J Vet Sci.
2016 Mar;17(1):119-122. 10.4142/jvs.2016.17.1.119.
Molecular cloning, purification and immunogenicity of recombinant Brucella abortus 544 malate dehydrogenase protein
- Affiliations
-
- 1Institute of Animal Medicine, College of Veterinary Medicine, Gyeongsang National University, Jinju 52828, Korea. kimsuk@gnu.ac.kr
- 2Department of Veterinary Paraclinical Sciences, College of Veterinary Medicine, University of the Philippines Los Baños, College, Laguna 4031, Philippines.
- 3Institute of Agriculture and Life Science, College of Veterinary Medicine, Gyeongsang National University, Jinju 52828, Korea.
- KMID: 2363344
- DOI: http://doi.org/10.4142/jvs.2016.17.1.119
Abstract
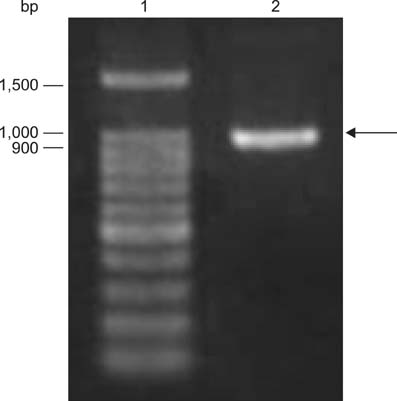
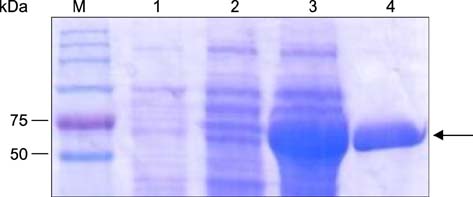
- The Brucella mdh gene was successfully cloned and expressed in E. coli. The purified recombinant malate dehydrogenase protein (rMDH) was reactive to Brucella-positive bovine serum in the early stage, but not reactive in the middle or late stage, and was reactive to Brucella-positive mouse serum in the late stage, but not in the early or middle stage of infection. In addition, rMDH did not react with Brucella-negative bovine or mouse sera. These results suggest that rMDH has the potential for use as a specific antigen in serological diagnosis for early detection of bovine brucellosis.
MeSH Terms
-
Animals
Antigens, Bacterial/*immunology
Brucella abortus/*enzymology/immunology
Brucellosis/diagnosis/*veterinary
Cattle
Cattle Diseases/*diagnosis
Cloning, Molecular
Enzyme-Linked Immunosorbent Assay
Escherichia coli/genetics
Malate Dehydrogenase/*genetics/*immunology/isolation & purification
Mice
Recombinant Proteins/genetics/*immunology
Antigens, Bacterial
Malate Dehydrogenase
Recombinant Proteins
Figure
Cited by 1 articles
-
Tannic acid-mediated immune activation attenuates Brucella abortus infection in mice
Alisha W. B. Reyes, Huynh T. Hop, Lauren T. Arayan, Tran X. N. Huy, Wongi Min, Hu Jang Lee, Hong Hee Chang, Suk Kim
J Vet Sci. 2018;19(1):51-57. doi: 10.4142/jvs.2018.19.1.51.
Reference
-
1. Al Dahouk S, Tomaso H, Nöckler K, Neubauer H, Frangoulidis D. Laboratory-based diagnosis of brucellosis– a review of the literature. Part II: serological tests for brucellosis. Clin Lab. 2003; 49:577–589.2. Carvalho Neta AV, Mol JPS, Xavier MN, Paixão TA, Lage AP, Santos RL. Pathogenesis of bovine brucellosis. Vet J. 2010; 184:146–155.
Article3. Cassataro J, Pasquevich K, Bruno L, Wallach JC, Fossati CA, Baldi PC. Antibody reactivity to Omp31 from Brucella melitensis in human and animal infections by smooth and rough brucellae. Clin Diagn Lab Immunol. 2004; 11:111–114.
Article4. Clavijo E, Díaz R, Anguita Á, García A, Pinedo A, Smits HL. Comparison of a dipstick assay for detection of Brucella-specific immunoglobulin M antibodies with other tests for serodiagnosis of human brucellosis. Clin Diagn Lab Immunol. 2003; 10:612–615.
Article5. Han X, Tong Y, Tian M, Zhang Y, Sun X, Wang S, Qiu X, Ding C, Yu S. Enzymatic activity analysis and catalytic essential residues identification of Brucella abortus malate dehydrogenase. ScientificWorldJournal. 2014; 2014:973751.6. Ko J, Splitter GA. Molecular host-pathogen interaction in brucellosis: current understanding and future approaches to vaccine development for mice and humans. Clin Microbiol Rev. 2003; 16:65–78.
Article7. Lee JJ, Simborio HL, Reyes AWB, Kim DG, Hop HT, Min W, Her M, Jung SC, Yoo HS, Kim S. Proteomic analyses of the time course responses of mice infected with Brucella abortus 544 reveal immunogenic antigens. FEMS Microbiol Lett. 2014; 357:164–174.8. Lee JJ, Simborio HL, Reyes AWB, Kim DG, Hop HT, Min W, Her M, Jung SC, Yoo HS, Kim S. Immunoproteomic identification of immunodominant antigens independent of the time of infection in Brucella abortus 2308-challenged cattle. Vet Res. 2015; 46:17.9. Lowry JE, Goodridge L, Vernati G, Fluegel AM, Edwards WH, Andrews GP. Identification of Brucella abortus genes in elk (Cervus elaphus) using in vivo-induced antigen technology (IVIAT) reveals novel markers of infection. Vet Microbiol. 2010; 142:367–372.
Article10. Lowry JE, Isaak DD, Leonhardt JA, Vernati G, Pate JC, Andrews GP. Vaccination with Brucella abortus recombinant in vivo-induced antigens reduces bacterial load and promotes clearance in a mouse model for infection. PLoS One. 2011; 6:e17425.11. McGiven JA, Stack JA, Perrett LL, Tucker JD, Brew SD, Stubberfield E, MacMillan AP. Harmonisation of European tests for serological diagnosis of Brucella infection in bovines. Rev Sci Tech. 2006; 25:1039–1053.12. Olsen SC. Recent developments in livestock and wildlife brucellosis vaccination. Rev Sci Tech. 2013; 32:207–217.
Article13. Pajuaba AC, Silva DAO, Almeida KC, Cunha-Junior JP, Pirovani CP, Camillo LR, Mineo JR. Immunoproteomics of Brucella abortus reveals differential antibody profiles between S19-vaccinated and naturally infected cattle. Proteomics. 2012; 12:820–831.
Article14. Park SJ, Cotter PA, Gunsalus RP. Regulation of malate dehydrogenase (mdh) gene expression in Escherichia coli in response to oxygen, carbon, and heme availability. J Bacteriol. 1995; 177:6652–6656.
Article15. Seleem MN, Boyle SM, Sriranganathan N. Brucellosis: a re-emerging zoonosis. Vet Microbiol. 2010; 140:392–398.
Article16. Trant CG, Lacerda TLS, Carvalho NB, Azevedo V, Rosinha GMS, Salcedo SP, Gorvel JP, Oliveira SC. The Brucella abortus phosphoglycerate kinase mutant is highly attenuated and induces protection superior to that of vaccine strain 19 in immunocompromised and immunocompetent mice. Infect Immun. 2010; 78:2283–2291.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Protective effects of recombinant Brucella abortus Omp28 against infection with a virulent strain of Brucella abortus 544 in mice
- Diversity of Humoral Immune Responses to Recombinant Proteins of Brucella abortus Among Residents in Cheju Province
- In silico analysis of Brucella abortus Omp2b and in vitro expression of SOmp2b
- The virulence of Brucella abortus isolated from cattle in Korea
- Different invasion efficiencies of Brucella abortus wild-type and mutantsin RAW 264.7 and THP-1 phagocytic cells and HeLa non-phagocytic cells