Infect Chemother.
2017 Jun;49(2):135-139. 10.3947/ic.2017.49.2.135.
Comparison of Plasma Concentrations of Posaconazole with the Oral Suspension and Tablet in Korean Patients with Hematologic Malignancies
- Affiliations
-
- 1Department of Internal Medicine, Seoul National University College of Medicine and Seoul National University Hospital, Seoul, Korea. wbpark1@snu.ac.kr
- 2Department of Clinical Pharmacology and Therapeutics, Seoul National University College of Medicine and Seoul National University Hospital, Seoul, Korea.
- 3Department of Internal Medicine, Seoul National University College of Medicine and Seoul National University Bundang Hospital, Seongnam, Korea.
- 4Department of Laboratory Medicine, Seoul National University College of Medicine and Seoul National University Hospital, Seoul, Korea.
- KMID: 2383935
- DOI: http://doi.org/10.3947/ic.2017.49.2.135
Abstract
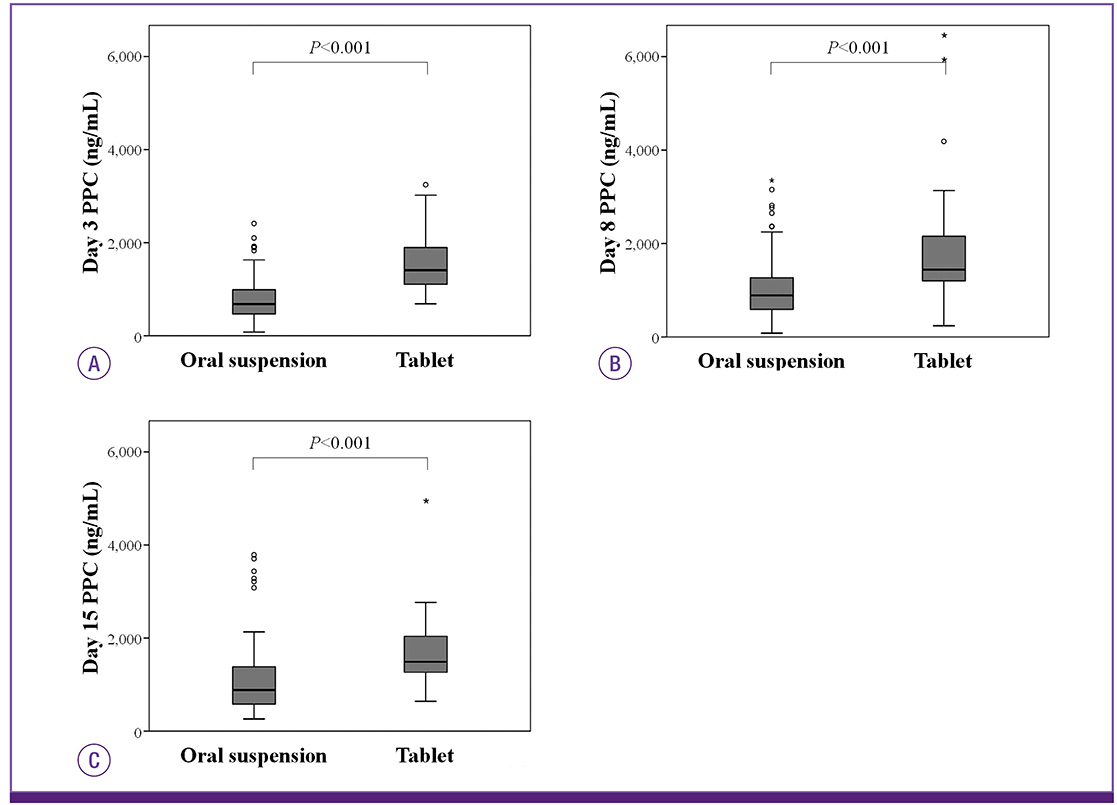
- The posaconazole tablet formulation was developed to have improved bioavailability compared to the oral suspension. Here, we compared posaconazole plasma concentration (PPC) with the posaconazole oral suspension versus the tablet in Korean patients undergoing remission induction chemotherapy for hematologic malignancies. PPC was measured at 3, 8, and 15 days of treatment with the oral suspension (174 patients) or the tablet (40 patients). At all time-points, mean PPC was significantly higher with the tablet compared to the oral suspension. Our findings suggest that posaconazole tablets generate an optimal PPC earlier and in more patients than the oral suspension among Korean patients.
Keyword
MeSH Terms
Figure
Cited by 1 articles
-
Advances in prophylaxis and treatment of invasive fungal infections: perspectives on hematologic diseases
Hyojin Ahn, Raeseok Lee, Sung-Yeon Cho, Dong-Gun Lee
Blood Res. 2022;57(S1):101-111. doi: 10.5045/br.2022.2022036.
Reference
-
1. Freifeld AG, Bow EJ, Sepkowitz KA, Boeckh MJ, Ito JI, Mullen CA, Raad II, Rolston KV, Young JA, Wingard JR; Infectious Diseases Society of America. Clinical practice guideline for the use of antimicrobial agents in neutropenic patients with cancer: 2010 update by the Infectious Diseases Society of America. Clin Infect Dis. 2011; 52:e56–e93.
Article2. Cornely OA, Maertens J, Winston DJ, Perfect J, Ullmann AJ, Walsh TJ, Helfgott D, Holowiecki J, Stockelberg D, Goh YT, Petrini M, Hardalo C, Suresh R, Angulo-Gonzalez D. Posaconazole vs. fluconazole or itraconazole prophylaxis in patients with neutropenia. N Engl J Med. 2007; 356:348–359.
Article3. Torres HA, Hachem RY, Chemaly RF, Kontoyiannis DP, Raad II. Posaconazole: a broad-spectrum triazole antifungal. Lancet Infect Dis. 2005; 5:775–785.
Article4. Chae H, Cho SY, Yu H, Cha K, Lee S, Kim M, Kim Y, Kim YJ, Kim HJ, Lee DG. Determination of posaconazole concentration with LC-MS/MS in adult patients with hematologic malignancy. Clin Chim Acta. 2015; 450:220–226.
Article5. Dolton MJ, Ray JE, Chen SC, Ng K, Pont L, McLachlan AJ. Multicenter study of posaconazole therapeutic drug monitoring: exposure-response relationship and factors affecting concentration. Antimicrob Agents Chemother. 2012; 56:5503–5510.
Article6. Hoenigl M, Raggam RB, Salzer HJ, Valentin T, Valentin A, Zollner-Schwetz I, Strohmeier AT, Seeber K, Wölfler A, Sill H, Krause R. Posaconazole plasma concentrations and invasive mould infections in patients with haematological malignancies. Int J Antimicrob Agents. 2012; 39:510–513.
Article7. Kang SH, Kim HS, Bae MN, Kim J, Yoo JY, Lee KY, Lee DG, Kim HJ. Fatal breakthrough mucormycosis in an acute myelogenous leukemia patient while on posaconazole prophylaxis. Infect Chemother. 2015; 47:49–54.
Article8. Guarascio AJ, Slain D. Review of the new delayed-release oral tablet and intravenous dosage forms of posaconazole. Pharmacotherapy. 2015; 35:208–219.
Article9. McKeage K. Posaconazole: a review of the gastro-resistant tablet and intravenous solution in invasive fungal infections. Drugs. 2015; 75:397–406.
Article10. Krishna G, Ma L, Martinho M, Preston RA, O'mara E. A new solid oral tablet formulation of posaconazole: a randomized clinical trial to investigate rising single-and multiple-dose pharmacokinetics and safety in healthy volunteers. J Antimicrob Chemother. 2012; 67:2725–2730.
Article11. Park WB, Cho JY, Park SI, Kim EJ, Yoon S, Yoon SH, Lee JO, Koh Y, Song KH, Choe PG, Yu KS, Kim ES, Bang SM, Kim NJ, Kim I, Oh MD, Kim HB, Song SH. Effectiveness of increasing the frequency of posaconazole syrup administration to achieve optimal plasma concentrations in patients with haematological malignancy. Int J Antimicrob Agents. 2016; 48:106–110.
Article12. Jang SH, Colangelo PM, Gobburu JV. Exposure-response of posaconazole used for prophylaxis against invasive fungal infections: evaluating the need to adjust doses based on drug concentrations in plasma. Clin Pharmacol Ther. 2010; 88:115–119.
Article13. Ashbee HR, Barnes RA, Johnson EM, Richardson MD, Gorton R, Hope WW. Therapeutic drug monitoring (TDM) of antifungal agents: guidelines from the British Society for Medical Mycology. J Antimicrob Chemother. 2014; 69:1162–1176.
Article14. Wiederhold NP. Pharmacokinetics and safety of posaconazole delayed-release tablets for invasive fungal infections. Clin Pharmacol. 2015; 8:1–8.
Article15. US Department of Health and Human Services. Common terminology criteria for adverse events (CTCAE) version 4.0. NIH publication No. 09-5410. National Cancer Institute;2009.16. Moton A, Krishna G, Wang Z. Tolerability and safety profile of posaconazole: evaluation of 18 controlled studies in healthy volunteers. J Clin Pharm Ther. 2009; 34:301–311.
Article17. Catanzaro A, Cloud GA, Stevens DA, Levine BE, Williams PL, Johnson RH, Rendon A, Mirels LF, Lutz JE, Holloway M, Galgiani JN. Safety, tolerance, and efficacy of posaconazole therapy in patients with nonmeningeal disseminated or chronic pulmonary coccidioidomycosis. Clin Infect Dis. 2007; 45:562–568.
Article18. Pham AN, Bubalo JS, Lewis JS 2nd. Comparison of posaconazole serum concentrations from haematological cancer patients on posaconazole tablet and oral suspension for treatment and prevention of invasive fungal infections. Mycoses. 2016; [Epub ahead of print].
Article19. Cornely OA, Duarte RF, Haider S, Chandrasekar P, Helfgott D, Jiménez JL, Candoni A, Raad I, Laverdiere M, Langston A, Kartsonis N, Van Iersel M, Connelly N, Waskin H. Phase 3 pharmacokinetics and safety study of a posaconazole tablet formulation in patients at risk for invasive fungal disease. J Antimicrob Chemother. 2016; 71:718–726.
Article20. Jung DS, Tverdek FP, Kontoyiannis DP. Switching from posaconazole suspension to tablets increases serum drug levels in leukemia patients without clinically relevant hepatotoxicity. Antimicrob Agents Chemother. 2014; 58:6993–6995.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Fatal Breakthrough Mucormycosis in an Acute Myelogenous Leukemia Patient while on Posaconazole Prophylaxis
- Incidence and Types of Constitutional Chromosomal Abnormalities in Patients with Hematologic Malignancies
- Comparison of the Concentrations of 8-MOP in both Plasma and Suction Blister Fluid after Oral Ingestion
- Extracorporeal Life Support in Patients with Hematologic Malignancies: A Single Center Experience
- Posaconazole versus Itraconazole as Prophylactic Antifungal Agents during Induction Chemotherapy for Acute Myeloid Leukemia: A Real-World Single Center Comparison