J Korean Med Sci.
2015 Oct;30(10):1503-1508. 10.3346/jkms.2015.30.10.1503.
Effect of Propofol and Desflurane on Immune Cell Populations in Breast Cancer Patients: A Randomized Trial
- Affiliations
-
- 1Department of Anesthesiology and Pain Medicine, School of Medicine, Ewha Womans University, Seoul, Korea. baikhj@ewha.ac.kr
- KMID: 2344186
- DOI: http://doi.org/10.3346/jkms.2015.30.10.1503
Abstract
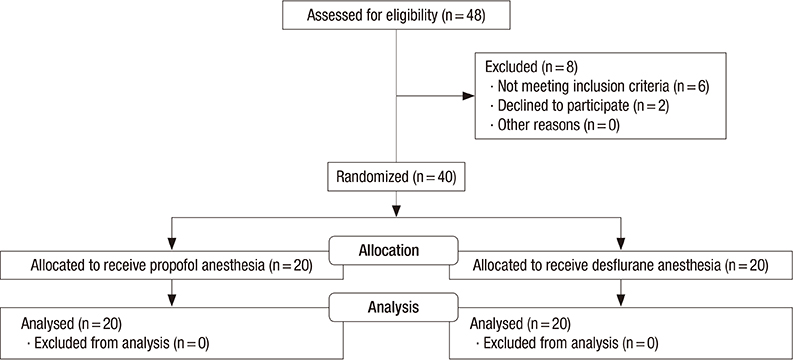
- Several factors can affect the perioperative immune function. We evaluated the effect of propofol and desflurane anesthesia on the surgery-induced immune perturbation in patients undergoing breast cancer surgery. The patients were randomly assigned to receive propofol (n = 20) or desflurane (n = 20) anesthesia. The total and differential white blood cell counts were determined with lymphocyte subpopulations before and 1 hr after anesthesia induction and at 24 hr postoperatively. Plasma concentrations of interleukin (IL)-2 and IL-4 were also measured. Both propofol and desflurane anesthesia preserved the IL-2/IL-4 and CD4+/CD8+ T cell ratio. Leukocytes were lower in the propofol group than in the desflurane group at 1 hr after induction (median [quartiles], 4.98 [3.87-6.31] vs. 5.84 [5.18-7.94] 10(3)/microL) and 24 hr postoperatively (6.92 [5.54-6.86] vs. 7.62 [6.22-9.21] 10(3)/microL). NK cells significantly decreased 1 hr after induction in the propofol group (0.41 [0.34-0.53] to 0.25 [0.21-0.33] 10(3)/microL), but not in the desflurane group (0.33 [0.29-0.48] to 0.38 [0.30-0.56] 10(3)/microL). Our findings indicate that both propofol and desflurane anesthesia for breast cancer surgery induce a favorable immune response in terms of preservation of IL-2/IL-4 and CD4+/CD8+ T cell ratio in the perioperative period. With respect to leukocytes and NK cells, desflurane anesthesia is associated with less adverse immune responses than propofol anesthesia during surgery for breast cancer. (Clinical trial registration at https://cris.nih.go.kr/cris number: KCT0000939)
Keyword
MeSH Terms
-
Adolescent
Adult
Aged
Aged, 80 and over
Anesthesia/adverse effects
Anesthetics, Inhalation/*therapeutic use
Anesthetics, Intravenous/*therapeutic use
Breast Neoplasms/immunology/*surgery
*CD4-CD8 Ratio
Female
Humans
Interleukin-2/blood
Interleukin-4/blood
Isoflurane/*analogs & derivatives/therapeutic use
Middle Aged
Postoperative Period
Propofol/*therapeutic use
Young Adult
Anesthetics, Inhalation
Anesthetics, Intravenous
Interleukin-2
Interleukin-4
Isoflurane
Propofol
Figure
Reference
-
1. Jemal A, Bray F, Center MM, Ferlay J, Ward E, Forman D. Global cancer statistics. CA Cancer J Clin. 2011; 61:69–90.2. Tavare AN, Perry NJ, Benzonana LL, Takata M, Ma D. Cancer recurrence after surgery: direct and indirect effects of anesthetic agents. Int J Cancer. 2012; 130:1237–1250.3. Tracey KJ. Reflex control of immunity. Nat Rev Immunol. 2009; 9:418–428.4. Mansi JL, Gogas H, Bliss JM, Gazet JC, Berger U, Coombes RC. Outcome of primary-breast-cancer patients with micrometastases: a long-term follow-up study. Lancet. 1999; 354:197–202.5. Heaney A, Buggy DJ. Can anaesthetic and analgesic techniques affect cancer recurrence or metastasis? Br J Anaesth. 2012; 109:i17–i28.6. Niwa H, Rowbotham DJ, Lambert DG, Buggy DJ. Can anesthetic techniques or drugs affect cancer recurrence in patients undergoing cancer surgery? J Anesth. 2013; 27:731–741.7. Green JS, Tsui BC. Impact of anesthesia for cancer surgery: continuing professional development. Can J Anaesth. 2013; 60:1248–1269.8. Neeman E, Ben-Eliyahu S. Surgery and stress promote cancer metastasis: new outlooks on perioperative mediating mechanisms and immune involvement. Brain Behav Immun. 2013; 30:S32–S40.9. Wakefield CH, Carey PD, Foulds S, Monson JR, Guillou PJ. Changes in major histocompatibility complex class II expression in monocytes and T cells of patients developing infection after surgery. Br J Surg. 1993; 80:205–209.10. White PF, Tang J, Wender RH, Yumul R, Stokes OJ, Sloninsky A, Naruse R, Kariger R, Norel E, Mandel S, et al. Desflurane versus sevoflurane for maintenance of outpatient anesthesia: the effect on early versus late recovery and perioperative coughing. Anesth Analg. 2009; 109:387–393.11. Mahmoud NA, Rose DJ, Laurence AS. Desflurane or sevoflurane for gynaecological day-case anaesthesia with spontaneous respiration? Anaesthesia. 2001; 56:171–174.12. Kushida A, Inada T, Shingu K. Enhancement of antitumor immunity after propofol treatment in mice. Immunopharmacol Immunotoxicol. 2007; 29:477–486.13. Melamed R, Bar-Yosef S, Shakhar G, Shakhar K, Ben-Eliyahu S. Suppression of natural killer cell activity and promotion of tumor metastasis by ketamine, thiopental, and halothane, but not by propofol: mediating mechanisms and prophylactic measures. Anesth Analg. 2003; 97:1331–1339.14. Müller-Edenborn B, Roth-Z'graggen B, Bartnicka K, Borgeat A, Hoos A, Borsig L, Beck-Schimmer B. Volatile anesthetics reduce invasion of colorectal cancer cells through down-regulation of matrix metalloproteinase-9. Anesthesiology. 2012; 117:293–301.15. Ke JJ, Zhan J, Feng XB, Wu Y, Rao Y, Wang YL. A comparison of the effect of total intravenous anaesthesia with propofol and remifentanil and inhalational anaesthesia with isoflurane on the release of pro- and anti-inflammatory cytokines in patients undergoing open cholecystectomy. Anaesth Intensive Care. 2008; 36:74–78.16. Tylman M, Sarbinowski R, Bengtson JP, Kvarnström A, Bengtsson A. Inflammatory response in patients undergoing colorectal cancer surgery: the effect of two different anesthetic techniques. Minerva Anestesiol. 2011; 77:275–282.17. Schilling T, Kozian A, Kretzschmar M, Huth C, Welte T, Bühling F, Hedenstierna G, Hachenberg T. Effects of propofol and desflurane anaesthesia on the alveolar inflammatory response to one-lung ventilation. Br J Anaesth. 2007; 99:368–375.18. Baki ED, Aldemir M, Kokulu S, Koca HB, Ela Y, Sivaci RG, öztürk NK, Emmiler M, Adali F, Uzel H. Comparison of the effects of desflurane and propofol anesthesia on the inflammatory response and s100beta protein during coronary artery bypass grafting. Inflammation. 2013; 36:1327–1333.19. Schreiber RD, Old LJ, Smyth MJ. Cancer immunoediting: integrating immunity's roles in cancer suppression and promotion. Science. 2011; 331:1565–1570.20. Decker D, Schondorf M, Bidlingmaier F, Hirner A, von Ruecker AA. Surgical stress induces a shift in the type-1/type-2 T-helper cell balance, suggesting down-regulation of cell-mediated and up-regulation of antibody-mediated immunity commensurate to the trauma. Surgery. 1996; 119:316–325.21. Cata JP, Bauer M, Sokari T, Ramirez MF, Mason D, Plautz G, Kurz A. Effects of surgery, general anesthesia, and perioperative epidural analgesia on the immune function of patients with non-small cell lung cancer. J Clin Anesth. 2013; 25:255–262.22. Inada T, Yamanouchi Y, Jomura S, Sakamoto S, Takahashi M, Kambara T, Shingu K. Effect of propofol and isoflurane anaesthesia on the immune response to surgery. Anaesthesia. 2004; 59:954–959.23. Liu S, Wang B, Li S, Zhou Y, An L, Wang Y, Lv H, Zhang G, Fang F, Liu Z, et al. Immune cell populations decrease during craniotomy under general anesthesia. Anesth Analg. 2011; 113:572–577.24. Raman D, Baugher PJ, Thu YM, Richmond A. Role of chemokines in tumor growth. Cancer Lett. 2007; 256:137–165.25. Han YF, Zhao J, Ma LY, Yin JH, Chang WJ, Zhang HW, Cao GW. Factors predicting occurrence and prognosis of hepatitis-B-virus-related hepatocellular carcinoma. World J Gastroenterol. 2011; 17:4258–4270.26. Snyder GL, Greenberg S. Effect of anaesthetic technique and other perioperative factors on cancer recurrence. Br J Anaesth. 2010; 105:106–115.27. Wada H, Seki S, Takahashi T, Kawarabayashi N, Higuchi H, Habu Y, Sugahara S, Kazama T. Combined spinal and general anesthesia attenuates liver metastasis by preserving TH1/TH2 cytokine balance. Anesthesiology. 2007; 106:499–506.28. Hori Y, Ibuki T, Hosokawa T, Tanaka Y. The effects of neurosurgical stress on peripheral lymphocyte subpopulations. J Clin Anesth. 2003; 15:1–8.29. Yeager MP, Procopio MA, DeLeo JA, Arruda JL, Hildebrandt L, Howell AL. Intravenous fentanyl increases natural killer cell cytotoxicity and circulating CD16(+) lymphocytes in humans. Anesth Analg. 2002; 94:94–99.30. Beilin B, Shavit Y, Hart J, Mordashov B, Cohn S, Notti I, Bessler H. Effects of anesthesia based on large versus small doses of fentanyl on natural killer cell cytotoxicity in the perioperative period. Anesth Analg. 1996; 82:492–497.31. Yardeni IZ, Beilin B, Mayburd E, Alcalay Y, Bessler H. Relationship between fentanyl dosage and immune function in the postoperative period. J Opioid Manag. 2008; 4:27–33.32. Sanders RD, Weimann J, Maze M. Biologic effects of nitrous oxide: a mechanistic and toxicologic review. Anesthesiology. 2008; 109:707–722.33. Fleischmann E, Marschalek C, Schlemitz K, Dalton JE, Gruenberger T, Herbst F, Kurz A, Sessler DI. Nitrous oxide may not increase the risk of cancer recurrence after colorectal surgery: a follow-up of a randomized controlled trial. BMC Anesthesiol. 2009; 9:1.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Optimal effect-site concentration of remifentanil for preventing development of hypertension during tracheal intubation with inhaled desflurane induction
- Effect of equipotent doses of propofol and sevoflurane on endoplasmic reticulum stress during breast cancer surgery
- Effect of remimazolam on intraoperative hemodynamic stability in patients undergoing cerebrovascular bypass surgery: a prospective randomized controlled trial
- The Effects of Desflurane on Anesthesia Induction
- A low dose of droperidol decreases the desflurane concentration needed during breast cancer surgery: a randomized double-blinded study