Yonsei Med J.
2016 Mar;57(2):350-357. 10.3349/ymj.2016.57.2.350.
Increasing Incidence of High-Level Tetracycline-Resistant Neisseria gonorrhoeae due to Clonal Spread and Foreign Import
- Affiliations
-
- 1Department of Laboratory Medicine, International St. Mary's Hospital, Catholic Kwandong University College of Medicine, Incheon, Korea.
- 2Department of Laboratory Medicine and Research Institute of Bacterial Resistance, Yonsei University College of Medicine, Seoul, Korea. leekcp@yuhs.ac
- KMID: 2374039
- DOI: http://doi.org/10.3349/ymj.2016.57.2.350
Abstract
- PURPOSE
The detection of high-level tetracycline-resistant strains of Neisseria gonorrhoeae (TRNG) can make important epidemiological contributions that are relevant to controlling infections from this pathogen. In this study, we aimed to determine the incidence of TRNG isolates over time and also to investigate the characteristics and genetic epidemiology of these TRNG isolates in Korea.
MATERIALS AND METHODS
The antimicrobial susceptibilities of 601 isolates of N. gonorrhoeae from 2004 to 2011 were tested by standard Clinical and Laboratory Standards Institute methods. To determine the molecular epidemiological relatedness, N. gonorrhoeae multi-antigen sequence typing was performed.
RESULTS
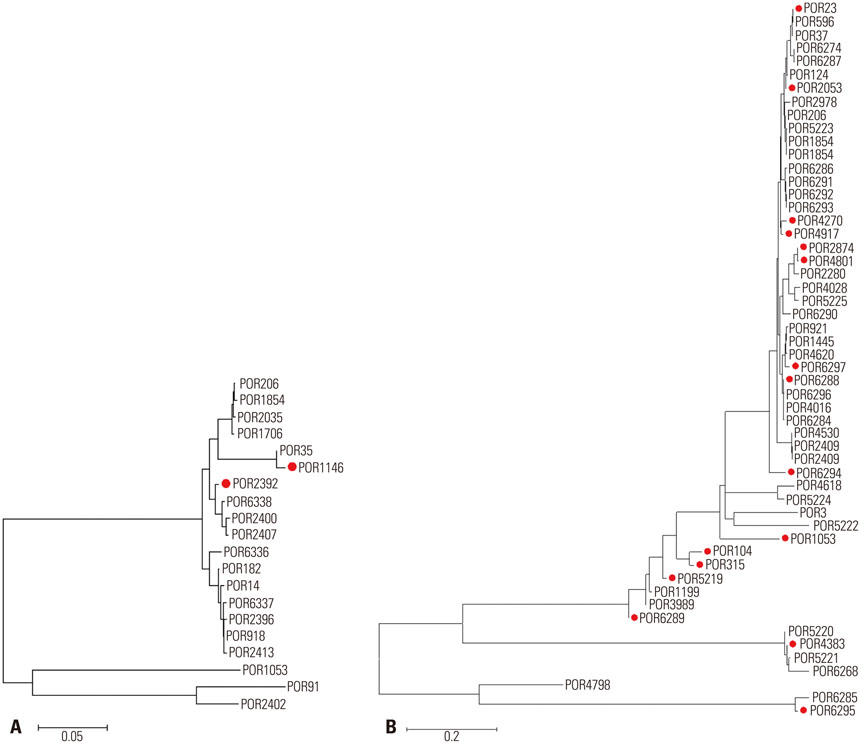
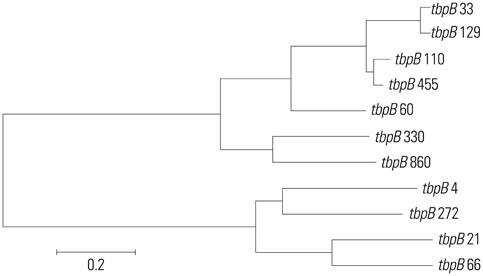
The incidence of TRNG increased from 2% in 2004 to 21% in 2011. The minimum inhibitory concentration distributions of ceftriaxone and susceptibility of ciprofloxacin in TRNG were different from non-TRNG and varied according to the year of isolation. Most of the TRNG isolates collected from 2004 to 2007 exhibited genetic relatedness, with sequence type (ST) 1798 being the most common. From 2008 to 2011, the STs of the isolates became more variable and introduction of genetically unrelated TRNG were noted.
CONCLUSION
The increased incidence of TRNG strains until 2007 appears to be due, at least in part, to clonal spread. However, we propose that the emergence of various STs since 2008 could be associated with foreign import.
MeSH Terms
-
Anti-Bacterial Agents/*pharmacology
Ceftriaxone/pharmacology
Ciprofloxacin/pharmacology
DNA, Bacterial/analysis
Drug Resistance, Multiple, Bacterial/*genetics
Gonorrhea/drug therapy/epidemiology/microbiology
Humans
Incidence
Microbial Sensitivity Tests
Molecular Epidemiology
Neisseria gonorrhoeae/*drug effects/*genetics/isolation & purification
Republic of Korea/epidemiology
Sequence Analysis, DNA
Tetracycline/pharmacology
Tetracyclines/*pharmacology
Anti-Bacterial Agents
Ciprofloxacin
Ceftriaxone
DNA, Bacterial
Tetracycline
Tetracyclines
Figure
Cited by 1 articles
-
History of Syphilis and Gonorrhea in Korea
Jae-Ki Choi, Seung-Ju Lee, Jin-Hong Yoo
Infect Chemother. 2019;51(2):210-216. doi: 10.3947/ic.2019.51.2.210.
Reference
-
1. WHO. Global incidence and prevalence of selected curable sexually transmitted infections - 2008. accessed on 2015 Jan 5. Available at: http://www.who.int/reproductivehealth/publications/rtis/2008_STI_estimates.pdf.2. Unemo M, Ison CA, Cole M, Spiteri G, van de Laar M, Khotenashvili L. Gonorrhoea and gonococcal antimicrobial resistance surveillance networks in the WHO European Region, including the independent countries of the former Soviet Union. Sex Transm Infect. 2013; 89:Suppl 4. iv42–iv46.
Article3. Centers for Disease Control and Prevention. Transmitted Disease Surveillance 2012. accessed on 2015 Jan 5. Available at: http://www.cdc.gov/sTD/stats12/Surv2012.pdf.4. Centers for Disease Control and Prevention. Antibiotic Resistance Threats in the United States, 2013. accessed on 2015 Jan 5. Available at: http://www.cdc.gov/drugresistance/threat-report-2013/pdf/ar-threats-2013-508.pdf.5. Health Insurance Review & Assessment Service. Statistics for Diseases and Treatment in Korea. accessed on 2015 Jan 5. Available at: http://www.hira.or.kr/rd/dissdic/infoSickList.do?pgmid=HIRAA020044020100.6. Elias J, Frosch M, Vogel U. Neisseria. In : Versalovic J, Carroll KC, Funke G, Jorgensen JH, Landry ML, Warnock DW, editors. Manual of Clinical Microbiology. 10th ed. Washington, DC: ASM Press;2011. p. 559–573.7. Chacko MR, Wiemann CM, Smith PB. Chlamydia and gonorrhea screening in asymptomatic young women. J Pediatr Adolesc Gynecol. 2004; 17:169–178.
Article8. Barbee LA. Preparing for an era of untreatable gonorrhea. Curr Opin Infect Dis. 2014; 27:282–287.
Article9. Unemo M, Golparian D, Shafer WM. Challenges with gonorrhea in the era of multi-drug and extensively drug resistance - are we on the right track? Expert Rev Anti Infect Ther. 2014; 12:653–656.
Article10. WHO. Global action plan to control the spread and impact of antimicrobial resistance in Neisseria gonorrhoeae. accessed on 2015 Jan 5. Available at: http://apps.who.int/iris/bitstream/10665/44863/1/9789241503501_eng.pdf.11. Chong Y, Kim SO, Yi KN, Lee SY. Penicillin and tetracycline susceptibility of Neisseria gonorrhoeae strains isolated during 1966 to 1975. Yonsei Med J. 1976; 17:46–51.
Article12. Lee K, Shin JW, Lim JB, Kim YA, Yong D, Oh HB, et al. Emerging antimicrobial resistance, plasmid profile and pulsed-field gel electrophoresis pattern of the endonuclease-digested genomic DNA of Neisseria gonorrhoeae. Yonsei Med J. 2000; 41:381–386.
Article13. Clinical and Laboratory Standards Institute. Performance Standards for Antimicrobial Susceptibility Testing; Twenty-Third Informational Supplement. Wayne, PA: Clinical and Laboratory Standards Institute;2013.14. Clinical and Laboratory Standards Institute. Performance Standards for Antimicrobial Disk Susceptibility Tests; Approved Standard. 11th ed. Wayne, PA: Clinical and Laboratory Standards Institute;2012.15. Clinical and Laboratory Standards Institute. Methods for dilution antimicrobial susceptibility tests for bacteria that grow aerobically; approved standard. 9th ed. Wayne, PA: Clinical and Laboratory Standards Institute;2012.16. Martin IM, Ison CA, Aanensen DM, Fenton KA, Spratt BG. Rapid sequence-based identification of gonococcal transmission clusters in a large metropolitan area. J Infect Dis. 2004; 189:1497–1505.
Article17. Ameyama S, Onodera S, Takahata M, Minami S, Maki N, Endo K, et al. Mosaic-like structure of penicillin-binding protein 2 Gene (penA) in clinical isolates of Neisseria gonorrhoeae with reduced susceptibility to cefixime. Antimicrob Agents Chemother. 2002; 46:3744–3749.
Article18. Ohnishi M, Saika T, Hoshina S, Iwasaku K, Nakayama S, Watanabe H, et al. Ceftriaxone-resistant Neisseria gonorrhoeae, Japan. Emerg Infect Dis. 2011; 17:148–149.19. Wright DJ, Azadian B. Cephalosporin resistance in gonorrhoea. Lancet Infect Dis. 2013; 13:728–730.
Article20. Sparling PF, Sarubbi FA Jr, Blackman E. Inheritance of low-level resistance to penicillin, tetracycline, and chloramphenicol in Neisseria gonorrhoeae. J Bacteriol. 1975; 124:740–749.
Article21. Morse SA, Johnson SR, Biddle JW, Roberts MC. High-level tetracycline resistance in Neisseria gonorrhoeae is result of acquisition of streptococcal tetM determinant. Antimicrob Agents Chemother. 1986; 30:664–670.
Article22. Lee H, Hong SG, Soe Y, Yong D, Jeong SH, Lee K, et al. Trends in antimicrobial resistance of Neisseria gonorrhoeae isolated from Korean patients from 2000 to 2006. Sex Transm Dis. 2011; 38:1082–1086.
Article23. Kim HJ, Seo Y, Kim WH, Lee Y, Lee H, Lee K, et al. Antimicrobial resistance and molecular epidemiologic characteristics of Neisseria gonorrhoeae isolated from Korea in 2013. Ann Clin Microbiol. 2013; 16:182–187.
Article24. Palmer HM, Young H. Dramatic increase in a single genotype of TRNG ciprofloxacin-resistant Neisseria gonorrhoeae isolates in men who have sex with men. Int J STD AIDS. 2006; 17:254–256.
Article25. Fernandez Cobo M, Galarza P, Sparo M, Buscemi L, Pizarro MR, Fiorito S. Characterization of an outbreak of tetM-containing Neisseria gonorrhoeae in Argentina. Int J STD AIDS. 1999; 10:169–173.
Article26. Bala M, Jain RK, Ray K. Antimicrobial susceptibility profile of resistance phenotypes of Neisseria gonorrheae in India. Sex Transm Dis. 2008; 35:588–591.
Article27. Roberts MC, Knapp JS. Host range of the conjugative 25.2-megadalton tetracycline resistance plasmid from Neisseria gonorrhoeae and related species. Antimicrob Agents Chemother. 1988; 32:488–491.
Article28. Greco V, Ng LK, Catana R, Li H, Dillon JA. Molecular epidemiology of Neisseria gonorrheae isolates with plasmid-mediated tetracycline resistance in Canada: temporal and geographical trends (1986-1997). Microb Drug Resist. 2003; 9:353–360.
Article29. Moodley P, Hoppenbrouwers J, Bohlken L, Sturm AW. Emergence of TetM-mediated tetracycline resistance in rural South Africa. J Antimicrob Chemother. 2001; 48:142–143.
Article30. Olsen B, Hadad R, Fredlund H, Unemo M. The Neisseria gonorrhoeae population in Sweden during 2005-phenotypes, genotypes and antibiotic resistance. APMIS. 2008; 116:181–189.
Article31. WHO Western Pacific Gonococcal Antimicrobial Surveillance Programme. Surveillance of antibiotic resistance in Neisseria gonorrhoeae in the WHO Western Pacific Region, 2004. Commun Dis Intell Q Rep. 2006; 30:129–132.32. Lahra MM. WHO Western Pacific and South East Asian Gonococcal Antimicrobial Surveillance Programme. Surveillance of antibiotic resistance in Neisseria gonorrhoeae in the WHO Western Pacific and South East Asian Regions, 2010. Commun Dis Intell Q Rep. 2012; 36:95–100.33. Chisholm SA, Unemo M, Quaye N, Johansson E, Cole MJ, Ison CA, et al. Molecular epidemiological typing within the European Gonococcal Antimicrobial Resistance Surveillance Programme reveals predominance of a multidrug-resistant clone. Euro Surveill. 2013; (18):pii: 20358.
Article34. Cámara J, Serra J, Ayats J, Bastida T, Carnicer-Pont D, Andreu A, et al. Molecular characterization of two high-level ceftriaxone-resistant Neisseria gonorrhoeae isolates detected in Catalonia, Spain. J Antimicrob Chemother. 2012; 67:1858–1860.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Antimicrobial Resistance of Neisseria gonorrhoeae Isolated in Korea
- Surveillance for Spectinomycin Resistant Neisseria Gonorrhoeae
- Antimicrobial Resistance of Neisseria gonorrhoeae Isolated in Korea
- Treatment of Neisseria gonorrhoeae in the Era of Multidrug Resistance
- Prevalence of PPNG in Seoul, Korea ( 1983 ~ 1984 )