Nat Prod Sci.
2015 Dec;21(4):268-272. 10.20307/nps.2015.21.4.268.
Anti-inflammatory Effect of Dactyloquinone B and Cyclospongiaquinone-1 Mixture in RAW264.7 Macrophage and ICR Mice
- Affiliations
-
- 1Department of Biomedical Chemistry, Konkuk University, Chungju 380-701, Korea.
- 2Department of Chemistry, University of Iowa, Iowa City, IA 52242, United States.
- 3College of Pharmacy, Keimyung University, Daegu 704-701, Korea. gsjeong@kmu.ac.kr
- 4College of Pharmacy, Chungnam National University; Daejeon 305-764, Korea. mkna@cnu.ac.kr
- KMID: 2312908
- DOI: http://doi.org/10.20307/nps.2015.21.4.268
Abstract
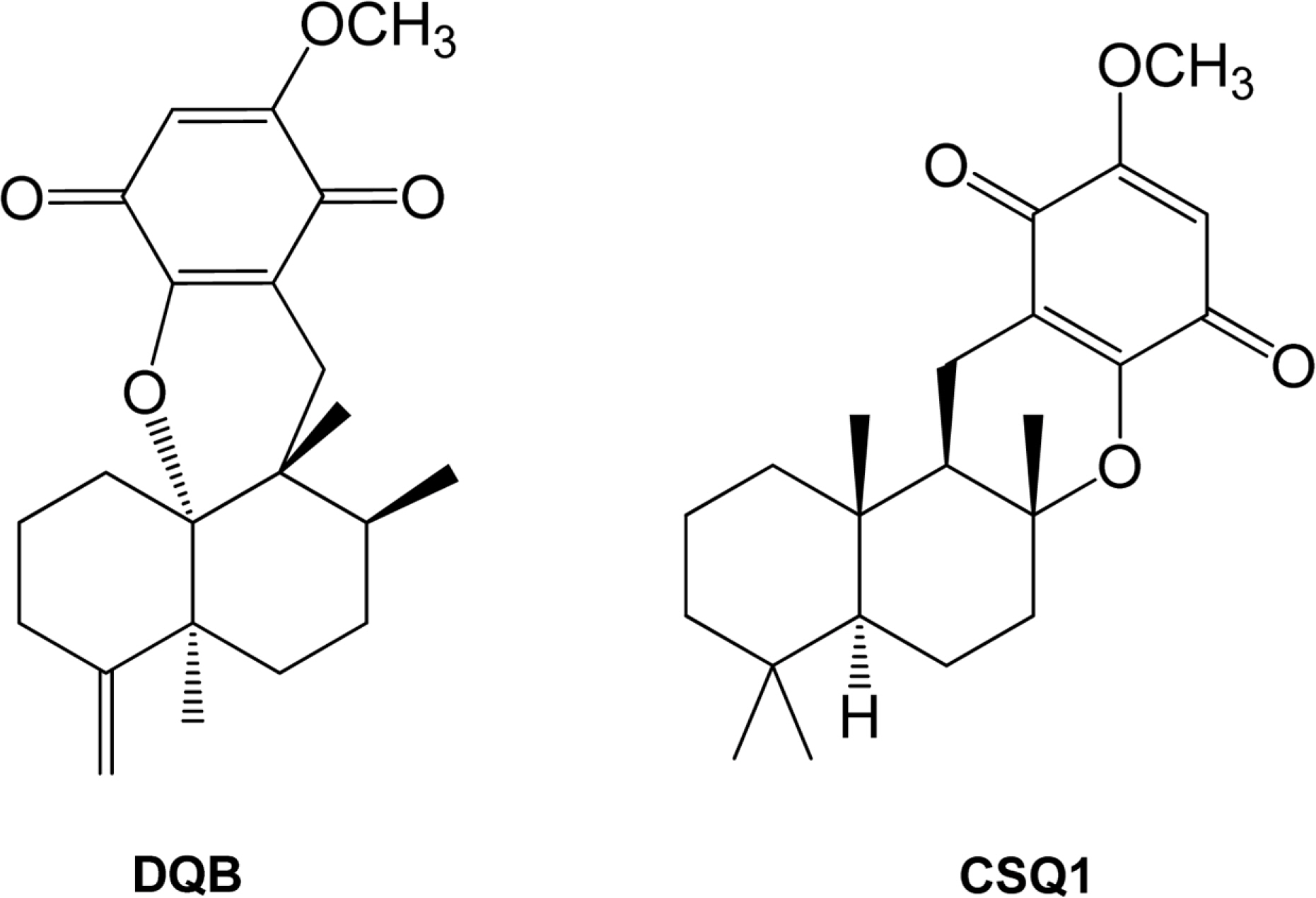
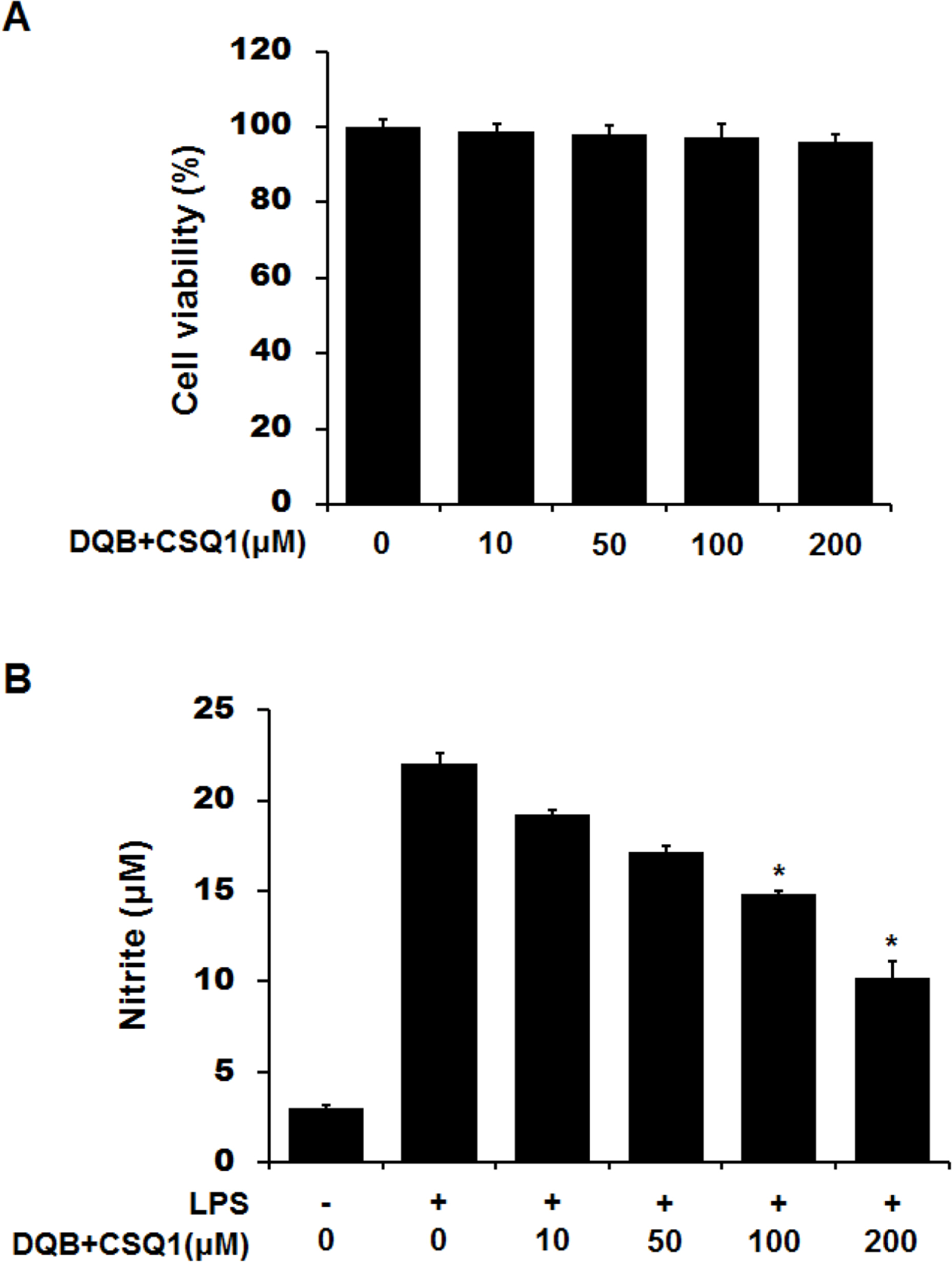
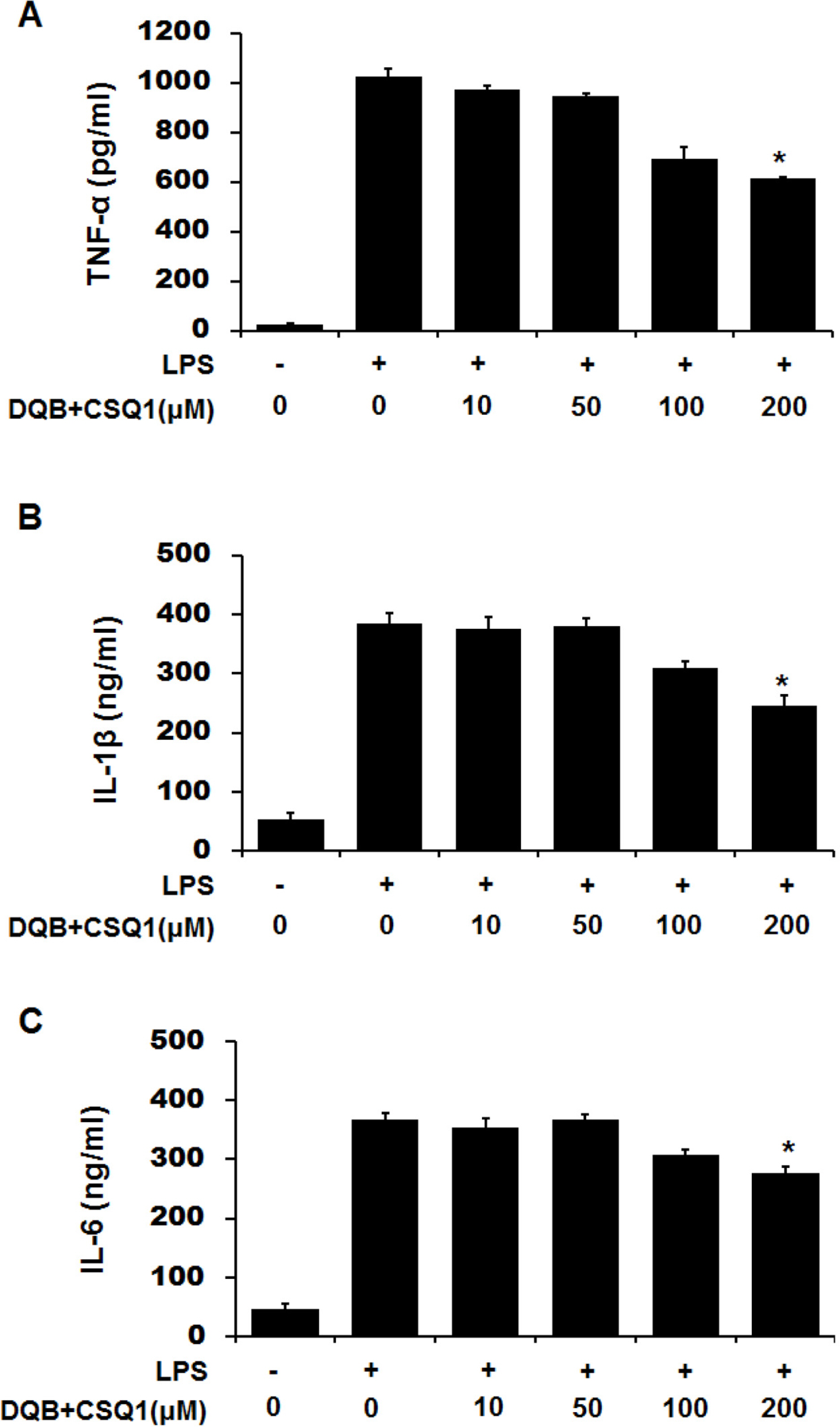
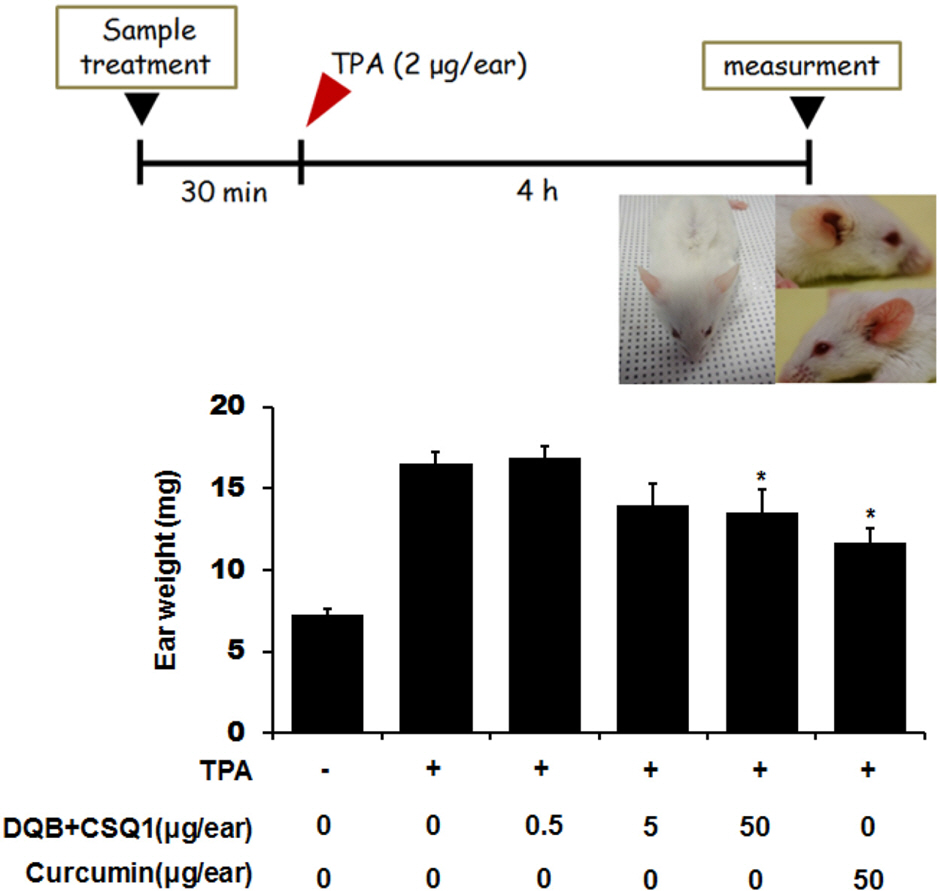
- Sesquiterpene-quinone is a class of secondary metabolites frequently encountered from marine sponge. The present study was designed to examine the anti-inflammatory action of sponge-derived dactyloquinone B (DQB) and cyclospongiaquinone-1 (CSQ1) mixture using lipopolysaccharide (LPS)-induced inflammatory responses. We measured the production of nitric oxide (NO), tumor necrosis factor-alpha (TNF-alpha), interleukin-1beta (IL-1beta), and interleukin-6 (IL-6) and expression of inducible nitric oxide synthase (iNOS) and cyclooxygenase-2 (COX-2) protein. TNF-alpha, IL-1beta, and IL-6 production, which increased by treatment with LPS, were significantly inhibited by DQB and CSQ1 mixture. It also decreased the production of NO production, and iNOS and COX-2 expression. Furthermore, it reduced 12-O-tetradecanoylphorbol 13-acetate (TPA)-induced ear edema of ICR mice. These results demonstrate that sesquiterpene-quinone, DQB and CSQ1 mixture, might serve as a chemical pipeline for the development of anti-inflammatory agent.
MeSH Terms
Figure
Reference
-
(1). Blunt J. W., Copp B. R., Keyzers R. A., Munro M. H., Prinsep M. R.Nat. Prod. Rep. 2014; 31:160–258.(2). Marcos I. S., Conde A., Moro R. F., Basabe P., Diez D., Urones J. G.Mini Rev. Org. Chem. 2010; 7:230–254.(3). Luibrand R. T., Erdman T. R., Vollmer J. J., Scheuer P. J., Finer J., Clardy J.Tetrahedron. 1979; 35:609–612.(4). Capon R. J., MacLeod J. K. J.Org. Chem. 1987; 52:5059–5060.(5). Takizawa P. A., Yucel J. K., Veit B., Faulkner D. J., Deerinck T., Soto G., Ellisman M., Malhotra V.Cell. 1993; 73:1079–1090.(6). Kazlauskas R., Murphy P. T., Warren R. G., Wells R. J., Blount J. F.Aust. J. Chem. 1978; 31:2685–2697.(7). Fernandez A., Alvarez E., Alvarez-Manzaneda R., Chahboun R., Alvarez-Manzaneda E.Chem. Comm. 2014; 50:13100–13102.(8). Mitome H., Nagasawa T., Miyaoka H., Yamada Y., van Soest R. W. J.Nat. Prod. 2001; 64:1506–1508.(9). Mitome H., Nagasawa T., Miyaoka H., Yamada Y., van Soest R. W.Tetrahedron. 2002; 58:1693–1696.(10). Mitome H., Nagasawa T., Miyaoka H., Yamada Y., van Soest R. W. J.Nat. Prod. 2003; 66:46–50.(11). Ovenden S. P., Nielson J. L., Liptrot C. H., Willis R. H., Tapiolas D. M., Wright A. D., Motti C. A. J.Nat. Prod. 2011; 74:65–68.(12). Jankam A., Somerville M. J., Hooper J. N. A., Brecknell D. J., Suksamrarn A., Garson M. J.Tetrahedron. 2007; 63:1577–1582.(13). Lee D. S., Jeong G. S., Li B., Park H., Kim Y. C.Int. Immunopharmacol. 2010; 10:850–858.(14). Lee D. S., Jang J. H., Ko W., Kim K. S., Sohn J. H., Kang M. S., Ahn J. S., Kim Y. C., Oh H.Mar. Drugs. 2013; 11:1409–1426.(15). Hwang I. H., Oh J., Zhou W., Park S., Kim J.-H., Chittiboyina A. G., Ferreira D., Song G. Y., Oh S., Na M., Hamann M. T. J.Nat. Prod. 2015; 78:453–461.(16). Agut J., Tarrida N., Sacristan A., Ortiz J. A.Methods Find. Exp. Clin. Pharmacol. 1996; 18:233–234.(17). Liebel F., Lyte P., Garay M., Babad J., Southall M. D.Arch. Dermatol. Res. 2006; 298:191–199.(18). Rao T. S., Currie J. L., Shaffer A. F., Isakson P. C.Inflammation. 1993; 17:723–741.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Inhibitory Effects of Paeonia Suffruticosa on the Expression of Inducible Nitric Oxide Synthase Gene in RAW264.7 Macrophages
- Quercetin-3-O-β-D-Glucuronide Suppresses Lipopolysaccharide-Induced JNK and ERK Phosphorylation in LPS-Challenged RAW264.7 Cells
- Anti-inflammatory and Anti-pruritic Effects of Portulaca oleracea L. Extract Using In Vitro and In Vivo Inflammation Model: LPS-treated Raw264.7 Cells, Keratinocytes, NC/Nga Mice and Hairless SKH-1 Mice
- Meconopsis quintuplinervia Regel Improves Cutibacterium acnes-Induced Inflammatory Responses in a Mouse Ear Edema Model and Suppresses Pro-Inflammatory Chemokine Production via the MAPK and NF-κB Pathways in RAW264.7 Cells
- Anti-inflammatory and immune enhancing activities of PB203 in mouse macrophage RAW264.7 cells