J Bacteriol Virol.
2016 Sep;46(3):159-166. 10.4167/jbv.2016.46.3.159.
Antiviral Activity of Corylus heterophylla Fisch Against Porcine Epidemic Diarrhea Virus Infection
- Affiliations
-
- 1Department of Beauty Science, Kwangju Women's University, Gwangju, Korea. rerived@kwu.ac.kr
- 2Department of Health Administration, Kwangju Women's University, Gwangju, Korea.
- 3Natural Medicine Research Center, Korea Research Institute of Bioscience & Biotechnology, Cheong-ju, Chungbuk, Korea.
- 4Department of Clinical Pathology, Daejeon Health Sciences College, Daejeon, Korea. artemis3993@gmail.com
- KMID: 2353851
- DOI: http://doi.org/10.4167/jbv.2016.46.3.159
Abstract
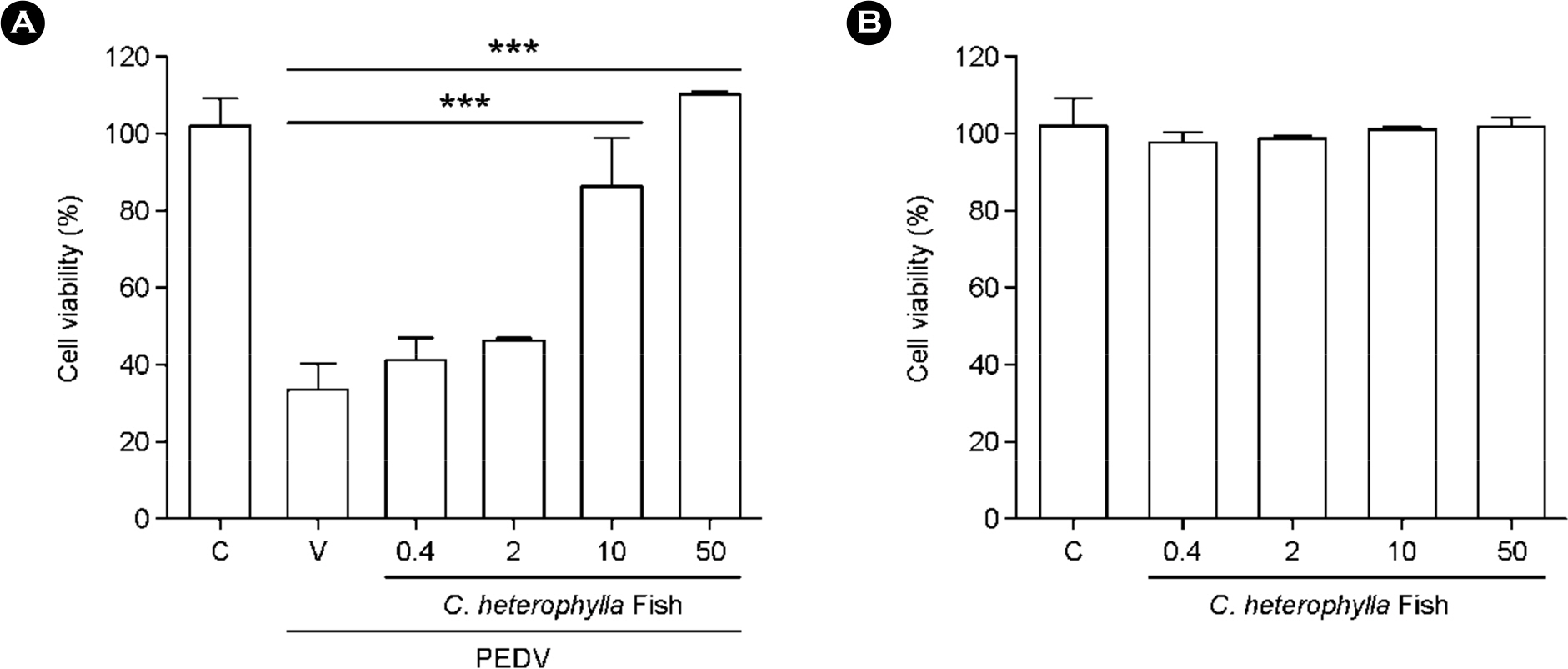
- The porcine epidemic diarrhea virus (PEDV) has recently been shown to cause huge economic losses in the global pork industry. Our results demonstrated that the extract dose-dependently inhibited the replication of PEDV and reduced the visible cytopathic effect (CPE). Treatment with C. heterophylla Fisch extract resulted in marked reduction of PEDVinduced cytokine and chemokine expression. The antiviral activity of C. heterophylla Fisch extract on PEDV replication was found to be primarily exerted at the early stages after infection. Taken together, our data indicate that C. heterophylla Fisch extract may be a good therapeutic agent for use against PEDV and also a potential candidate to be evaluated against other human and animal coronaviruses.
Figure
Reference
-
1). Wood EN. An apparently new syndrome of porcine epidemic diarrhoea. Vet Rec. 1977; 100:243–4.
Article2). Temeeyasen G, Srijangwad A, Tripipat T, Tipsombatboon P, Piriyapongsa J, Phoolcharoen W, et al. Genetic diversity of ORF3 and spike genes of porcine epidemic diarrhea virus in Thailand. Infect Genet Evol. 2014; 21:205–13.
Article3). Sun D, Wang X, Wei S, Chen J, Feng L. Epidemiology and vaccine of porcine epidemic diarrhea virus in China: a mini-review. J Vet Med Sci. 2016; 78:355–63.
Article4). Jung K, Saif LJ. Porcine epidemic diarrhea virus infection: Etiology, epidemiology, pathogenesis and immunoprophylaxis. Vet J. 2015; 204:134–43.
Article5). Gerber PF, Opriessnig T. Detection of immunoglobulin (Ig) A antibodies against porcine epidemic diarrhea virus (PEDV) in fecal and serum samples. MethodsX. 2015; 2:368–73.
Article6). Kweon CH, Kwon BJ, Lee JG, Kwon GO, Kang YB. Derivation of attenuated porcine epidemic diarrhea virus (PEDV) as vaccine candidate. Vaccine. 1999; 17:2546–53.
Article7). Song DS, Yang JS, Oh JS, Han JH, Park BK. Differentiation of a Vero cell adapted porcine epidemic diarrhea virus from Korean field strains by restriction fragment length polymorphism analysis of ORF 3. Vaccine. 2003; 21:1833–42.
Article8). Shen H, Zhang C, Guo P, Liu Z, Zhang J. Effective inhibition of porcine epidemic diarrhea virus by RNA interference in vitro. Virus Genes. 2015; 51:252–9.9). Järvinen P, Palmé A, Orlando Morales L, Lännenpää M, Keinänen M, Sopanen T, et al. Phylogenetic relationships of Betula species (Betulaceae) based on nuclear ADH and chloroplast matK sequences. Am J Bot. 2004; 91:1834–45.10). Zhao TT, Zhang J, Liang LS, Ma QH, Chen X, Zong JW, et al. Expression and Functional Analysis of WRKY Transcription Factors in Chinese Wild Hazel, Corylus heterophylla Fisch. PloS One. 2015; 10:e0135315.11). Plosker GL, Hurst M. Paclitaxel: a pharmacoeconomic review of its use in non-small cell lung cancer. Pharmacoeconomics. 2001; 19:1111–34.12). Kumar S, Mahdi H, Bryant C, Shah JP, Garg G, Munkarah A. Clinical trials and progress with paclitaxel in ovarian cancer. Int J Womens Health. 2010; 2:411–27.
Article13). Gradishar WJ. Taxanes for the treatment of metastatic breast cancer. Breast Cancer. 2012; 6:159–71.
Article14). Choi HJ, Kim JH, Lee CH, Ahn YJ, Song JH, Baek SH, et al. Antiviral activity of quercetin 7-rhamnoside against porcine epidemic diarrhea virus. Antiviral Res. 2009; 81:77–81.
Article15). Chiang LC, Chiang W, Liu MC, Lin CC. In vitro antiviral activities of Caesalpinia pulcherrima and its related flavonoids. J Antimicrob Chemother. 2003; 52:194–8.16). Briskin DP. Medicinal plants and phytomedicines. Linking plant biochemistry and physiology to human health. Plant Physiol. 2000; 124:507–14.
Article17). Jassim SA, Naji MA. Novel antiviral agents: a medicinal plant perspective. J Appl Microbiol. 2003; 95:412–27.
Article18). Vlietinck AJ, Vanden Berghe DA. Can ethnopharmacology contribute to the development of antiviral drugs? J Ethnopharmacol. 1991; 32:141–53.
Article19). Mukhtar M, Arshad M, Ahmad M, Pomerantz RJ, Wigdahl B, Parveen Z. Antiviral potentials of medicinal plants. Virus Res. 2008; 131:111–20.
Article20). Li Y, Jiang R, Ooi LS, But PP, Ooi VE. Antiviral triterpenoids from the medicinal plant Schefflera heptaphylla. Phytother Res. 2007; 21:466–70.21). del Barrio G, Parra F. Evaluation of the antiviral activity of an aqueous extract from Phyllanthus orbicularis. J Ethnopharmacol. 2000; 72:317–22.
Article22). Opitz B, van Laak V, Eitel J, Suttorp N. Innate immune recognition in infectious and noninfectious diseases of the lung. Am J Respir Crit Care Med. 2010; 181:1294–309.
Article23). Gazit R, Gruda R, Elboim M, Arnon TI, Katz G, Achdout H, et al. Lethal influenza infection in the absence of the natural killer cell receptor gene Ncr1. Nat Immunol. 2006; 7:517–23.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Recent vaccine technology in industrial animals
- Sequence analysis of the spike gene of Porcine epidemic diarrhea virus isolated from South China during 2011–2015
- Reemergence of porcine epidemic diarrhea virus on Jeju Island
- Genetic diversity of nucleocapsid genes of recent porcine epidemic diarrhea viruses isolated in Korea
- Porcine epidemic diarrhea: a review of current epidemiology and available vaccines