J Korean Acad Periodontol.
2009 Aug;39(Suppl):231-237.
Co-expression of CdtA and CdtC subunits of cytolethal distending toxin from Aggregatibacter actinomycetemcomitans
- Affiliations
-
- 1Department of Periodontology, College of Dentistry, Chonbuk National University, Korea. cbuperio@chonbuk.ac.kr
- 2Department of Oral Microbiology and Institute of Oral Bioscience, Brain Korea 21 Project, Chonbuk National University, Korea.
Abstract
-
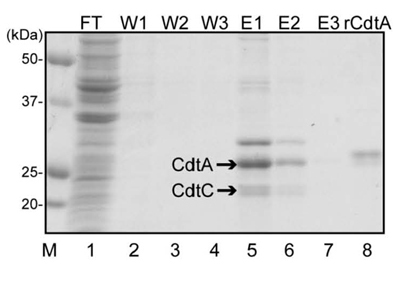
PURPOSE: Cytolethal distending toxin (CDT) is a family of heat-labile cytotoxins produced by several gram-negative mucosa-associated pathogens, including Aggregatibacter actinomycetemcomitans. CDT is well known to be capable of inducing growth arrest, morphological alterations, and eventually death in various cells. CDT belongs to a tripartite AB2 toxin (CdtB: the enzymatic A subunit ; CdtA and CdtC: the heterodimeric B subunit). Previous studies proposed that CdtA and CdtC together bind to a cell surface receptor and glycolipids act as a receptor for A. actinomycetemcomitans CDT (AaCDT). In this study, recombinant CdtA and CdtC proteins of AaCDT were co-expressed in a bacterial expression system and tested for their affinity for GM1 ganglioside.
METHODS
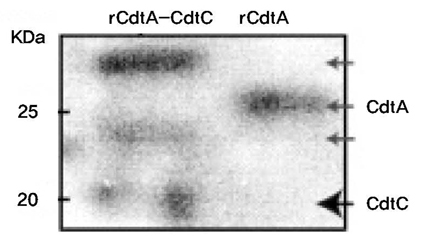
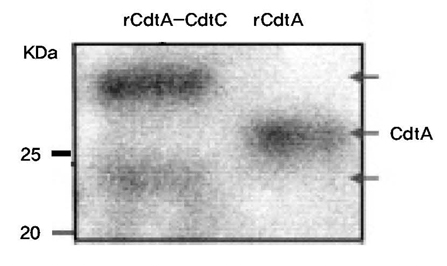
The genes for CdtA and CdtC from A. actinomycetemcomitans Y4 were utilized to construct the expression vectors, pRSET-cdtA and pET28a-cdtC. Both CdtA and CdtC proteins were expressed in Escherichia coli BL21(DE3) and then purified using hexahistidine (His6) tag. The identity of purified protein was confirmed by anti-His6 antibody and monoclonal anti-CdtA antibody. Furthermore, the affinity of recombinant protein to GM1 ganglioside was checked through ELISA.
RESULTS
Recombinant CdtA and CdtC proteins were expressed as soluble proteins and reacted to anti-His6 and monoclonal anti-CdtA antibodies. ELISA revealed that purified soluble CdtA-CdtC protein bound to GM1 ganglioside, while CdtA alone did not.
CONCLUSIONS
Co-expression of CdtA and CdtC proteins enhanced the solubility of the proteins in E. coli, leading to convenient preparation of active CdtA-CdtC, a critical material for the study of AaCDT pathogenesis.
MeSH Terms
Figure
Reference
-
1. Henderson B, Nair SP, Ward JM, Wilson M. Molecular pathogenicity of the oral opportunistic pathogen Actinobacillus actinomycetemcomitans. Annu Rev Microbiol. 2003. 57:29–55.
Article2. Lara-Tejero M, Galan JE. Cytolethal distending toxin: limited damage as a strategy to modulate cellular functions. Trends Microbiol. 2002. 10:147–152.
Article3. Leung WK, Ngai VKS, Yau JYY, et al. Characterization of Actinobacillus actinomycetemcomitans isolated from young Chinese aggressive periodontitis patients. J Periodontal Res. 2005. 40:258–268.
Article4. Ahmed HJ, Svensson LA, Cope LD, et al. Prevalence of cdtABC genes encoding cytolethal distending toxin among Haemophilus ducreyi and Actinobacillus actinomycetemcomitans strains. J Med Microbiol. 2001. 50:860–864.
Article5. Fabris AS, DiRienzo JM, Wikstrom M, Mayer MP. Detection of cytolethal distending toxin activity and cdt genes in Actinobacillus actinomycetemcomitansisolates from geographically diverse populations. Oral Microbiol Immunol. 2002. 17:231–238.
Article6. Belibasakis G, Johansson A, Wang Y, et al. Inhibited proliferation of human periodontal ligament cells and gingival fibroblasts by Actinobacillus actinomycetemcomitans: involvement of the cytolethal distending toxin. Eur J Oral Sci. 2002. 110:366–373.
Article7. Kanno F, Korostoff J, Volgina A, DiRienzo JM. Resistance of human periodontal ligament fibroblasts to the cytolethal distending toxin of Actinobacillus actinomycetemcomitans. J Periodontol. 2005. 76:1189–1201.
Article8. DiRienzo JM, Song M, Wan LSY, Ellen RP. Kinetics of KB and HEp-2 cell responses to an invasive, cytolethal distending toxin producing strain of Actinobacillus actinomycetemcomitans. Oral Microbiol Immunol. 2002. 17:245–251.
Article9. Belibasakis GN, Johansson A, Wang Y, et al. The cytolethal distending toxin induces receptor activator of NF-kappaB ligand expression in human gingival fibroblasts and periodontal ligament cells. Infect Immun. 2005. 73:342–351.
Article10. Belibasakis GN, Brage M, Lagergård T, Johansson A. Cytolethal distending toxin upregulates RANKL expression in Jurkat T-cells. APMIS. 2008. 116:499–506.
Article11. Mogi M, Otogoto J, Ota N, Togari A. Differential expression of RANKL and osteoprotegerin in gingival crevicular fluid of patients with periodontitis. J Dent Res. 2004. 83:166–169.
Article12. Pickett CL, Cottle DL, Pesci EC, Bikah G. Cloning, sequencing, and expression of the Escherichia coli cytolethal distending toxin genes. Infect Immun. 1994. 62:1046–1051.
Article13. Elwell CA, Dreyfus LA. DNase I homologous residues in CdtB are critical for cytolethal distending toxin-mediated cell cycle arrest. Mol Microbiol. 2000. 37:952–963.
Article14. Nesic D, Hsu Y, Stebbins CE. Assembly and function of a bacterial genotoxin. Nature. 2004. 429:429–433.
Article15. Peres SY, Marches O, Daigle F, et al. A new cytolethal distending toxin (CDT) from Escherichia coli producing CNF2 blocks HeLa cell division in G2/M phase. Mol Microbiol. 1997. 24:1095–1107.16. Lee RB, Hassane DC, Cottle DL, Pickett CL. Interactions of Campylobacter jejuni cytolethal distending toxin subunits CdtA and CdtC with HeLa cells. Infect Immun. 2003. 71:4883–4890.
Article17. McSweeney LA, Dreyfus LA. Carbohydrate-binding specificity of the Escherichia coli cytolethal distending toxin CdtA-II and CdtC-II subunits. Infect Immun. 2005. 73:2051–2060.
Article18. Mise K, Akifusa S, Watarai S, et al. Involvement of ganglioside GM3 in G2/M cell cycle arrest of human monocytic cells induced by Actinobacillus actinomycetemcomitans cytolethal distending toxin. Infect Immun. 2005. 73:4846–4852.
Article19. Yamada T, Komoto J, Saiki K, Konishi K, Takusagawa F. Variation of loop sequence alters stability of cytolethal distending toxin (CDT): crystal structure of CDT from Actinobacillus actinomycetemcomitans. Protein Sci. 2006. 15:362–372.
Article20. Boesze-Battaglia K, Besack D, McKay T, et al. Cholesterol-rich membrane microdomains mediate cell cycle arrest induced by Actinobacillus actinomycetemcomitans cytolethal-distending toxin. Cell Microbiol. 2006. 8:823–836.
Article21. Saiki K, Konishi K, Gomi T, Nishihara T, Yoshikawa M. Reconstitution and purification of cytolethal distending toxin of Actinobacillus actinomycetemcomitans. Microbiol Immunol. 2001. 45:497–506.
Article22. Ko SY, Jeong DK, Ryu SH, Kim HS. Cloning and protein expression of Actinobacillus actinomycetemcomitans cytolethal distending toxin subunit CdtA. J Korean Acad Periodontol. 2007. 37:339–351.
Article23. Lee ES, Park SY, Lee EU, Kim HS. Cloning and protein expression of Aggregatibacter actinomycetemcomitans cytolethal distending toxin C. J Korean Acad Periodontol. 2008. 38:317–324.
Article24. Mao X, DiRienzo JM. Functional studies of the recombinant subunits of a cytolethal distending holotoxin. Cell Microbiol. 2002. 4:245–255.
Article25. Nesic D, Stebbins CE. Mechanisms of assembly and cellular interactions for the bacterial genotoxin CDT. PLoS Pathog. 2005. 1:214–224.
Article26. Ueno Y, Ohara M, Kawamoto T, et al. Biogenesis of the Actinobacillus actinomycetemcomitans cytolethal distending toxin holotoxin. Infect Immun. 2006. 74:3480–3487.
Article27. Lagergard T, Lundqvist A, Wising C, Gabrielsson V, Ahlman K. Formaldehyde treatment increases the immunogenicity and decreases the toxicity of Haemophilus ducreyi cytolethal distending toxin. Vaccine. 2007. 25:3606–3614.
Article28. Mbwana J, Ahmed HJ, Ahlman K, et al. Specificity of antibodies directed against the cytolethal distending toxin of Haemophilus ducreyi in patients with chancroid. Microb Pathog. 2003. 35:133–137.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Cloning and protein expression of Actinobacillus actinomycetemcomitans cytolethal distending toxin subunit CdtA
- Production of the polyclonal subunit C protein antibody against Aggregatibacter actinomycetemcomitans cytolethal distending toxin
- Cloning and protein expression of Aggregatibacter actinomycetemcomitans cytolethal distending toxin C
- Purification and refolding of the recombinant subunit B protein of the Aggregatibacter actinomycetemcomitans cytolethal distending toxin
- Virulence-associated Genome Sequences of Pasteurella canis and Unique Toxin Gene Prevalence of P. canis and Pasteurella multocida Isolated from Humans and Companion Animals