J Korean Med Sci.
2016 Aug;31(8):1215-1223. 10.3346/jkms.2016.31.8.1215.
OTX1 Contributes to Hepatocellular Carcinoma Progression by Regulation of ERK/MAPK Pathway
- Affiliations
-
- 1Department of Anesthesiology, Shanghai Pulmonary Hospital, Tongji University, School of Medicine, Shanghai, China.
- 2Department of Oncology, Quzhou People's Hospital in Zhejiang Province, Quzhou Zhejiang, China.
- 3Department of Pathology, Affiliated Hospital Cancer Center, Academy of Military Medical Sciences, Beijing, China.
- 4Department of Oncology, Lishui Central Hospital, Lishui Hospital of Zhejiang University, Lishui, Zhejiang, China.
- 5Department of Pathology, Zhejiang Cancer Hospital, Hangzhou Zhejiang, China. wmj1999@126.com
- KMID: 2373748
- DOI: http://doi.org/10.3346/jkms.2016.31.8.1215
Abstract
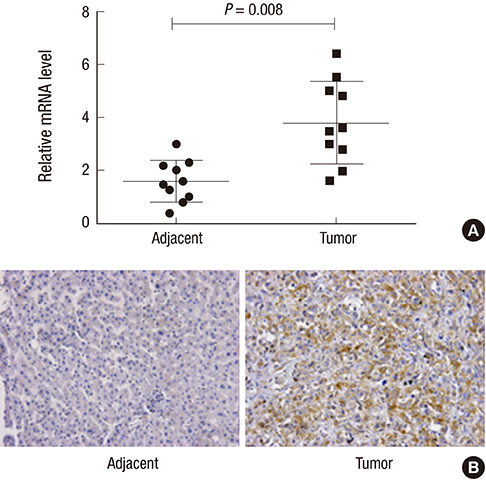
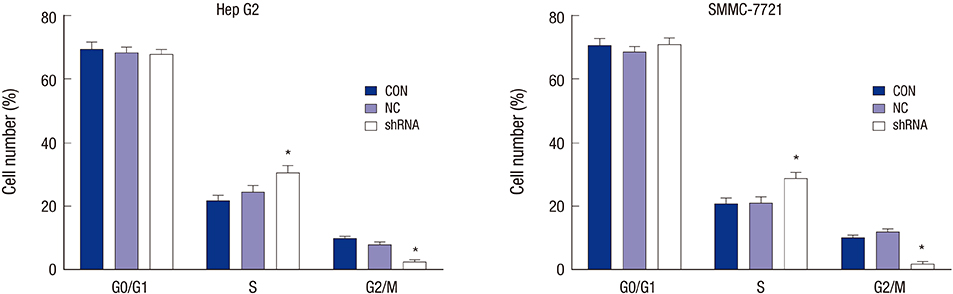
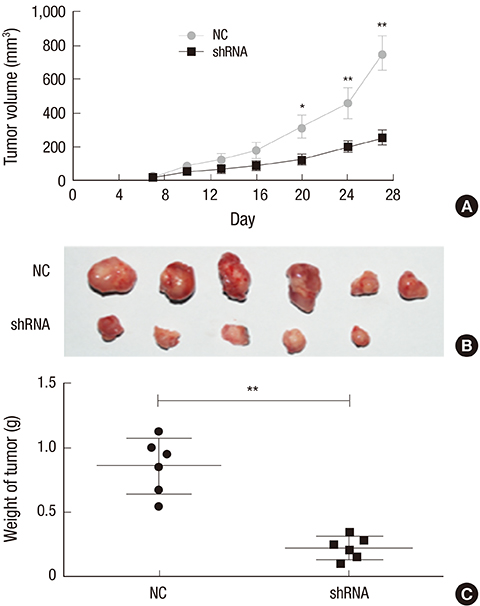
- Orthodenticlehomeobox 1 (OTX1) overexpression had previously been associated with the progression of several tumors. The present study aimed to determine the expression and role of OTX1 in human hepatocellular carcinoma (HCC). The expression level of OTX1 was examined by quantitative real-time PCR (qRT-PCR) in 10 samples of HCC and paired adjacent non-cancerous tissues, and by immunohistochemistry (IHC) analysis in 128 HCC samples and matched controls. The relationship between OTX1 expression and the clinicopathological features werealso analyzed. Furthermore, the effects of OTX1 knockdown on cell proliferation and migration were determined in HCC cell lines. Axenograft mouse model was also established to investigate the role of OTX1 in HCC tumor growth. TheqRT-PCR and IHC analyses revealed that OTX1 was significantly elevated in HCC tissues compared with the paired non-cancerous controls. Expression of OTX1 was positively correlated with nodal metastasis status (P = 0.009) and TNM staging (P = 0.001) in HCC tissues. In addition, knockdown of OTX1 by shRNA significantly inhibited the proliferation and migration, and induced cell cycle arrest in S phase in vitro. Tumor growth was markedly inhibited by OTX1 silencing in the xenograft. Moreover, OTX1 silencing was causable for the decreased phosphorylation level of ERK/MAPK signaling. In conclusion, OTX1 contributes to HCC progression possibly by regulation of ERK/MAPK pathway. OTX1 may be a novel target for molecular therapy towards HCC.
Keyword
MeSH Terms
-
Aged
Animals
Blotting, Western
Carcinoma, Hepatocellular/metabolism/*pathology
Cell Line, Tumor
Cell Movement
Cell Proliferation
Disease Progression
Female
Gene Expression Regulation, Neoplastic
Humans
Immunohistochemistry
Liver/metabolism/pathology
Liver Neoplasms/metabolism/*pathology
Lymphatic Metastasis
MAP Kinase Signaling System
Male
Mice
Mice, Inbred BALB C
Mice, Nude
Middle Aged
Neoplasm Staging
Otx Transcription Factors/antagonists & inhibitors/genetics/*metabolism
Phosphorylation
RNA Interference
Real-Time Polymerase Chain Reaction
S Phase Cell Cycle Checkpoints
Transplantation, Heterologous
Otx Transcription Factors
Figure
Reference
-
1. El-Serag HB, Rudolph KL. Hepatocellular carcinoma: epidemiology and molecular carcinogenesis. Gastroenterology. 2007; 132:2557–2576.2. El-Serag HB, Kanwal F. Epidemiology of hepatocellular carcinoma in the United States: where are we? Where do we go? Hepatology. 2014; 60:1767–1775.3. Bosch FX, Ribes J, Díaz M, Cléries R. Primary liver cancer: worldwide incidence and trends. Gastroenterology. 2004; 127:S5–16.4. Llovet JM, Burroughs A, Bruix J. Hepatocellular carcinoma. Lancet. 2003; 362:1907–1917.5. Shin JW, Chung YH. Molecular targeted therapy for hepatocellular carcinoma: current and future. World J Gastroenterol. 2013; 19:6144–6155.6. Chan SL, Mok T, Ma BB. Management of hepatocellular carcinoma: beyond sorafenib. Curr Oncol Rep. 2012; 14:257–266.7. Wilhelm SM, Carter C, Tang L, Wilkie D, McNabola A, Rong H, Chen C, Zhang X, Vincent P, McHugh M, et al. BAY 43-9006 exhibits broad spectrum oral antitumor activity and targets the RAF/MEK/ERK pathway and receptor tyrosine kinases involved in tumor progression and angiogenesis. Cancer Res. 2004; 64:7099–7109.8. Chang YS, Adnane J, Trail PA, Levy J, Henderson A, Xue D, Bortolon E, Ichetovkin M, Chen C, McNabola A, et al. Sorafenib (BAY 43-9006) inhibits tumor growth and vascularization and induces tumor apoptosis and hypoxia in RCC xenograft models. Cancer Chemother Pharmacol. 2007; 59:561–574.9. Klein WH, Li X. Function and evolution of Otx proteins. Biochem Biophys Res Commun. 1999; 258:229–233.10. Acampora D, Postiglione MP, Avantaggiato V, Di Bonito M, Simeone A. The role of Otx and Otp genes in brain development. Int J Dev Biol. 2000; 44:669–677.11. Larsen KB, Lutterodt M, Rath MF, Møller M. Expression of the homeobox genes PAX6, OTX2, and OTX1 in the early human fetal retina. Int J Dev Neurosci. 2009; 27:485–492.12. Pagani IS, Terrinoni A, Marenghi L, Zucchi I, Chiaravalli AM, Serra V, Rovera F, Sirchia S, Dionigi G, Miozzo M, et al. The mammary gland and the homeobox gene Otx1. Breast J. 2010; 16:Suppl 1. S53–6.13. Omodei D, Acampora D, Russo F, De Filippi R, Severino V, Di Francia R, Frigeri F, Mancuso P, De Chiara A, Pinto A, et al. Expression of the brain transcription factor OTX1 occurs in a subset of normal germinal-center B cells and in aggressive Non-Hodgkin Lymphoma. Am J Pathol. 2009; 175:2609–2617.14. Zakrzewska M, Grešner SM, Zakrzewski K, Zalewska-Szewczyk B, Liberski PP. Novel gene expression model for outcome prediction in paediatric medulloblastoma. J Mol Neurosci. 2013; 51:371–379.15. de Haas T, Oussoren E, Grajkowska W, Perek-Polnik M, Popovic M, Zadravec-Zaletel L, Perera M, Corte G, Wirths O, van Sluis P, et al. OTX1 and OTX2 expression correlates with the clinicopathologic classification of medulloblastomas. J Neuropathol Exp Neurol. 2006; 65:176–186.16. Terrinoni A, Pagani IS, Zucchi I, Chiaravalli AM, Serra V, Rovera F, Sirchia S, Dionigi G, Miozzo M, Frattini A, et al. OTX1 expression in breast cancer is regulated by p53. Oncogene. 2011; 30:3096–3103.17. Yu K, Cai XY, Li Q, Yang ZB, Xiong W, Shen T, Wang WY, Li YF. OTX1 promotes colorectal cancer progression through epithelial-mesenchymal transition. Biochem Biophys Res Commun. 2014; 444:1–5.18. Li LL, Xue AM, Li BX, Shen YW, Li YH, Luo CL, Zhang MC, Jiang JQ, Xu ZD, Xie JH, et al. JMJD2A contributes to breast cancer progression through transcriptional repression of the tumor suppressor ARHI. Breast Cancer Res. 2014; 16:R56.19. Whittaker S, Marais R, Zhu AX. The role of signaling pathways in the development and treatment of hepatocellular carcinoma. Oncogene. 2010; 29:4989–5005.20. Wang S, Huang X, Li Y, Lao H, Zhang Y, Dong H, Xu W, Li JL, Li M. RN181 suppresses hepatocellular carcinoma growth by inhibition of the ERK/MAPK pathway. Hepatology. 2011; 53:1932–1942.21. Pratilas CA, Solit DB. Targeting the mitogen-activated protein kinase pathway: physiological feedback and drug response. Clin Cancer Res. 2010; 16:3329–3334.22. Villanueva A, Minguez B, Forner A, Reig M, Llovet JM. Hepatocellular carcinoma: novel molecular approaches for diagnosis, prognosis, and therapy. Annu Rev Med. 2010; 61:317–328.23. Thomas MB, Abbruzzese JL. Opportunities for targeted therapies in hepatocellular carcinoma. J Clin Oncol. 2005; 23:8093–8108.24. Llovet JM, Ricci S, Mazzaferro V, Hilgard P, Gane E, Blanc JF, de Oliveira AC, Santoro A, Raoul JL, Forner A, et al. Sorafenib in advanced hepatocellular carcinoma. N Engl J Med. 2008; 359:378–390.25. Chen H, Sukumar S. Role of homeobox genes in normal mammary gland development and breast tumorigenesis. J Mammary Gland Biol Neoplasia. 2003; 8:159–175.26. Jeon K, Lim H, Kim JH, Han D, Lee ER, Yang GM, Song MK, Kim JH, Cho SG. Bax inhibitor-1 enhances survival and neuronal differentiation of embryonic stem cells via differential regulation of mitogen-activated protein kinases activities. Biochim Biophys Acta. 2012; 1823:2190–2200.27. Burotto M, Chiou VL, Lee JM, Kohn EC. The MAPK pathway across different malignancies: a new perspective. Cancer. 2014; 120:3446–3456.28. Huynh H, Nguyen TT, Chow KH, Tan PH, Soo KC, Tran E. Over-expression of the mitogen-activated protein kinase (MAPK) kinase (MEK)-MAPK in hepatocellular carcinoma: its role in tumor progression and apoptosis. BMC Gastroenterol. 2003; 3:19.29. Ito Y, Sasaki Y, Horimoto M, Wada S, Tanaka Y, Kasahara A, Ueki T, Hirano T, Yamamoto H, Fujimoto J, et al. Activation of mitogen-activated protein kinases/extracellular signal-regulated kinases in human hepatocellular carcinoma. Hepatology. 1998; 27:951–958.30. Sun Y, Tang S, Jin X, Zhang C, Zhao W, Xiao X. Opposite effects of JNK and p38 MAPK signaling pathways on furazolidone-stimulated S phase cell cycle arrest of human hepatoblastoma cell line. Mutat Res. 2013; 755:24–29.31. Sun Y, Tang S, Jin X, Zhang C, Zhao W, Xiao X. Involvement of the p38 MAPK signaling pathway in S-phase cell-cycle arrest induced by Furazolidone in human hepatoma G2 cells. J Appl Toxicol. 2013; 33:1500–1505.32. Iguchi T, Miyakawa Y, Yamamoto K, Kizaki M, Ikeda Y. Nitrogen-containing bisphosphonates induce S-phase cell cycle arrest and apoptosis of myeloma cells by activating MAPK pathway and inhibiting mevalonate pathway. Cell Signal. 2003; 15:719–727.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Ras Mitogen-activated Protein Kinase Signaling and Kinase Suppressor of Ras as Therapeutic Targets for Hepatocellular Carcinoma
- The Extracellular Signal-Regulated Kinase Mitogen-Activated Protein Kinase Pathway in Osteoblasts
- Expression of Down Stream Molecules of RET (p-ERK, p-p38 MAPK, p-JNK and p-AKT) in Papillary Thyroid Carcinomas
- Anti-proliferative Effect of 15,16-Dihydrotanshinone I Through Cell Cycle Arrest and the Regulation of AMP-activated Protein Kinase/Akt/mTOR and Mitogen-activated Protein Kinase Signaling Pathway in Human Hepatocellular Carcinoma Cells
- Induction of Integrin Signaling by Steroid Sulfatase in Human Cervical Cancer Cells