Immune Netw.
2016 Dec;16(6):337-343. 10.4110/in.2016.16.6.337.
Folate Receptor-Targeted Diagnostics and Therapeutics for Inflammatory Diseases
- Affiliations
-
- 1Department of Pharmaceutical Engineering, Cheongju University, Cheongju 28503, Korea. ysyi@cju.ac.kr
- KMID: 2362798
- DOI: http://doi.org/10.4110/in.2016.16.6.337
Abstract
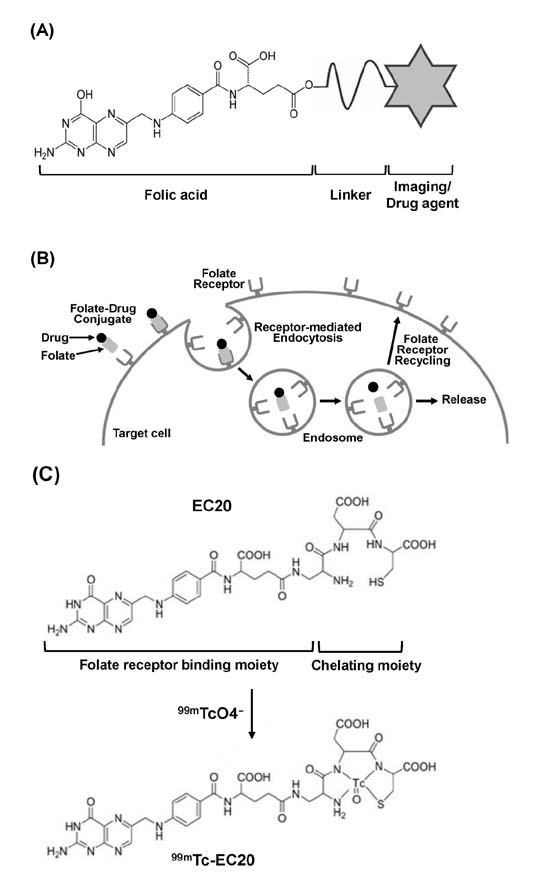
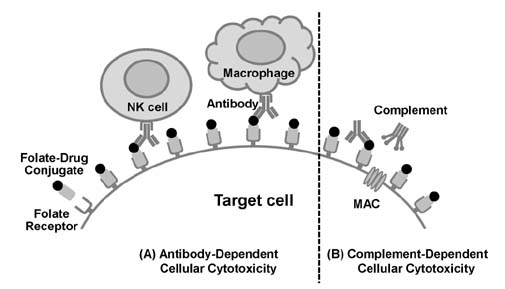
- Inflammation, an innate immune response mediated by macrophages, forms the first line of defence to protect our body from the invasion of various pathogens. Although inflammation is a defensive response, chronic inflammation has been regarded as the major cause of many types of human diseases such as inflammatory/autoimmune diseases, cancers, neurological diseases, and cardiovascular diseases. Folate receptor (FR) is a cell surface glycosylphosphatidylinositol (GPI)-anchored glycoprotein, and its three isoforms, FR-α, FR-β, and FR-γ, are found in humans. Interestingly, FRs are highly expressed on a variety of cells, including cancer cells and activated macrophages, whereas their expression on normal cells is undetectable, indicating that FR-targeting could be a good selective strategy for the diagnosis and therapeutic treatment of cancers and activated macrophage-mediated inflammatory diseases. Previous studies successfully showed FR-targeted imaging of many types of cancers in animal models as well as human patients. Recently, a number of emerging studies have found that activated macrophages, which are critical players for a variety of inflammatory diseases, highly express FRs, and selective targeting of these FR-positive activated macrophages is a good approach to diagnose and treat inflammatory diseases. In this review, we describe the characteristics and structure of FRs, and further discuss FR-targeted diagnostics and therapeutics of human diseases, in particular, activated macrophage-mediated inflammatory diseases.
MeSH Terms
Figure
Cited by 2 articles
-
Regulatory Roles of the Caspase-11 Non-Canonical Inflammasome in Inflammatory Diseases
Young-Su Yi
Immune Netw. 2018;18(6):. doi: 10.4110/in.2018.18.e41.Role of inflammasomes in inflammatory autoimmune rheumatic diseases
Young-Su Yi
Korean J Physiol Pharmacol. 2018;22(1):1-15. doi: 10.4196/kjpp.2018.22.1.1.
Reference
-
1. Low PS, Henne WA, Doorneweerd DD. Discovery and development of folic-acid-based receptor targeting for imaging and therapy of cancer and inflammatory diseases. Acc Chem Res. 2008; 41:120–129.
Article2. Lu JY, Lowe DA, Kennedy MD, Low PS. Folate-targeted enzyme prodrug cancer therapy utilizing penicillin-V amidase and a doxorubicin prodrug. J Drug Target. 1999; 7:43–53.
Article3. Antony AC. The biological chemistry of folate receptors. Blood. 1992; 79:2807–2820.
Article4. Salazar MD, Ratnam M. The folate receptor: what does it promise in tissue-targeted therapeutics? Cancer Metastasis Rev. 2007; 26:141–152.
Article5. Elnakat H, Ratnam M. Distribution, functionality and gene regulation of folate receptor isoforms: implications in targeted therapy. Adv Drug Deliv Rev. 2004; 56:1067–1084.
Article6. Low PS, Antony AC. Folate receptor-targeted drugs for cancer and inflammatory diseases. Adv Drug Deliv Rev. 2004; 56:1055–1058.
Article7. Kamen B. Folate and antifolate pharmacology. Semin Oncol. 1997; 24:S18.8. Goh YI, Koren G. Folic acid in pregnancy and fetal outcomes. J Obstet Gynaecol. 2008; 28:3–13.
Article9. Reddy JA, Low PS. Folate-mediated targeting of therapeutic and imaging agents to cancers. Crit Rev Ther Drug Carrier Syst. 1998; 15:587–627.
Article10. Spiegelstein O, Eudy JD, Finnell RH. Identification of two putative novel folate receptor genes in humans and mouse. Gene. 2000; 258:117–125.
Article11. Shen F, Wu M, Ross JF, Miller D, Ratnam M. Folate receptor type gamma is primarily a secretory protein due to lack of an efficient signal for glycosylphosphatidylinositol modification: protein characterization and cell type specificity. Biochemistry. 1995; 34:5660–5665.
Article12. Lacey SW, Sanders JM, Rothberg KG, Anderson RG, Kamen BA. Complementary DNA for the folate binding protein correctly predicts anchoring to the membrane by glycosyl-phosphatidylinositol. J Clin Invest. 1989; 84:715–720.
Article13. Shen F, Ross JF, Wang X, Ratnam M. Identification of a novel folate receptor, a truncated receptor, and receptor type beta in hematopoietic cells: cDNA cloning, expression, immunoreactivity, and tissue specificity. Biochemistry. 1994; 33:1209–1215.
Article14. Wang X, Shen F, Freisheim JH, Gentry LE, Ratnam M. Differential stereospecificities and affinities of folate receptor isoforms for folate compounds and antifolates. Biochem Pharmacol. 1992; 44:1898–1901.
Article15. Maziarz KM, Monaco HL, Shen F, Ratnam M. Complete mapping of divergent amino acids responsible for differential ligand binding of folate receptors alpha and beta. J Biol Chem. 1999; 274:11086–11091.
Article16. Shen F, Zheng X, Wang J, Ratnam M. Identification of amino acid residues that determine the differential ligand specificities of folate receptors alpha and beta. Biochemistry. 1997; 36:6157–6163.
Article17. Weitman SD, Lark RH, Coney LR, Fort DW, Frasca V, Zurawski VR Jr, Kamen BA. Distribution of the folate receptor GP38 in normal and malignant cell lines and tissues. Cancer Res. 1992; 52:3396–3401.18. Weitman SD, Weinberg AG, Coney LR, Zurawski VR, Jennings DS, Kamen BA. Cellular localization of the folate receptor: potential role in drug toxicity and folate homeostasis. Cancer Res. 1992; 52:6708–6711.19. Toffoli G, Cernigoi C, Russo A, Gallo A, Bagnoli M, Boiocchi M. Overexpression of folate binding protein in ovarian cancers. Int J Cancer. 1997; 74:193–198.
Article20. Wu M, Gunning W, Ratnam M. Expression of folate receptor type alpha in relation to cell type, malignancy, and differentiation in ovary, uterus, and cervix. Cancer Epidemiol Biomarkers Prev. 1999; 8:775–782.21. Ross JF, Wang H, Behm FG, Mathew P, Wu M, Booth R, Ratnam M. Folate receptor type beta is a neutrophilic lineage marker and is differentially expressed in myeloid leukemia. Cancer. 1999; 85:348–357.
Article22. Ratnam M, Marquardt H, Duhring JL, Freisheim JH. Homologous membrane folate binding proteins in human placenta: cloning and sequence of a cDNA. Biochemistry. 1989; 28:8249–8254.
Article23. Reddy JA, Haneline LS, Srour EF, Antony AC, Clapp DW, Low PS. Expression and functional characterization of the beta-isoform of the folate receptor on CD34(+) cells. Blood. 1999; 93:3940–3948.
Article24. Pan XQ, Zheng X, Shi G, Wang H, Ratnam M, Lee RJ. Strategy for the treatment of acute myelogenous leukemia based on folate receptor beta-targeted liposomal doxorubicin combined with receptor induction using all-trans retinoic acid. Blood. 2002; 100:594–602.
Article25. Nakashima-Matsushita N, Homma T, Yu S, Matsuda T, Sunahara N, Nakamura T, Tsukano M, Ratnam M, Matsuyama T. Selective expression of folate receptor beta and its possible role in methotrexate transport in synovial macrophages from patients with rheumatoid arthritis. Arthritis Rheum. 1999; 42:1609–1616.
Article26. Turk MJ, Breur GJ, Widmer WR, Paulos CM, Xu LC, Grote LA, Low PS. Folate-targeted imaging of activated macrophages in rats with adjuvant-induced arthritis. Arthritis Rheum. 2002; 46:1947–1955.
Article27. Mathias CJ, Lewis MR, Reichert DE, Laforest R, Sharp TL, Lewis JS, Yang ZF, Waters DJ, Snyder PW, Low PS, Welch MJ, Green MA. Preparation of 66Ga- and 68Ga-labeled Ga(III)-deferoxamine-folate as potential folate-receptor-targeted PET radiopharmaceuticals. Nucl Med Biol. 2003; 30:725–731.
Article28. Siegel BA, Dehdashti F, Mutch DG, Podoloff DA, Wendt R, Sutton GP, Burt RW, Ellis PR, Mathias CJ, Green MA, Gershenson DM. Evaluation of 111In-DTPA-folate as a receptor-targeted diagnostic agent for ovarian cancer: initial clinical results. J Nucl Med. 2003; 44:700–707.29. Reddy JA, Xu LC, Parker N, Vetzel M, Leamon CP. Preclinical evaluation of (99m)Tc-EC20 for imaging folate receptor-positive tumors. J Nucl Med. 2004; 45:857–866.30. Paulos CM, Turk MJ, Breur GJ, Low PS. Folate receptor-mediated targeting of therapeutic and imaging agents to activated macrophages in rheumatoid arthritis. Adv Drug Deliv Rev. 2004; 56:1205–1217.
Article31. Ayala-Lopez W, Xia W, Varghese B, Low PS. Imaging of atherosclerosis in apoliprotein e knockout mice: targeting of a folate-conjugated radiopharmaceutical to activated macrophages. J Nucl Med. 2010; 51:768–774.
Article32. Shen J, Chelvam V, Cresswell G, Low PS. Use of folate-conjugated imaging agents to target alternatively activated macrophages in a murine model of asthma. Mol Pharm. 2013; 10:1918–1927.
Article33. Mathias CJ, Wang S, Waters DJ, Turek JJ, Low PS, Green MA. Indium-111-DTPA-folate as a potential folate-receptor-targeted radiopharmaceutical. J Nucl Med. 1998; 39:1579–1585.34. Xia W, Hilgenbrink AR, Matteson EL, Lockwood MB, Cheng JX, Low PS. A functional folate receptor is induced during macrophage activation and can be used to target drugs to activated macrophages. Blood. 2009; 113:438–446.
Article35. Kraus VB, McDaniel G, Huebner JL, Stabler TV, Pieper CF, Shipes SW, Petry NA, Low PS, Shen J, McNearney TA, Mitchell P. Direct in vivo evidence of activated macrophages in human osteoarthritis. Osteoarthritis Cartilage. 2016; 24:1613–1621.
Article36. Hilgenbrink AR, Low PS. Folate receptor-mediated drug targeting: from therapeutics to diagnostics. J Pharm Sci. 2005; 94:2135–2146.
Article37. Lu Y, Sega E, Low PS. Folate receptor-targeted immunotherapy: induction of humoral and cellular immunity against hapten-decorated cancer cells. Int J Cancer. 2005; 116:710–719.
Article38. Lu Y, You F, Vlahov I, Westrick E, Fan M, Low PS, Leamon CP. Folate-targeted dinitrophenyl hapten immunotherapy: effect of linker chemistry on antitumor activity and allergic potential. Mol Pharm. 2007; 4:695–706.
Article39. Wen Y, Graybill WS, Previs RA, Hu W, Ivan C, Mangala LS, Zand B, Nick AM, Jennings NB, Dalton HJ, Sehgal V, Ram P, Lee JS, Vivas-Mejia PE, Coleman RL, Sood AK. Immunotherapy targeting folate receptor induces cell death associated with autophagy in ovarian cancer. Clin Cancer Res. 2015; 21:448–459.
Article40. Liang X, Luo M, Wei XW, Ma CC, Yang YH, Shao B, Liu YT, Liu T, Ren J, Liu L, He ZY, Wei YQ. A folate receptor-targeted lipoplex delivering interleukin-15 gene for colon cancer immunotherapy. Oncotarget. 2016; DOI: 10.18632/oncotarget.10537.
Article41. Paulos CM, Varghese B, Widmer WR, Breur GJ, Vlashi E, Low PS. Folate-targeted immunotherapy effectively treats established adjuvant and collagen-induced arthritis. Arthritis Res Ther. 2006; 8:R77.42. Varghese B, Haase N, Low PS. Depletion of folate-receptor-positive macrophages leads to alleviation of symptoms and prolonged survival in two murine models of systemic lupus erythematosus. Mol Pharm. 2007; 4:679–685.
Article43. Yi YS, Ayala-Lopez W, Kularatne SA, Low PS. Folate-targeted hapten immunotherapy of adjuvant-induced arthritis: comparison of hapten potencies. Mol Pharm. 2009; 6:1228–1236.
Article44. Furusho Y, Miyata M, Matsuyama T, Nagai T, Li H, Akasaki Y, Hamada N, Miyauchi T, Ikeda Y, Shirasawa T, Ide K, Tei C. Novel therapy for atherosclerosis using recombinant immunotoxin against folate receptor beta-expressing macrophages. J Am Heart Assoc. 2012; 1:e003079.45. Ferrero-Miliani L, Nielsen OH, Andersen PS, Girardin SE. Chronic inflammation: importance of NOD2 and NALP3 in interleukin-1beta generation. Clin Exp Immunol. 2007; 147:227–235.
Article46. Park YJ, Chung MK, Hwang D, Kim WU. Proteomics in rheumatoid arthritis research. Immune Netw. 2015; 15:177–185.
Article47. Yan S, Yim LY, Lu L, Lau CS, Chan VS. MicroRNA regulation in systemic lupus erythematosus pathogenesis. Immune Netw. 2014; 14:138–148.
Article48. Bae S, Kim H, Yu YS, Lee NE, Kong JM, Kim HR, Hwang YI, Song YW, Kang JS, Lee WJ. Identification of CM1 as a pathogenic factor in inflammatory diseases and cancer. Immune Netw. 2011; 11:175–181.
Article49. Woo Y, Jeong D, Chung DH, Kim HY. The roles of innate lymphoid cells in the development of asthma. Immune Netw. 2014; 14:171–181.
Article50. Italiani P, Boraschi D. New Insights Into Tissue Macrophages: From their origin to the development of memory. Immune Netw. 2015; 15:167–176.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Effect of paternal folate deficiency on placental folate content and folate receptor alpha expression in rats
- Crosstalk Mechanisms Following Targeted Therapy in Head and Neck Cancer
- Folate Receptor Targeted Imaging Using Poly (ethylene glycol)-folate: In Vitro and In Vivo Studies
- Sulfasalazine-induced Megaloblastic Anemia in Normal Serum Folate Level
- Blood Folate Level Determined by a Microplate Reader and Folate Intake Measured by a Weighted Food Record