Nat Prod Sci.
2016 Jun;22(2):93-101. 10.20307/nps.2016.22.2.93.
Qualitative and Quantitative Analysis of Thirteen Marker Components in Traditional Korean Formula, Samryeongbaekchul-san using an Ultra-Performance Liquid Chromatography Equipped with Electrospray Ionization Tandem Mass Spectrometry
- Affiliations
-
- 1K-her Research Center, Korea Institute of Oriental Medicine, 1672 Yuseongdae-ro, Yuseong-gu, Daejeon 34054, Korea. hkshin@kiom.re.kr
- KMID: 2328873
- DOI: http://doi.org/10.20307/nps.2016.22.2.93
Abstract
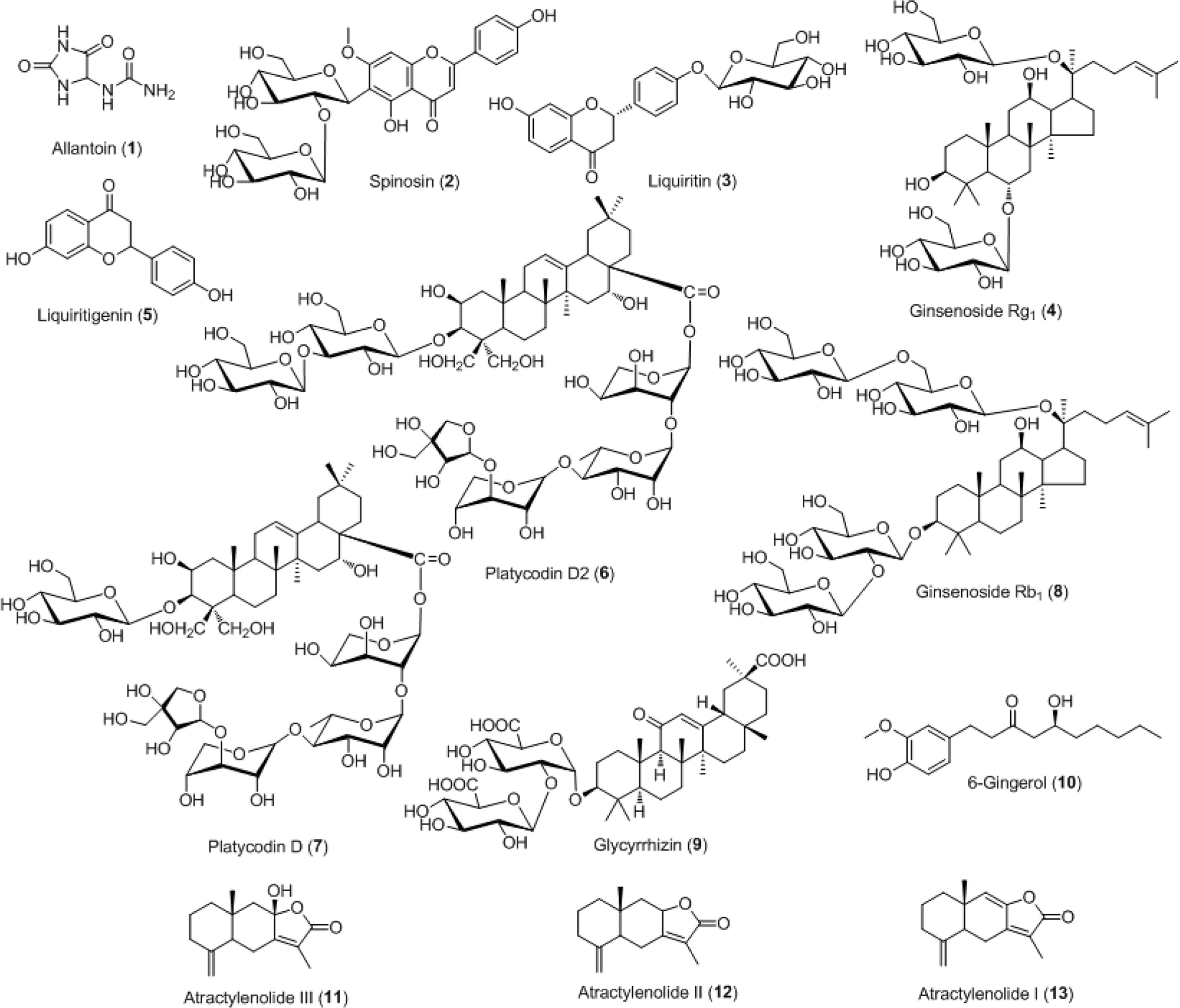
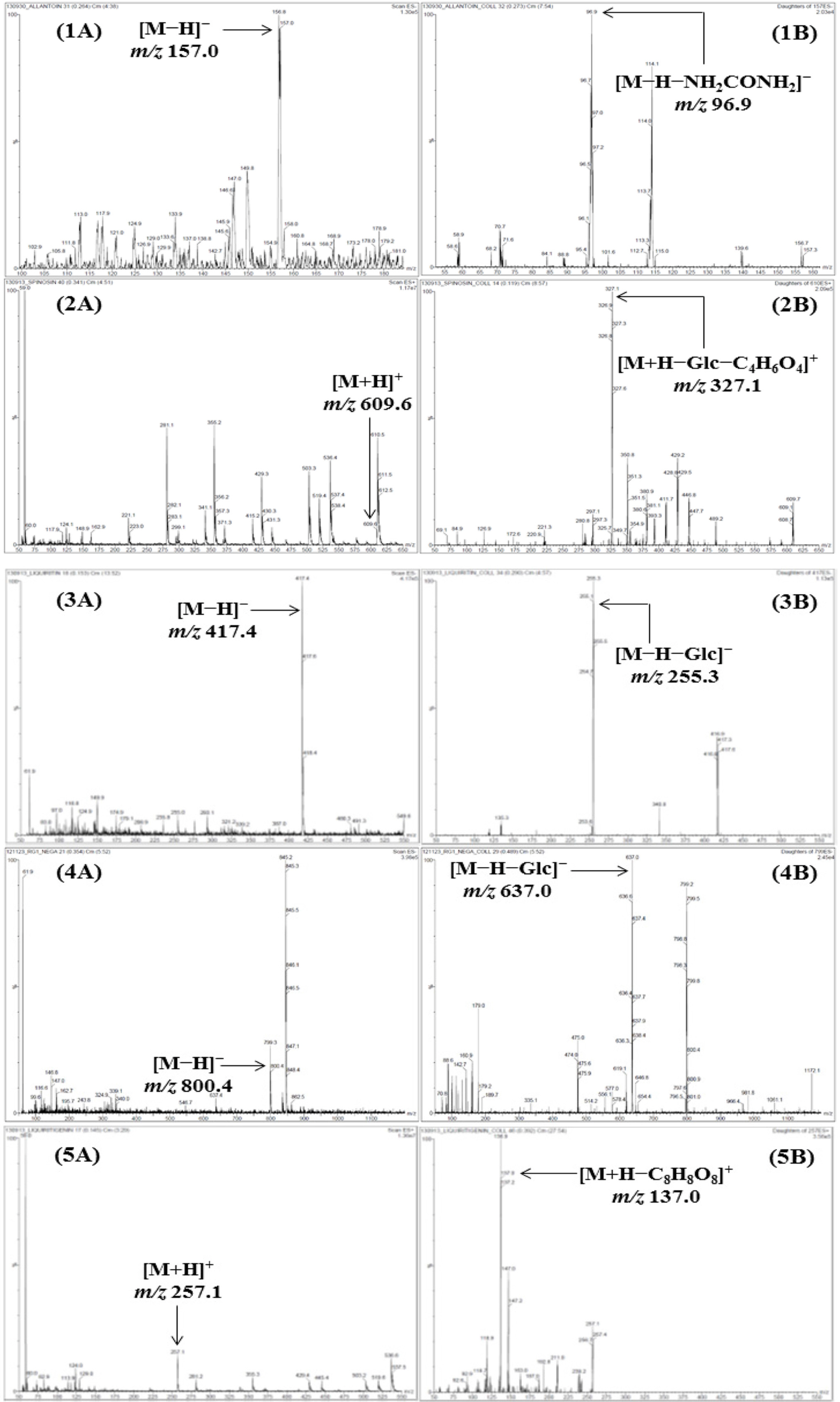
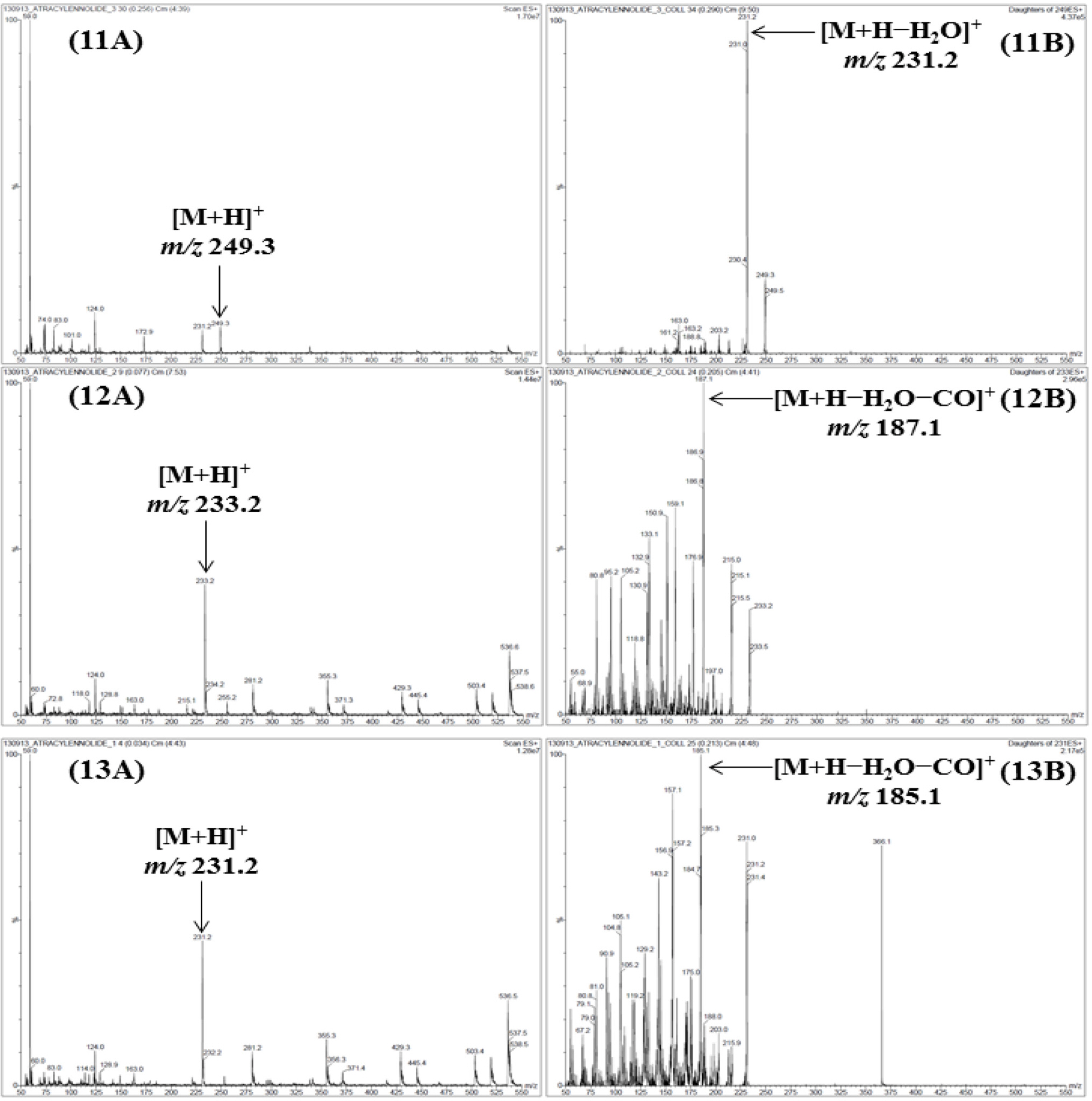
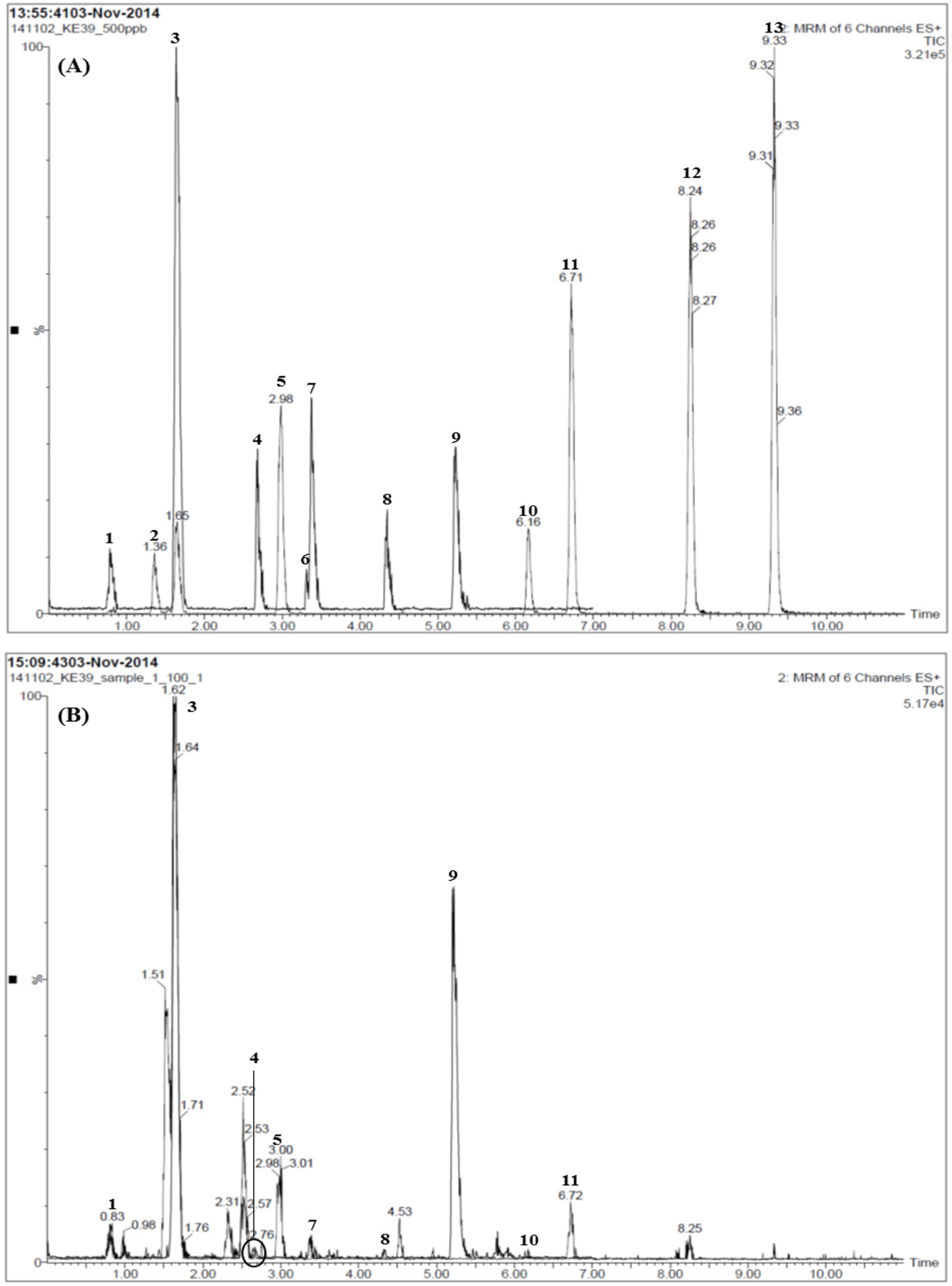
- For efficient quality control of the Samryeongbaekchul-san decoction, a powerful and accurate an ultra-performance liquid chromatography (UPLC) coupled with electrospray ionization (ESI) tandem mass spectrometry (MS) method was developed for quantitative analysis of the thirteen constituents: allantoin (1), spinosin (2), liquiritin (3), ginsenoside Rg1 (4), liquiritigenin (5), platycodin D2 (6), platycodin D (7), ginsenoside Rb1 (8), glycyrrhizin (9), 6-gingerol (10), atractylenolide III (11), atractylenolide II (12), and atractylenolide I (13). Separation of the compounds 1 - 13 was performed on a UPLC BEH Câ‚₈ column (2.1 × 100 mm, 1.7 µm) at a column temperature of 40 ℃ with a gradient solvent system of 0.1% (v/v) formic acid aqueous-acetonitrile. The flow rate and injection volume were 0.3 mL/min and 2.0 µL. Calibration curves of all compounds were showed good linearity with values of the correlation coefficient ≥ 0.9920 within the test ranges. The values of limits of detection and quantification for all analytes were 0.04 - 4.53 ng/mL and 0.13 - 13.60 ng/mL. The result of an experiment, compounds 2, 6, 12, and 13 were not detected while compounds 1, 3 - 5, and 7 - 11 were detected with 1,570.42, 5,239.85, 299.35, 318.88, 562.27, 340.87, 12,253.69, 73.80, and 115.01 µg/g, respectively.
MeSH Terms
Figure
Reference
-
(1). Qiu J.Nat. Rev. Drug Discov. 2007; 6:506–507.(2). Wang L., Zhou G. B., Liu P., Song J. H., Liang Y., Yan X. J., Xu F., Wang B. S., Mao J. H., Shen Z. X., Chen S. J., Chen Z.Proc. Natl. Acad. Sci. U. S. A. 2008; 105:4826–4831.(3). Chen H., Wu M., Kubo K. Y. J.Ethnopharmacol. 2012; 142:80–85.(4). Lee S. R.Korean J. Orient. Physiol. Pathol. 2009; 23:374–380.(5). Heo J.Dongeuibogam; Namsandang: Seoul. 2004; 440.(6). Yang Q. H., Xu Y. J., Liu Y. Z., Liang Y. J., Feng G. F., Zhang Y. P., Xing H. J., Yan H. Z, Li Y. Y.Evid. Based Complement. Alternat. Med. 2014; 2014:671013–671021.(7). Yang Q. H., Xu Y. J., Feng G. F., Hu C. F., Zhang Y. P., Cheng S. B., Wagn Y. P., Gong X. W. Afr. J.Tradit. Complement. Altern. Med. 2013; 11:213–221.(8). Liu X., Jia Y., Iiu Q., Wu J.Lishizhen Med. Mater. Med. Res. 2011; 22:2671–2673.(9). You Y., Liu U., Gao S.Chinese J. Experiment. Trad. Med. Formulae. 2012; 18:136–140.(10). Turner R., Stamp L. K., Kettle A. J. J.Chromatogr. B Analyt. Technol. Biomed. Life Sci. 2012; 891-892:85–89.(11). Ren Y., Wang P., Wu C., Zhang J., Niu C.Biomed. Chromatogr. 2013; 27:17–26.
Article(12). Wang Y., Yang L., He Y. Q., Wang C. H., Welbeck E. W., Bligh S. W., Wang Z. T.Rapid Commun. Mass Spectrom. 2008; 22:1767–1778.(13). Zhao J., Su C., Yang C., Liu M., Tang L., Su W., Liu Z. J.Pharm. Biomed. Anal. 2012; 64-65:94–97.(14). Tan G., Zhu Z., Jing J., Lv L., Lou Z., Zhang G., Chai Y.Biomed. Chromatogr. 2011; 25:913–924.(15). Na Y. C., Ha Y. W., Kim Y. S., Kim K. J. J.Chromatogr. A. 2008; 1189:467–475.(16). Chen L., Qi J., Chang Y. X., Zhu D., Yu B. J.Pharm. Biomed. Anal. 2009; 50:127–137.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Quantitative Analysis of Eleven Bioactive Constituents of a Traditional Herbal Medicine, Yeonggyechulgam-tang using, Liquid Chromatography-Electrospray Ionization Tandem Mass Spectrometry
- Quantification of apixaban in human plasma using ultra performance liquid chromatography coupled with tandem mass spectrometry
- Simultaneous Screening of 177 Drugs of Abuse in Urine Using Ultra-performance Liquid Chromatography with Tandem Mass Spectrometry in Drug-intoxicated Patients
- Determination of candesartan or olmesartan in hypertensive patient plasma using UPLC-MS/MS
- Mass spectrometry based cellular phosphoinositides profiling and phospholipid analysis: A brief review