Korean J Physiol Pharmacol.
2016 Mar;20(2):139-145. 10.4196/kjpp.2016.20.2.139.
Myosin VI contributes to malignant proliferation of human glioma cells
- Affiliations
-
- 1Neurosurgical Department of Huashan Hospital, Fudan University, Shanghai 200040, China. dr_zhongping@163.com
- 2Neurosurgical Department of Huadong Hospital, Fudan University, Shanghai 200040, China.
- KMID: 2388669
- DOI: http://doi.org/10.4196/kjpp.2016.20.2.139
Abstract
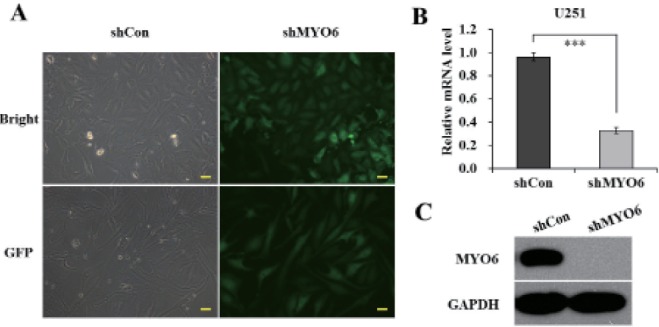
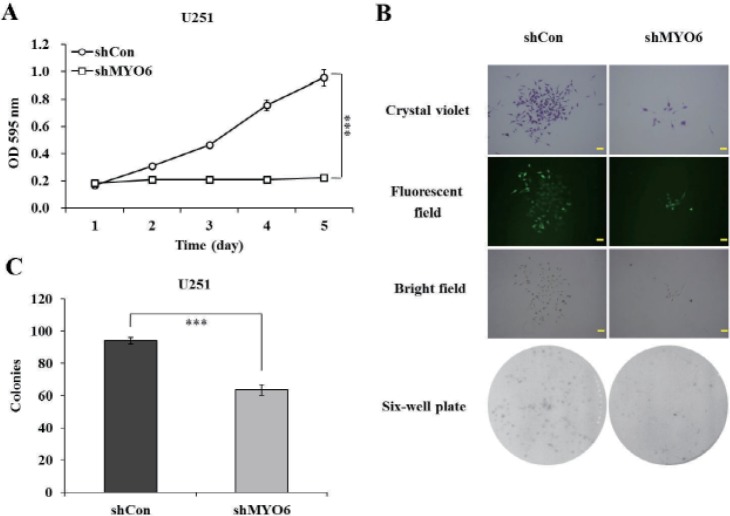
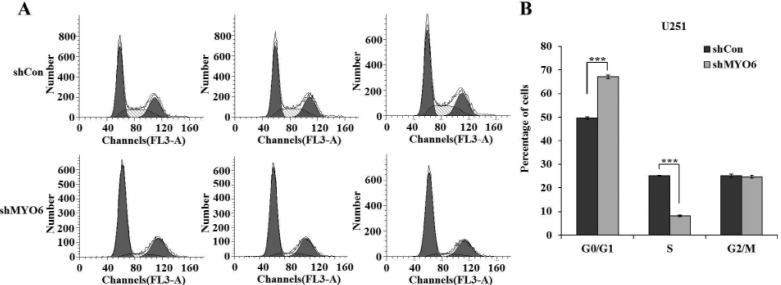
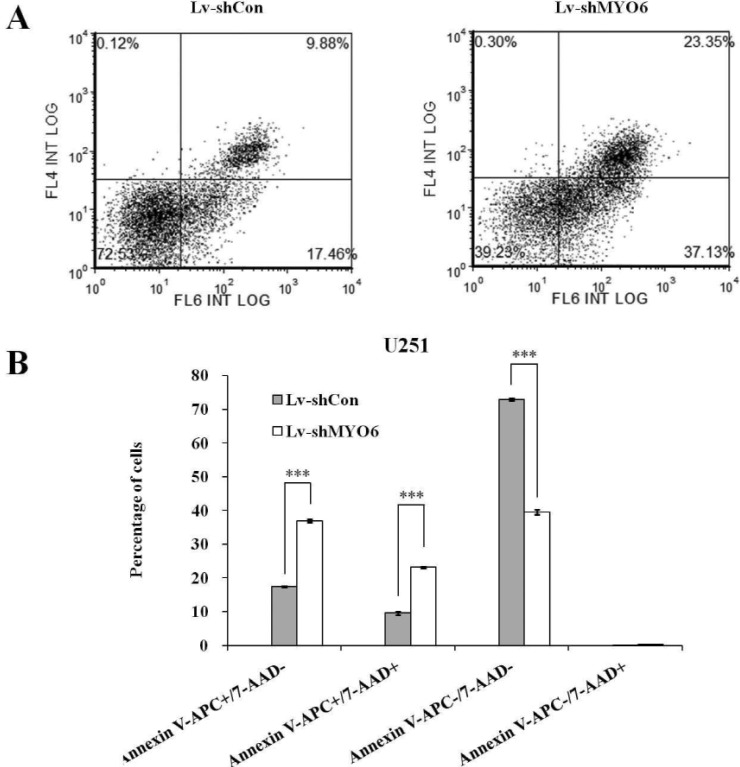
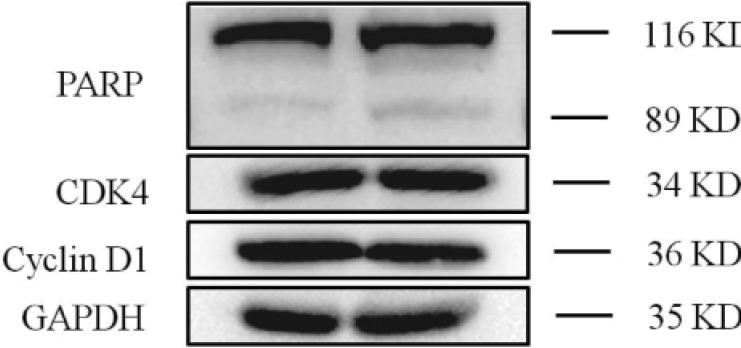
- Previously characterized as a backward motor, myosin VI (MYO6), which belongs to myosin family, moves toward the minus end of the actin track, a direction opposite to all other known myosin members. Recent researches have illuminated the role of MYO6 in human cancers, particularly in prostate cancer. However, the role of MYO6 in glioma has not yet been determined. In this study, to explore the role of MYO6 in human glioma, lentivirus-delivered short hairpin RNA (shRNA) targeting MYO6 was designed to stably down-regulate its endogenous expression in glioblastoma cells U251. Knockdown of MYO6 signifi cantly inhibited viability and proliferation of U251 cells in vitro. Moreover, the cell cycle of U251 cells was arrested at G0/G1 phase with the absence of MYO6, which could contribute to the suppression of cell proliferation. In conclusion, we firstly identified the crucial involvement of MYO6 in human glioma. The inhibition of MYO6 by shRNA might be a potential therapeutic method in human glioma.
Keyword
MeSH Terms
Figure
Reference
-
1. Annibali D, Whitfield JR, Favuzzi E, Jauset T, Serrano E, Cuartas I, Redondo-Campos S, Folch G, Gonzàlez-Juncà A, Sodir NM, Massó-Vallés D, Beaulieu ME, Swigart LB, Mc Gee MM, Somma MP, Nasi S, Seoane J, Evan GI, Soucek L. Myc inhibition is effective against glioma and reveals a role for Myc in proficient mitosis. Nat Commun. 2014; 5:4632. PMID: 25130259.
Article2. Huse JT, Holland EC. Targeting brain cancer: advances in the molecular pathology of malignant glioma and medulloblastoma. Nat Rev Cancer. 2010; 10:319–331. PMID: 20414201.
Article3. Constantin A, Elkhaled A, Jalbert L, Srinivasan R, Cha S, Chang SM, Bajcsy R, Nelson SJ. Identifying malignant transformations in recurrent low grade gliomas using high resolution magic angle spinning spectroscopy. Artif Intell Med. 2012; 55:61–70. PMID: 22387185.
Article4. Stupp R, Hegi ME, Mason WP, van den Bent MJ, Taphoorn MJ, Janzer RC, Ludwin SK, Allgeier A, Fisher B, Belanger K, Hau P, Brandes AA, Gijtenbeek J, Marosi C, Vecht CJ, Mokhtari K, Wesseling P, Villa S, Eisenhauer E, Gorlia T, Weller M, Lacombe D, Cairncross JG, Mirimanoff RO. European organisation for research and treatment of cancer brain tumour and radiation oncology groups. national cancer institute of Canada clinical trials group. Effects of radiotherapy with concomitant and adjuvant temozolomide versus radiotherapy alone on survival in glioblastoma in a randomised phase III study: 5-year analysis of the EORTCNCIC trial. Lancet Oncol. 2009; 10:459–466. PMID: 19269895.
Article5. Mirimanoff RO. High-grade gliomas: reality and hopes. Chin J Cancer. 2014; 33:1–3. PMID: 24384235.
Article6. Qiu ZK, Shen D, Chen YS, Yang QY, Guo CC, Feng BH, Chen ZP. Enhanced MGMT expression contributes to temozolomide resistance in glioma stem-like cells. Chin J Cancer. 2014; 33:115–122. PMID: 23958055.
Article7. Seog DH, Han J. Sorting nexin 17 interacts directly with kinesin superfamily KIF1Bbeta protein. Korean J Physiol Pharmacol. 2008; 12:199–204. PMID: 19967056.8. Hartman MA, Finan D, Sivaramakrishnan S, Spudich JA. Principles of unconventional myosin function and targeting. Annu Rev Cell Dev Biol. 2011; 27:133–155. PMID: 21639800.
Article9. Wells AL, Lin AW, Chen LQ, Safer D, Cain SM, Hasson T, Carragher BO, Milligan RA, Sweeney HL. Myosin VI is an actin-based motor that moves backwards. Nature. 1999; 401:505–508. PMID: 10519557.
Article10. Buss F, Spudich G, Kendrick-Jones J. Myosin VI: cellular functions and motor properties. Annu Rev Cell Dev Biol. 2004; 20:649–676. PMID: 15473855.
Article11. Friedman TB, Griffith AJ. Human nonsyndromic sensorineural deafness. Annu Rev Genomics Hum Genet. 2003; 4:341–402. PMID: 14527306.
Article12. Buss F, Kendrick-Jones J. How are the cellular functions of myosin VI regulated within the cell? Biochem Biophys Res Commun. 2008; 369:165–175. PMID: 18068125.
Article13. Liao YW, Wu XM, Jia J, Wu XL, Hong T, Meng LX, Wu XY. Myosin VI contributes to maintaining epithelial barrier function. J Biomed Sci. 2013; 20:68. PMID: 24028494.
Article14. Self T, Sobe T, Copeland NG, Jenkins NA, Avraham KB, Steel KP. Role of myosin VI in the differentiation of cochlear hair cells. Dev Biol. 1999; 214:331–341. PMID: 10525338.
Article15. Ahmed ZM, Morell RJ, Riazuddin S, Gropman A, Shaukat S, Ahmad MM, Mohiddin SA, Fananapazir L, Caruso RC, Husnain T, Khan SN, Riazuddin S, Griffith AJ, Friedman TB, Wilcox ER. Mutations of MYO6 are associated with recessive deafness, DFNB37. Am J Hum Genet. 2003; 72:1315–1322. PMID: 12687499.
Article16. Melchionda S, Ahituv N, Bisceglia L, Sobe T, Glaser F, Rabionet R, Arbones ML, Notarangelo A, Di Iorio E, Carella M, Zelante L, Estivill X, Avraham KB, Gasparini P. MYO6, the human homologue of the gene responsible for deafness in Snell's waltzer mice, is mutated in autosomal dominant nonsyndromic hearing loss. Am J Hum Genet. 2001; 69:635–640. PMID: 11468689.
Article17. Szczyrba J, Löprich E, Wach S, Jung V, Unteregger G, Barth S, Grobholz R, Wieland W, Stöhr R, Hartmann A, Wullich B, Grässer F. The microRNA profile of prostate carcinoma obtained by deep sequencing. Mol Cancer Res. 2010; 8:529–538. PMID: 20353999.
Article18. Wei S, Dunn TA, Isaacs WB, De Marzo AM, Luo J. GOLPH2 and MYO6: putative prostate cancer markers localized to the Golgi apparatus. Prostate. 2008; 68:1387–1395. PMID: 18543251.
Article19. Demichelis F, Setlur SR, Beroukhim R, Perner S, Korbel JO, Lafargue CJ, Pflueger D, Pina C, Hofer MD, Sboner A, Svensson MA, Rickman DS, Urban A, Snyder M, Meyerson M, Lee C, Gerstein MB, Kuefer R, Rubin MA. Distinct genomic aberrations associated with ERG rearranged prostate cancer. Genes Chromosomes Cancer. 2009; 48:366–380. PMID: 19156837.20. Järvinen AK, Autio R, Kilpinen S, Saarela M, Leivo I, Grénman R, Mäkitie AA, Monni O. High-resolution copy number and gene expression microarray analyses of head and neck squamous cell carcinoma cell lines of tongue and larynx. Genes Chromosomes Cancer. 2008; 47:500–509. PMID: 18314910.
Article21. Tamaki K, Kamakura M, Nakamichi N, Taniura H, Yoneda Y. Upregulation of Myo6 expression after traumatic stress in mouse hippocampus. Neurosci Lett. 2008; 433:183–187. PMID: 18261850.
Article22. Takarada T, Kou M, Nakamichi N, Ogura M, Ito Y, Fukumori R, Kokubo H, Acosta GB, Hinoi E, Yoneda Y. Myosin VI reduces proliferation, but not differentiation, in pluripotent P19 cells. PLoS One. 2013; 8:e63947. PMID: 23691122.
Article23. Lafaurie-Janvore J. Temporal regulation of abscission, the last step of cell division. Biol Aujourdhui. 2013; 207:133–148. PMID: 24103343.24. Sharma T, Kumari P, Pincha N, Mutukula N, Saha S, Jana SS, Ta M. Inhibition of non-muscle myosin II leads to G0/G1 arrest of Wharton's jelly-derived mesenchymal stromal cells. Cytotherapy. 2014; 16:640–652. PMID: 24210786.
Article25. Cameron RS, Liu C, Pihkala JP. Myosin 16 levels fluctuate during the cell cycle and are downregulated in response to DNA replication stress. Cytoskeleton (Hoboken). 2013; 70:328–348. PMID: 23596177.
Article27. Dunn TA, Chen S, Faith DA, Hicks JL, Platz EA, Chen Y, Ewing CM, Sauvageot J, Isaacs WB, De Marzo AM, Luo J. A novel role of myosin VI in human prostate cancer. Am J Pathol. 2006; 169:1843–1854. PMID: 17071605.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Retraction notice to: Myosin VI contributes to malignant proliferation of human glioma cells
- Inhibitory Effects of Toxoplasma Antigen on Proliferation and Invasion of Human Glioma Cells
- The PPARgamma Agonist Rosiglitazone Inhibits Glioma Cell Proliferation and Migration in vitro and Glioma Tumor Growth in vivo
- Tetrandrine Exerts a Radiosensitization Effect on Human Glioma through Inhibiting Proliferation by Attenuating ERK Phosphorylation
- Differential Effect of Genistein and Calphostin C on Phospholipase C Activation and Cell Proliferation in T98G Human Glioblastoma and Hs 683 Human Glioma Cells