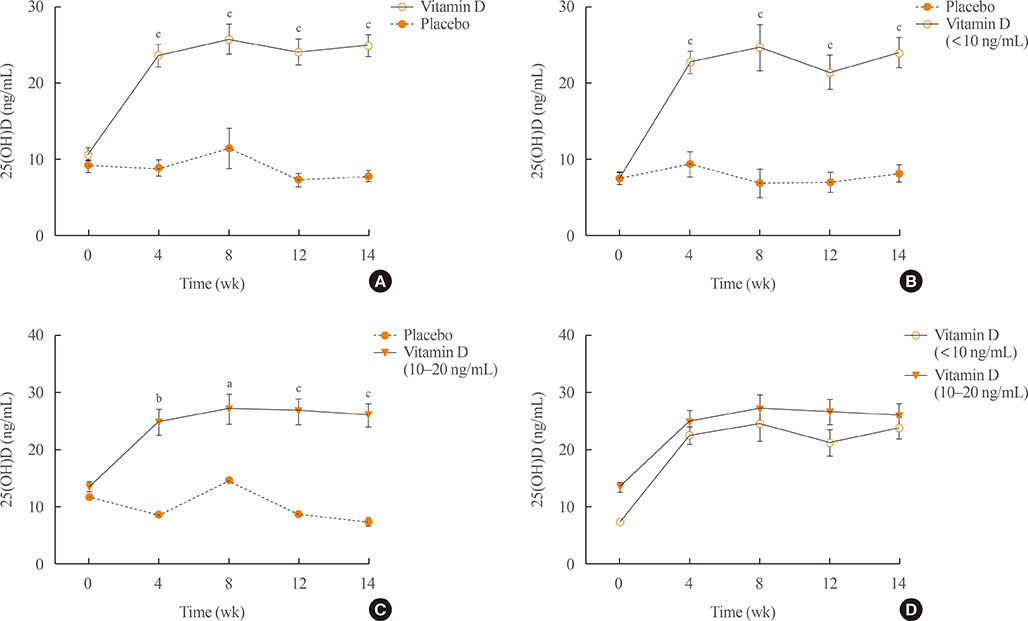
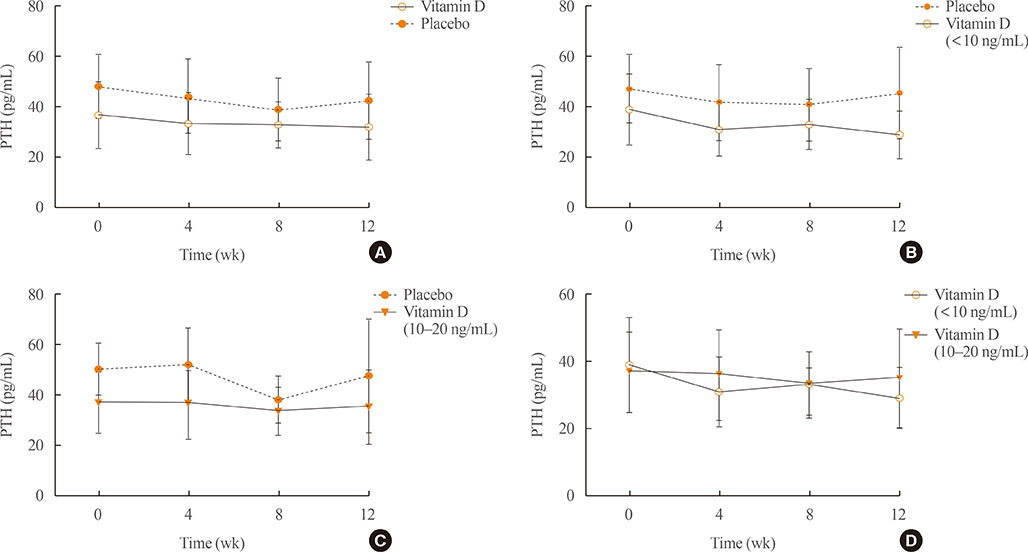
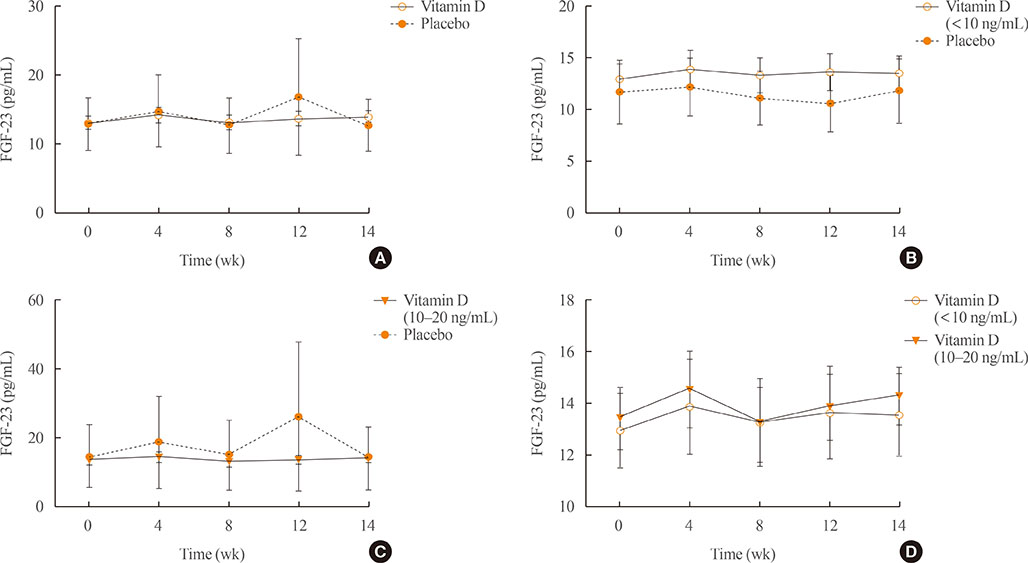
A total of 34 subjects with vitamin D deficiency (defined by serum 25-hydroxyvitamin D levels below 20 ng/mL) were randomly assigned to either the vitamin D injection group (200,000 units) or placebo treatment group. Serum calcium, phosphate, urine calcium/creatinine, serum 25-hydroxyvitamin D, fibroblast growth factor 23, sclerostin, parathyroid hormone, and dickkopf-1 levels were serially measured after treatment.
RESULTS
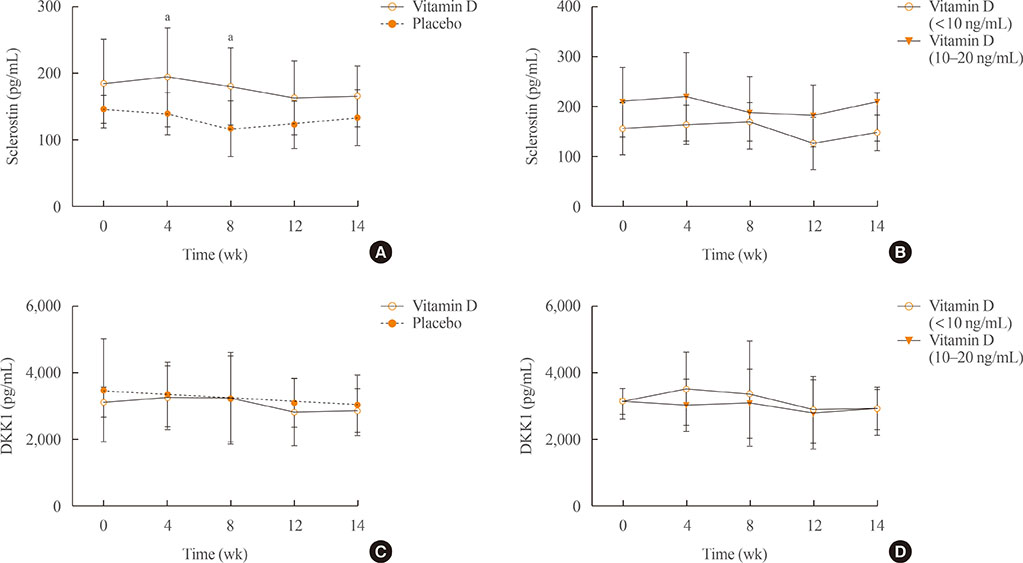
Comparing the vitamin D injection group with the placebo group, no significant changes were observed in serum fibroblast growth factor 23, parathyroid hormone, or dickkopf-1 levels. Serum sclerostin concentrations transiently increased at week 4 in the vitamin D group. However, these elevated levels declined later and there were no statistically significant differences as compared with baseline levels.
CONCLUSION
Serum fibroblast factor 23, sclerostin, parathyroid hormone, and dickkopf-1 levels were not affected significantly by single intramuscular injection of vitamin D₃.
MeSH Terms
Figure
Cited by 1 articles
-
Vitamin D Deficiency at Mid-Pregnancy Is Associated with a Higher Risk of Postpartum Glucose Intolerance in Women with Gestational Diabetes Mellitus
Kyung-Soo Kim, Seok Won Park, Yong-Wook Cho, Soo-Kyung Kim
Endocrinol Metab. 2020;35(1):97-105. doi: 10.3803/EnM.2020.35.1.97.
Reference
-
1. Hofbauer LC, Dunstan CR, Spelsberg TC, Riggs BL, Khosla S. Osteoprotegerin production by human osteoblast lineage cells is stimulated by vitamin D, bone morphogenetic protein-2, and cytokines. Biochem Biophys Res Commun. 1998; 250:776–781.2. Lin J, Manson JE, Lee IM, Cook NR, Buring JE, Zhang SM. Intakes of calcium and vitamin D and breast cancer risk in women. Arch Intern Med. 2007; 167:1050–1059.3. Kamen D, Aranow C. Vitamin D in systemic lupus erythematosus. Curr Opin Rheumatol. 2008; 20:532–537.4. Judd SE, Tangpricha V. Vitamin D deficiency and risk for cardiovascular disease. Am J Med Sci. 2009; 338:40–44.5. Choi HS, Kim KA, Lim CY, Rhee SY, Hwang YC, Kim KM, et al. Low serum vitamin D is associated with high risk of diabetes in Korean adults. J Nutr. 2011; 141:1524–1528.6. Yu S, Fang H, Han J, Cheng X, Xia L, Li S, et al. The high prevalence of hypovitaminosis D in China: a multicenter vitamin D status survey. Medicine (Baltimore). 2015; 94:e585.7. Rizzoli R, Eisman JA, Norquist J, Ljunggren O, Krishnarajah G, Lim SK, et al. Risk factors for vitamin D inadequacy among women with osteoporosis: an international epidemiological study. Int J Clin Pract. 2006; 60:1013–1019.8. Anderson PH, May BK, Morris HA. Vitamin D metabolism: new concepts and clinical implications. Clin Biochem Rev. 2003; 24:13–26.9. Jurutka PW, Bartik L, Whitfield GK, Mathern DR, Barthel TK, Gurevich M, et al. Vitamin D receptor: key roles in bone mineral pathophysiology, molecular mechanism of action, and novel nutritional ligands. J Bone Miner Res. 2007; 22:Suppl 2. V2–V10.10. Heikinheimo RJ, Inkovaara JA, Harju EJ, Haavisto MV, Kaarela RH, Kataja JM, et al. Annual injection of vitamin D and fractures of aged bones. Calcif Tissue Int. 1992; 51:105–110.11. Shimada T, Hasegawa H, Yamazaki Y, Muto T, Hino R, Takeuchi Y, et al. FGF-23 is a potent regulator of vitamin D metabolism and phosphate homeostasis. J Bone Miner Res. 2004; 19:429–435.12. Liu S, Tang W, Zhou J, Stubbs JR, Luo Q, Pi M, et al. Fibroblast growth factor 23 is a counter-regulatory phosphaturic hormone for vitamin D. J Am Soc Nephrol. 2006; 17:1305–1315.13. Seiler S, Reichart B, Roth D, Seibert E, Fliser D, Heine GH. FGF-23 and future cardiovascular events in patients with chronic kidney disease before initiation of dialysis treatment. Nephrol Dial Transplant. 2010; 25:3983–3989.14. Faul C, Amaral AP, Oskouei B, Hu MC, Sloan A, Isakova T, et al. FGF23 induces left ventricular hypertrophy. J Clin Invest. 2011; 121:4393–4408.15. Nasrallah MM, El-Shehaby AR, Salem MM, Osman NA, El Sheikh E, Sharaf El Din UA. Fibroblast growth factor-23 (FGF-23) is independently correlated to aortic calcification in haemodialysis patients. Nephrol Dial Transplant. 2010; 25:2679–2685.16. Turner C, Dalton N, Inaoui R, Fogelman I, Fraser WD, Hampson G. Effect of a 300,000-IU loading dose of ergocalciferol (vitamin D2) on circulating 1,25(OH)2-vitamin D and fibroblast growth factor-23 (FGF-23) in vitamin D insufficiency. J Clin Endocrinol Metab. 2013; 98:550–556.17. Atkins GJ, Rowe PS, Lim HP, Welldon KJ, Ormsby R, Wijenayaka AR, et al. Sclerostin is a locally acting regulator of late-osteoblast/preosteocyte differentiation and regulates mineralization through a MEPE-ASARM-dependent mechanism. J Bone Miner Res. 2011; 26:1425–1436.18. Morvan F, Boulukos K, Clement-Lacroix P, Roman Roman S, Suc-Royer I, Vayssiere B, et al. Deletion of a single allele of the Dkk1 gene leads to an increase in bone formation and bone mass. J Bone Miner Res. 2006; 21:934–945.19. Sankaralingam A, Roplekar R, Turner C, Dalton RN, Hampson G. Changes in dickkopf-1 (DKK1) and sclerostin following a loading dose of vitamin D 2 (300,000 IU). J Osteoporos. 2014; 2014:682763.20. Acibucu F, Dokmetas HS, Acibucu DO, Kilicli F, Aydemir M, Cakmak E. Effect of vitamin D treatment on serum sclerostin level. Exp Clin Endocrinol Diabetes. 2017; 125:634–637.21. Chung YS, Chung DJ, Kang MI, Kim IJ, Koh JM, Min YK, et al. Vitamin D repletion in Korean postmenopausal women with osteoporosis. Yonsei Med J. 2016; 57:923–927.22. Burns J, Paterson CR. Single dose vitamin D treatment for osteomalacia in the elderly. Br Med J (Clin Res Ed). 1985; 290:281–282.23. Saito H, Maeda A, Ohtomo S, Hirata M, Kusano K, Kato S, et al. Circulating FGF-23 is regulated by 1alpha,25-dihydroxyvitamin D3 and phosphorus in vivo. J Biol Chem. 2005; 280:2543–2549.24. Uzum AK, Salman S, Telci A, Boztepe H, Tanakol R, Alagol F, et al. Effects of vitamin D replacement therapy on serum FGF23 concentrations in vitamin D-deficient women in short term. Eur J Endocrinol. 2010; 163:825–831.25. Wolf M, Koch TA, Bregman DB. Effects of iron deficiency anemia and its treatment on fibroblast growth factor 23 and phosphate homeostasis in women. J Bone Miner Res. 2013; 28:1793–1803.26. Wolf M, White KE. Coupling fibroblast growth factor 23 production and cleavage: iron deficiency, rickets, and kidney disease. Curr Opin Nephrol Hypertens. 2014; 23:411–419.27. Romagnoli E, Mascia ML, Cipriani C, Fassino V, Mazzei F, D'Erasmo E, et al. Short and long-term variations in serum calciotropic hormones after a single very large dose of ergocalciferol (vitamin D2) or cholecalciferol (vitamin D3) in the elderly. J Clin Endocrinol Metab. 2008; 93:3015–3020.28. Gatti D, Viapiana O, Fracassi E, Idolazzi L, Dartizio C, Povino MR, et al. Sclerostin and DKK1 in postmenopausal osteoporosis treated with denosumab. J Bone Miner Res. 2012; 27:2259–2263.29. Gatti D, Viapiana O, Adami S, Idolazzi L, Fracassi E, Rossini M. Bisphosphonate treatment of postmenopausal osteoporosis is associated with a dose dependent increase in serum sclerostin. Bone. 2012; 50:739–742.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Efficacy and Safety of Intramuscular Injections of Vitamin D3 B.O.N. on Serum Vitamin D Levels in Adults with Vitamin D Deficiency: A Prospective, Single-Center, Open-Label Study
- Efficacy and safety of vitamin D3 B.O.N intramuscular injection in Korean adults with vitamin D deficiency
- Vitamin B12 Deficiency after a Total Gastrectomy in Patients with Gastric Cancer
- Vitamin D Deficiency Rickets
- Gestational Diabetes Mellitus, Fetal Growth and Vitamin D