J Breast Cancer.
2014 Mar;17(1):25-32.
Dimethyl Sulfoxide Suppresses Mouse 4T1 Breast Cancer Growth by Modulating Tumor-Associated Macrophage Differentiation
- Affiliations
-
- 1State Key Laboratory of Biotherapy and Cancer Center, West China Hospital, West China Medical School, Sichuan University, Chengdu, China. yuquawei@vip.sina.com
- 2Department of Medical Oncology, West China Hospital, West China Medical School, Sichuan University, Chengdu, China.
Abstract
- PURPOSE
The universal organic solvent dimethyl sulfoxide (DMSO) can be used as a differentiation inducer of many cancer cells and has been widely used as a solvent in laboratories. However, its effects on breast cancer cells are not well understood. The aim of this study is to investigate the effect and associated mechanisms of DMSO on mouse breast cancer.
METHODS
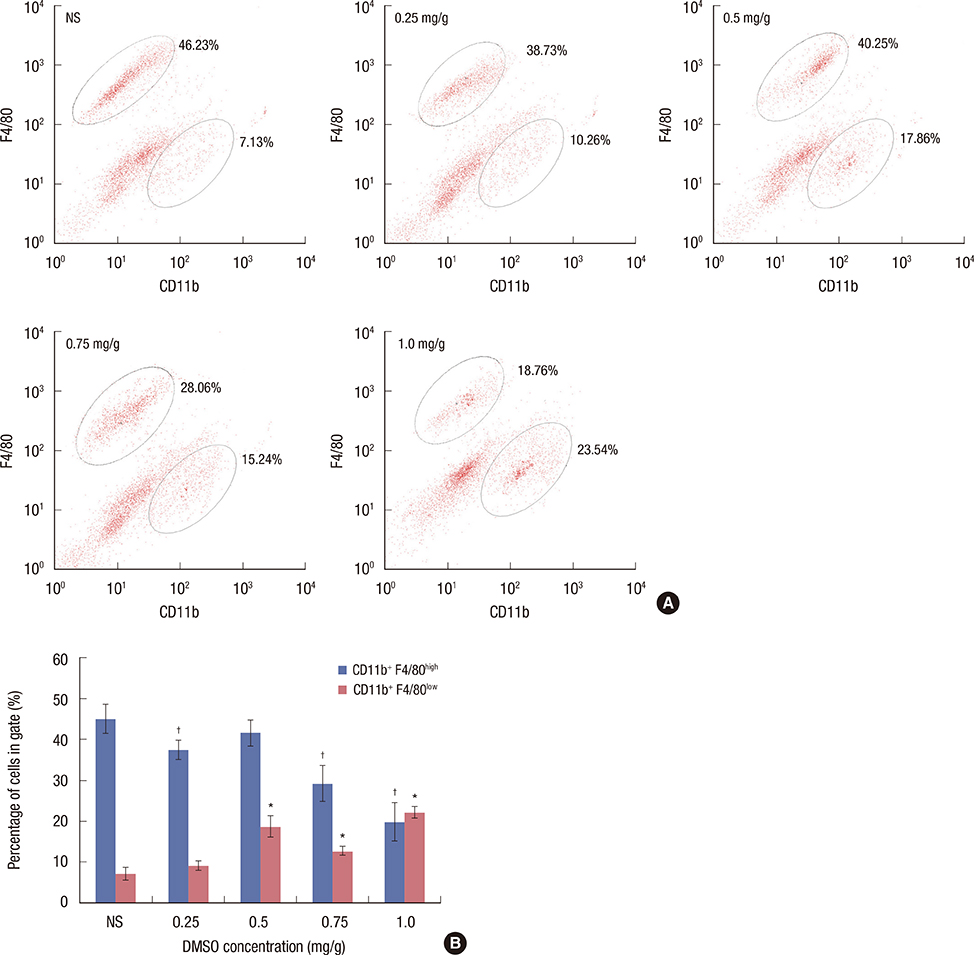
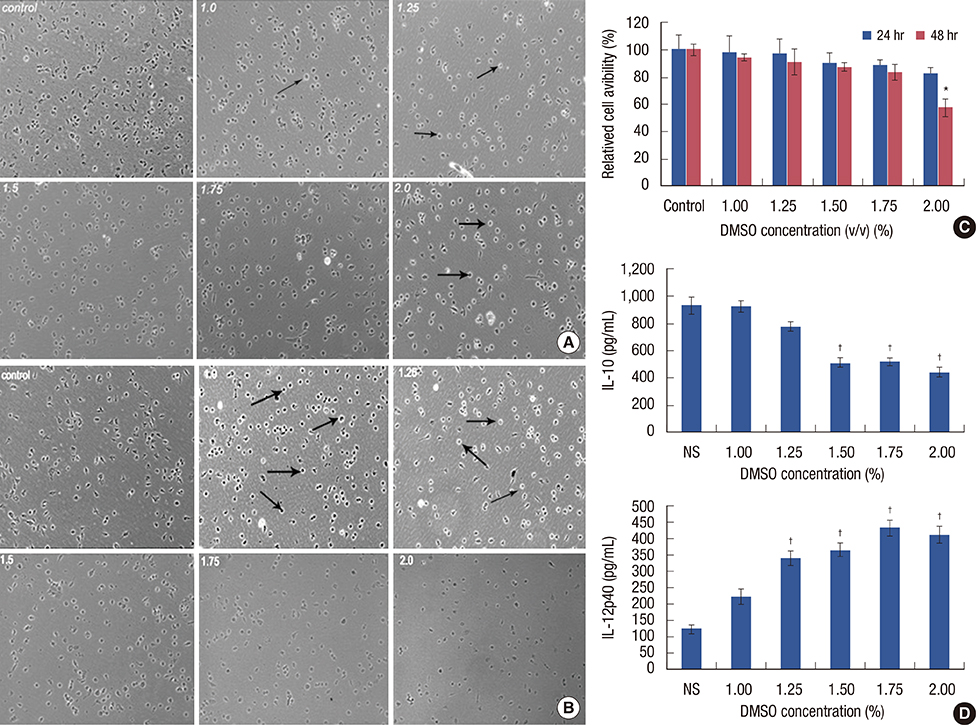
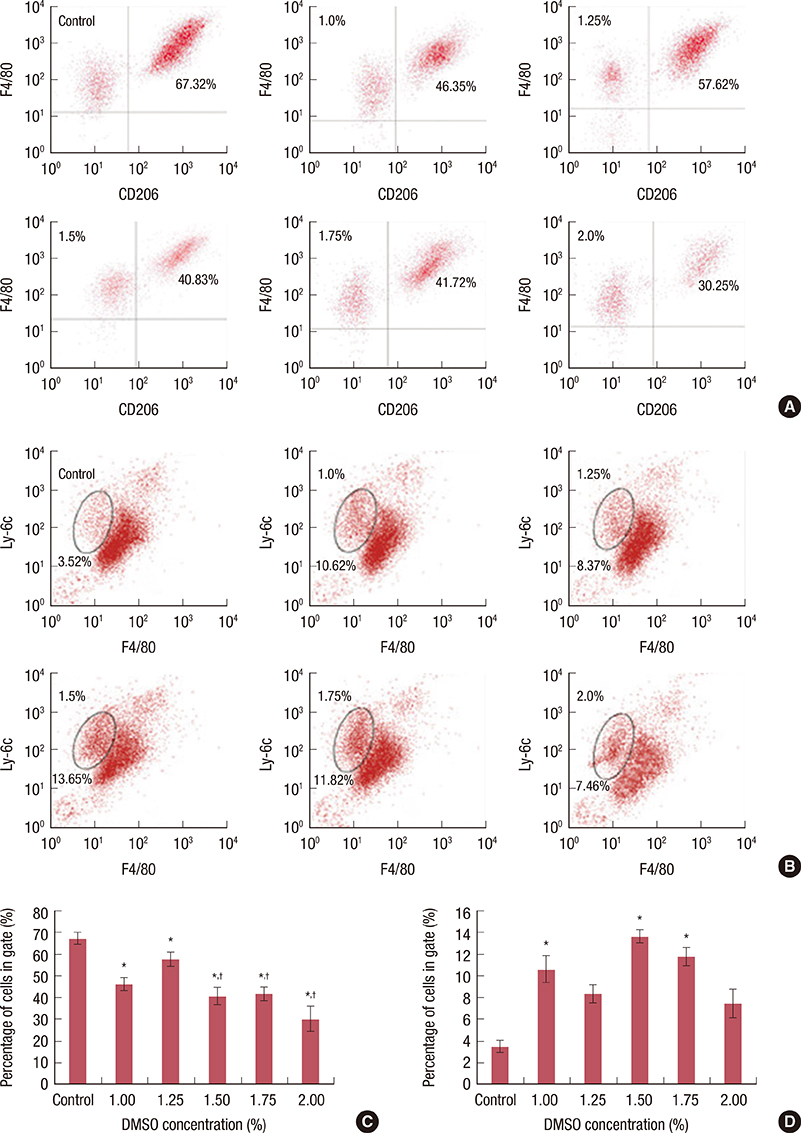
We applied DMSO to observe the effect on tumors in a mouse breast cancer model. Tumor-associated macrophages (TAMs) were tested by flow cytometry. Ex vivo tumor microenvironment was imitated by 4T1 cultured cell conditioned medium. Enzyme-linked immunosorbent assays were performed to detect interleukin (IL)-10 and IL-12 expression in medium. To investigate the cytotoxicity of DMSO on TAMs, 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assays were performed.
RESULTS
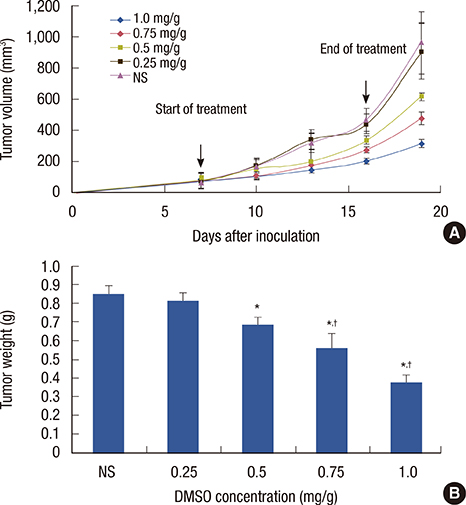
We found that DMSO produced tumor retardation when injected into mouse peritoneal cavities in a certain concentration range (0.5-1.0 mg/g). Furthermore, as detected by flow cytometry, TAM subtypes were found to be transformed. We further imitated a tumor microenvironment in vitro by using 4T1 cultured cell conditioned medium. Similarly, by using low concentration DMSO (1.0%-2.0% v/v), TAMs were induced to polarize to the classically activated macrophage (M1-type) and inhibited from polarizing into the alternatively activated macrophage (M2-type) in the conditioned medium. IL-10 expression in tumors was reduced, while IL-12 was increased compared with the control. Furthermore, we reported that 2.0% (v/v) DMSO could lead to cytotoxicity in peritoneal macrophages after 48 hours in MTT assays.
CONCLUSION
Our findings suggest that DMSO could exert antitumor effects in 4T1 cancer-bearing mice by reversing TAM orientation and polarization from M2- to M1-type TAMs. These data may provide novel insight into studying breast cancer immunotherapy.
Keyword
MeSH Terms
-
Animals
Breast Neoplasms*
Breast*
Cells, Cultured
Culture Media, Conditioned
Dimethyl Sulfoxide*
Enzyme-Linked Immunosorbent Assay
Flow Cytometry
Immunotherapy
Interleukin-10
Interleukin-12
Interleukins
Macrophages*
Macrophages, Peritoneal
Mice*
Tumor Microenvironment
Culture Media, Conditioned
Dimethyl Sulfoxide
Interleukin-10
Interleukin-12
Interleukins
Figure
Reference
-
1. Huberman E, Heckman C, Langenbach R. Stimulation of differentiated functions in human melanoma cells by tumor-promoting agents and dimethyl sulfoxide. Cancer Res. 1979; 39(7 Pt 1):2618–2624.2. Breitman TR, He RY. Combinations of retinoic acid with either sodium butyrate, dimethyl sulfoxide, or hexamethylene bisacetamide synergistically induce differentiation of the human myeloid leukemia cell line HL60. Cancer Res. 1990; 50:6268–6273.3. Levy J, Terada M, Rifkind RA, Marks PA. Induction of erythroid differentiation by dimethylsulfoxide in cells infected with Friend virus: relationship to the cell cycle. Proc Natl Acad Sci U S A. 1975; 72:28–32.
Article4. Collins SJ, Ruscetti FW, Gallagher RE, Gallo RC. Terminal differentiation of human promyelocytic leukemia cells induced by dimethyl sulfoxide and other polar compounds. Proc Natl Acad Sci U S A. 1978; 75:2458–2462.
Article5. Ferrero D, Tarella C, Gallo E, Ruscetti FW, Breitman TR. Terminal differentiation of the human promyelocytic leukemia cell line, HL-60, in the absence of cell proliferation. Cancer Res. 1982; 42:4421–4426.6. Takasaki S, Leive L. Induction by dimethyl sulfoxide of Fc fragment-mediated phagocytosis in the mouse macrophage-like cell line P388D1. Proc Natl Acad Sci U S A. 1982; 79:4980–4984.
Article7. Takenaga K. Enhanced metastatic potential of cloned low-metastatic Lewis lung carcinoma cells treated in vitro with dimethyl sulfoxide. Cancer Res. 1984; 44:1122–1127.8. Fujiu K, Manabe I, Nagai R. Renal collecting duct epithelial cells regulate inflammation in tubulointerstitial damage in mice. J Clin Invest. 2011; 121:3425–3441.
Article9. Solinas G, Schiarea S, Liguori M, Fabbri M, Pesce S, Zammataro L, et al. Tumor-conditioned macrophages secrete migration-stimulating factor: a new marker for M2-polarization, influencing tumor cell motility. J Immunol. 2010; 185:642–652.
Article10. Mantovani A, Sozzani S, Locati M, Allavena P, Sica A. Macrophage polarization: tumor-associated macrophages as a paradigm for polarized M2 mononuclear phagocytes. Trends Immunol. 2002; 23:549–555.
Article11. Sica A, Schioppa T, Mantovani A, Allavena P. Tumour-associated macrophages are a distinct M2 polarised population promoting tumour progression: potential targets of anti-cancer therapy. Eur J Cancer. 2006; 42:717–727.
Article12. Chen J, Yao Y, Gong C, Yu F, Su S, Chen J, et al. CCL18 from tumor-associated macrophages promotes breast cancer metastasis via PITPNM3. Cancer Cell. 2011; 19:541–555.
Article13. Ivanović Matić S, Dinić S, Mihailović M, Grigorov I, Bogojević D, Poznanović G. Acute-phase protein expression in DMSO-intoxicated rats. Toxicol Lett. 2004; 147:153–159.
Article14. Caras I, Tucureanu C, Lerescu L, Pitica R, Melinceanu L, Neagu S, et al. Influence of tumor cell culture supernatants on macrophage functional polarization: in vitro models of macrophage-tumor environment interaction. Tumori. 2011; 97:647–654.
Article15. Wang CC, Lin SY, Lai YH, Liu YJ, Hsu YL, Chen JJ. Dimethyl sulfoxide promotes the multiple functions of the tumor suppressor HLJ1 through activator protein-1 activation in NSCLC cells. PLoS One. 2012; 7:e33772.
Article16. Pommier RF, Woltering EA, Milo G, Fletcher WS. Synergistic cytotoxicity between dimethyl sulfoxide and antineoplastic agents against ovarian cancer in vitro. Am J Obstet Gynecol. 1988; 159:848–852.17. Koiri RK, Trigun SK. Dimethyl sulfoxide activates tumor necrosis factorα-p53 mediated apoptosis and down regulates D-fructose-6-phosphate-2-kinase and lactate dehydrogenase-5 in Dalton's lymphoma in vivo. Leuk Res. 2011; 35:950–956.
Article18. Hoang BX, Tran DM, Tran HQ, Nguyen PT, Pham TD, Dang HV, et al. Dimethyl sulfoxide and sodium bicarbonate in the treatment of refractory cancer pain. J Pain Palliat Care Pharmacother. 2011; 25:19–24.
Article19. Zylber-Katz E, Glazer RI. Phospholipid- and Ca2+-dependent protein kinase activity and protein phosphorylation patterns in the differentiation of human promyelocytic leukemia cell line HL-60. Cancer Res. 1985; 45:5159–5164.20. Morley P, Whitfield JF. The differentiation inducer, dimethyl sulfoxide, transiently increases the intracellular calcium ion concentration in various cell types. J Cell Physiol. 1993; 156:219–225.
Article21. Tsao D, Morita A, Bella A Jr, Luu P, Kim YS. Differential effects of sodium butyrate, dimethyl sulfoxide, and retinoic acid on membrane-associated antigen, enzymes, and glycoproteins of human rectal adenocarcinoma cells. Cancer Res. 1982; 42:1052–1058.22. Scher BM, Scher W, Robinson A, Waxman S. DNA ligase and DNase activities in mouse erythroleukemia cells during dimethyl sulfoxide-induced differentiation. Cancer Res. 1982; 42:1300–1306.23. Hollebeeck S, Raas T, Piront N, Schneider YJ, Toussaint O, Larondelle Y, et al. Dimethyl sulfoxide (DMSO) attenuates the inflammatory response in the in vitro intestinal Caco-2 cell model. Toxicol Lett. 2011; 206:268–275.
Article24. Kelly KA, Hill MR, Youkhana K, Wanker F, Gimble JM. Dimethyl sulfoxide modulates NF-kappa B and cytokine activation in lipopolysaccharide-treated murine macrophages. Infect Immun. 1994; 62:3122–3128.
Article25. Wang YC, He F, Feng F, Liu XW, Dong GY, Qin HY, et al. Notch signaling determines the M1 versus M2 polarization of macrophages in antitumor immune responses. Cancer Res. 2010; 70:4840–4849.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Effect of Dimethyl Sulfoxide (DMSO) Monotherapy in Treatment of Interstitial Cystitis
- Peanut sprout tea extract inhibits lung metastasis of 4T1 murine mammary carcinoma cells by suppressing the crosstalk between cancer cells and macrophages in BALB/c mice
- Effect of growth factors and differentiation inducer DMSO on the anaplastic thyroid carcinoma cell line, SNU-80
- The Effect of 5% Imiquimod Cream on C57BL/6 Mouse Melanoma Growth
- Effects of Trypsin, Collagenase and Dimethyl Sulfoxide on Dissociation of Rat Heart Cells