Lab Anim Res.
2010 Jun;26(2):139-144. 10.5625/lar.2010.26.2.139.
Fractionation of DNases Specific to Haemonchus contortus Intestine by Phenyl Sepharose Column
- Affiliations
-
- 1College of Veterinary Medicine, Kyungpook National University, Daegu, Korea. dmkwak@knu.ac.kr
- KMID: 2312059
- DOI: http://doi.org/10.5625/lar.2010.26.2.139
Abstract
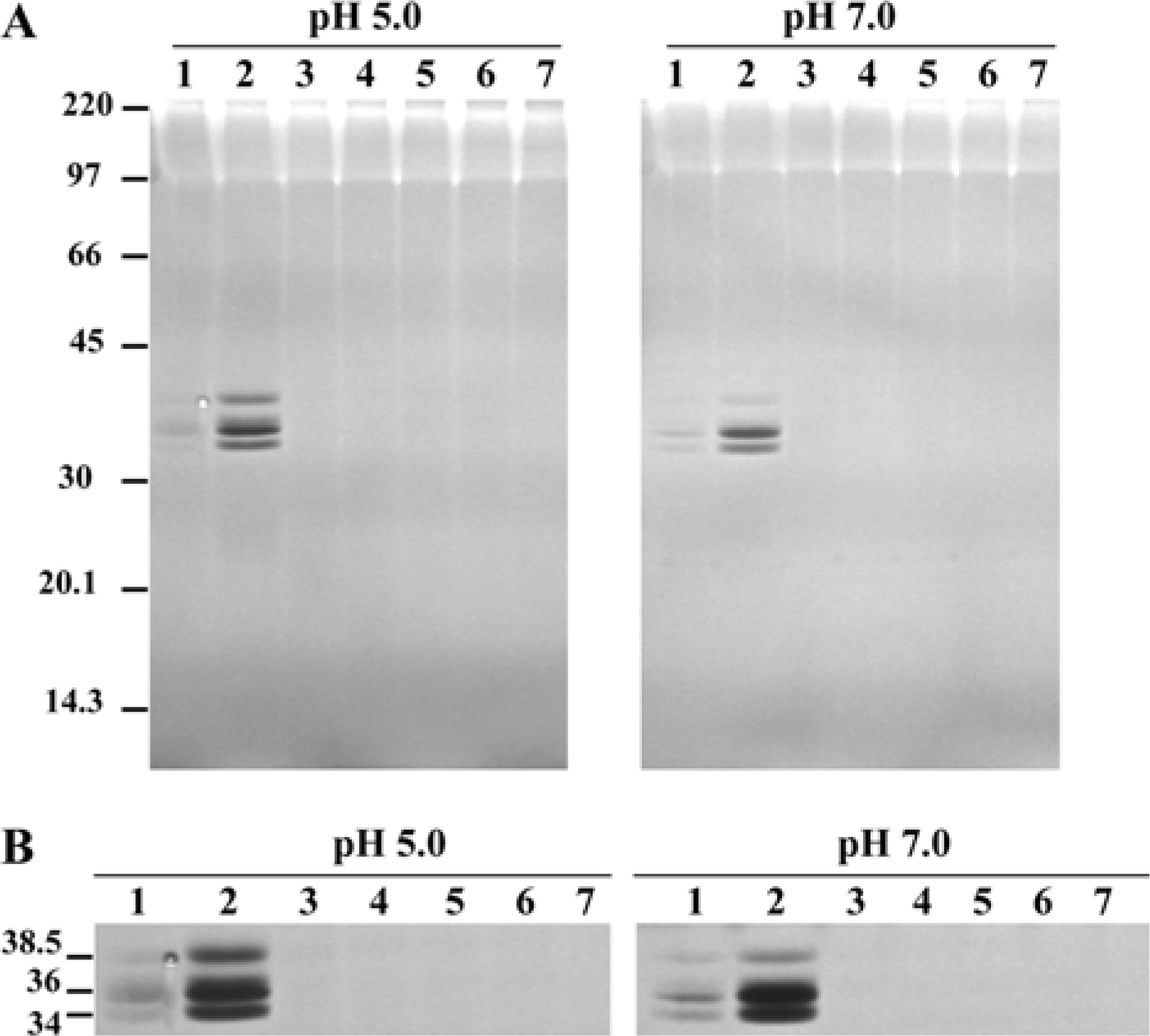
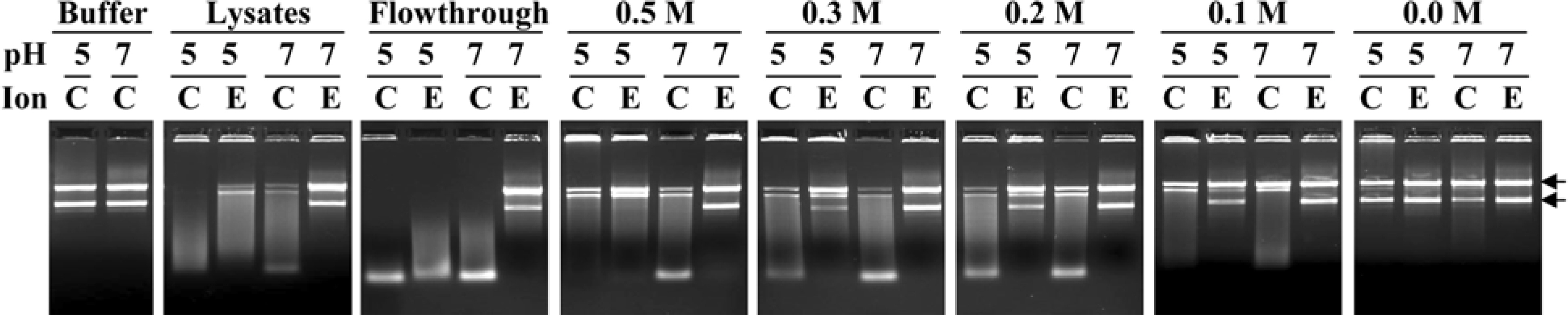
- Multiple DNases were identified from Haemonchus contortus intestine based on previous studies. The DNases detected at 34, 36 and 38.5 kDa had diverse characteristics. Some of them had characteristics similar to those of mammalians and others had unusual characteristics. This study was carried out to fractionate worm intestinal DNases from other proteins using phenyl Sepharose chromatographic methods. All DNases detected from Haemonchus contortus intestine were fractionated in the flowthrough of phenyl Sepharose, indicating the worm DNases are hydrophilic. The DNases were enriched five-fold in the flowthrough fraction while additional steps are required for isolation of the worm DNases. Thus, fractionation with phenyl Sepharose could be used as a good initial step to enrich and separate DNases from other proteins.
MeSH Terms
Figure
Reference
-
Barry M.A.., Eastman A.1993. Identification of deoxyribonuclease II as an endonuclease involved in apoptosis. Arch. Biochem. Biophys. 300(1):440–450.
ArticleColes G.C.., Jackson F.., Pomroy W.E.., Prichard R.K.., von Samson-Himmelstjerna G.., Silvestre A.., Taylor M.A.., Vercruysse J.2006. ).The detection of anthelmintic resistance in nematodes of veterinary importance. Vet. Parasitol. 136(3-4):167–185.
ArticleEnari M.., Sakahira H.., Yokoyama H.., Okawa K.., Iwamatsu A.., Nagata S.1998. A caspase-activated DNase that degrades DNA during apoptosis, and its inhibitor ICAD. Nature. 391(6662):43–50.
ArticleHedgecock E.M.., Sulston J.E.., Thomson J.N.1983. Mutations affecting programmed cell deaths in the nematode Caenorhabditis elegans. Science. 220(4603):1277–1279.Jasmer D.P.., McGuire T.C.1991. Protective immunity to a blood-feeding nematode (Haemonchus contortus) induced by parasite gut antigens. Infect. Immun. 59(12):4412–4417.
ArticleJasmer D.P.., Yao C.., Rehman A.., Johnson S.2000. Multiple lethal effects induced by a benzimidazole anthelmintic in the anterior intestine of the nematode Haemonchus contortus. Mol. Biochem. Parasitol. 105(1):81–90.
ArticleKaranu F.N.., Rurangirwa F.R.., McGuire T.C.., Jasmer D.P.1993. Haemonchus contortus: identification of proteases with diverse characteristics in adult worm excretory-secretory products. Exp. Parasitol. 77(3):362–371.
ArticleKwak D.., Jasmer D.P.2003. ).Non-classic characteristics define prominent DNase activities from the intestine and other tissues of Haemonchus contortus. Exp. Parasitol. 104(3-4):131–139.
ArticleKwak D.., Jasmer D.P.2004. ).Intestinal DNases of 36 and 38.5kDa from the parasitic nematode Haemonchus contortus have non-classic DNase characteristics and produce DNA fragments with 3'-hydroxyls. Exp. Parasitol. 108(3-4):142–153.
ArticleLacey E.1988. The role of the cytoskeletal protein, tubulin, in the mode of action and mechanism of drug resistance to benzimidazoles. Int. J. Parasitol. 18(7):885–936.
ArticleLubega G.W.., Prichard R.K.1990. Specific interaction of benzimidazole anthelmintics with tubulin: high-affinity binding and benzimidazole resistance in Haemonchus contortus. Mol. Biochem. Parasitol. 38(2):221–232.
ArticleMak C.H.., Ko R.C.1999. Characterization of endonuclease activity from excretory/secretory products of a parasitic nematode, Trichinella spiralis. Eur. J. Biochem. 260(2):477–481.
ArticleParrish J.., Li L.., Klotz K.., Ledwich D.., Wang X.., Xue D.2001. Mitochondrial endonuclease G is important for apoptosis in C. elegans. Nature. 412(6842):90–94.Parrish J.Z.., Xue D.2003. Functional genomic analysis of apoptotic DNA degradation in C. elegans. Mol. Cell. 11(4):987–996.Ribeiro J.M.., Carson D.A.1993. Ca2+/Mg(2+)-dependent endonuclease from human spleen: purification, properties, and role in apoptosis. Biochemistry. 32(35):9129–9136.Rodriguez A.M.., Rodin D.., Nomura H.., Morton C.C.., Weremowicz S.., Schneider M.C.1997. Identification, localization, and expression of two novel human genes similar to deoxyribonuclease I. Genomics. 42(3):507–513.
ArticleSaraste A.., Pulkki K.2000. Morphologic and biochemical hallmarks of apoptosis. Cardiovasc. Res. 45(3):528–537.
ArticleShiokawa D.., Tanuma S.1998. ).Molecular cloning and expression of a cDNA encoding an apoptotic endonuclease DNase gamma. Biochem. J. 332(Pt 3):713–720.Shiokawa D.., Tanuma S.1999. DLAD, a novel mammalian divalent cation-independent endonuclease with homology to DNase II. Nucleic Acids Res. 27(20):4083–4089.
ArticleShiokawa D.., Tanuma S.2001. Characterization of human DNase I family endonucleases and activation of DNase gamma during apoptosis. Biochemistry. 40(1):143–152.Wu Y.C.., Stanfield G.M.., Horvitz H.R.2000. NUC-1, a Caenorhabditis elegans DNase II homolog, functions in an intermediate step of DNA degradation during apoptosis. Genes Dev. 14(5):536–548.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Cloning and characterization of a selenium-independent glutathione peroxidase (HC29) from adult Haemonchus contortus
- Characterization of HC58cDNA, a putative cysteine protease from the parasite Haemonchus contortus
- Development of a Lateral Flow Strip-Based Recombinase Polymerase Amplification Assay for the Detection of Haemonchus contortus in Goat Feces
- Erratum: Characterization of HC58cDNA, a putative cysteine protease from the parasite Haemonchus contortus
- A variant of ornithine aminotransferase from mouse small intestine