Korean J Urol.
2006 May;47(5):548-552. 10.4111/kju.2006.47.5.548.
Alterations in Calcium-Activated Potassium Channel Expressions in Human Prostate Cancer
- Affiliations
-
- 1Department of Urology, Catholic University of Daegu School of Medicine, Korea. dykim@mail.cu.ac.kr
- 2Department of Physiology, Kyungpook National University School of Medicine, Daegu, Korea.
- KMID: 2294143
- DOI: http://doi.org/10.4111/kju.2006.47.5.548
Abstract
- PURPOSE
Recent studies have shown that potassium (K+) and sodium channels are involved in prostate cell growth. However, a great many of the studies have been done in prostate cancer cell lines and there are only scant studies on prostate cancer and benign prostatic hypertrophy (BPH) tissue. The present study was aimed to evaluate the alterations of the calcium-activated K+ channel (KCa) expression in prostate cancer, and to compare them with the expression profiles in human BPH tissue to understand their potential role in the progression of prostate cancer.
MATERIALS AND METHODS
The prostate tissues obtained from radical prostatectomy (n=10) and transurethral resection of the prostate (n=18) were quickly frozen in liquid nitrogen for the RNA measurements. The protein and mRNA levels of the KCa subtypes and connexins were measured by performing immunoblot analysis and reverse-transcription polymerase chain reaction, respectively.
RESULTS
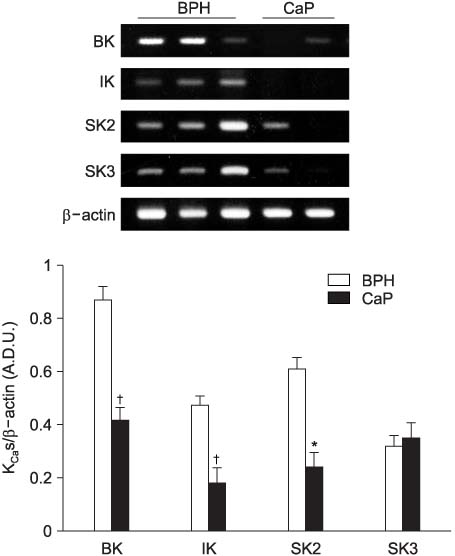
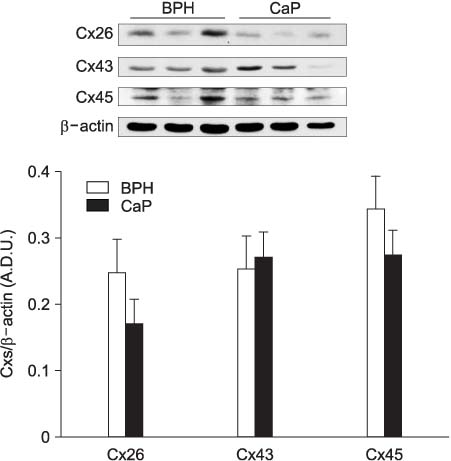
The mRNA levels of type 2 (SK2) and type 3 (SK3) small-conductance and large-conductance (BK) KCas in the prostate cancer tissues were decreased more than 50% compared with those in the BPH samples. In addition, the BK and SK2 protein levels in prostate cancer were also significantly lower than those in the BPH. As reported previously, the connexin 26 and 43 transcript signals in the prostate cancer were significantly reduced compared with those in the BPH samples.
CONCLUSIONS
These results suggest that the impaired expression of KCas may have a role in tumor progression via aberrant and uncontrolled prostate cell growth.
Keyword
MeSH Terms
-
Cell Line
Connexins
Humans*
Large-Conductance Calcium-Activated Potassium Channels
Nitrogen
Polymerase Chain Reaction
Potassium
Potassium Channels, Calcium-Activated*
Prostate*
Prostatectomy
Prostatic Hyperplasia
Prostatic Neoplasms*
RNA
RNA, Messenger
Small-Conductance Calcium-Activated Potassium Channels
Sodium Channels
Connexins
Large-Conductance Calcium-Activated Potassium Channels
Nitrogen
Potassium
Potassium Channels, Calcium-Activated
RNA
RNA, Messenger
Small-Conductance Calcium-Activated Potassium Channels
Sodium Channels
Figure
Reference
-
1. Gleave M, Miyake H, Chi K. Beyond simple castration: targeting the molecular basis of treatment resistance in advanced prostate cancer. Cancer Chemother Pharmacol. 2005. 56:Suppl 7. 47–57.2. Pienta KJ, Smith DC. Advances in prostate cancer chemotherapy: a new era begins. CA Cancer J Clin. 2005. 55:300–318.3. Abdul M, Hoosein N. Expression and activity of potassium ion channels in human prostate cancer. Cancer Lett. 2002. 186:99–105.4. Laniado ME, Fraser SP, Djamgoz MB. Voltage-gated K(+) channel activity in human prostate cancer cell lines of markedly different metastatic potential: distinguishing characteristics of PC-3 and LNCaP cells. Prostate. 2001. 46:262–274.5. Rybalchenko V, Prevarskaya N, Van Coppenolle F, Legrand G, Lemonnier L, Le Bourhis X, et al. Verapamil inhibits proliferation of LNCaP human prostate cancer cells influencing K+ channel gating. Mol Pharmacol. 2001. 59:1376–1387.6. Skryma RN, Prevarskaya NB, Dufy-Barbe L, Odessa MF, Audin J, Dufy B. Potassium conductance in the androgen-sensitive prostate cancer cell line LNCaP: involvement in cell proliferation. Prostate. 1997. 33:112–122.7. Diss JK, Archer SN, Hirano J, Fraser SP, Djamgoz MB. Expression profiles of voltage-gated Na(+) channel alpha-subunit genes in rat and human prostate cancer cell lines. Prostate. 2001. 48:165–178.8. Fraser SP, Grimes JA, Djamgoz MB. Effects of voltage-gated ion channel modulators on rat prostatic cancer cell proliferation: comparison of strongly and weakly metastatic cell lines. Prostate. 2000. 44:61–76.9. Grimes JA, Djamgoz MB. Electrophysiological characterization of voltage-gated Na+ current expressed in the highly metastatic Mat-LyLu cell line of rat prostate cancer. J Cell Physiol. 1998. 175:50–58.10. Gopalakrishnan M, Shieh CC. Potassium channel subtypes as molecular targets for overactive bladder and other urological disorders. Expert Opin Ther Targets. 2004. 8:437–458.11. Abdul M, Santo A, Hoosein N. Activity of potassium channel-blockers in breast cancer. Anticancer Res. 2003. 23:3347–3351.12. Parihar AS, Coghlan MJ, Gopalakrishnan M, Shieh CC. Effects of intermediate-conductance Ca2+-activated K+ channel modulators on human prostate cancer cell proliferation. Eur J Pharmacol. 2003. 471:157–164.13. Rane SG. The growth regulatory fibroblast IK channel is the prominent electrophysiological feature of rat prostatic cancer cells. Biochem Biophys Res Commun. 2000. 269:457–463.14. Ashcroft FM. Ion channels and disease. 2000. San Diego: Academic Press;125–160.15. Day ML, Pickering SJ, Johnson MH, Cook DI. Cell-cycle control of a large-conductance K+ channel in mouse early embryos. Nature. 1993. 365:560–562.16. Gee J, Tanaka M, Grossman HB. Connexin 26 is abnormally expressed in bladder cancer. J Urol. 2003. 169:1135–1137.17. Carruba G, Stefano R, Cocciadiferro L, Saladino F, Di Cristina A, Tokar E, et al. Intercellular communication and human prostate carcinogenesis. Ann N Y Acad Sci. 2002. 963:156–168.18. Habermann H, Ray V, Habermann W, Prins GS. Alterations in gap junction protein expression in human benign prostatic hyperplasia and prostate cancer. J Urol. 2002. 167:655–660.19. Grossman HB, Liebert M, Lee IW, Lee SW. Decreased connexin expression and intercellular communication in human bladder cancer cells. Cancer Res. 1994. 54:3062–3065.20. van Baren MJ, Heutink P. The PCR suite. Bioinformatics. 2004. 20:591–593.21. Tsai H, Werber J, Davia MO, Edelman M, Tanaka KE, Melman A, et al. Reduced connexin 43 expression in high grade, human prostatic adenocarcinoma cells. Biochem Biophys Res Commun. 1996. 227:64–69.22. Mehta PP, Lokeshwar BL, Schiller PC, Bendix MV, Ostenson RC, Howard GA, et al. Gap-junctional communication in normal and neoplastic prostate epithelial cells and its regulation by cAMP. Mol Carcinog. 1996. 15:18–32.23. Hossain MZ, Jagdale AB, Ao P, LeCiel C, Huang RP, Boynton AL. Impaired expression and posttranslational processing of connexin43 and downregulation of gap junctional communication in neoplastic human prostate cells. Prostate. 1999. 38:55–59.24. Govindarajan R, Zhao S, Song XH, Guo RJ, Wheelock M, Johnson KR, et al. Impaired trafficking of connexins in androgen-independent human prostate cancer cell lines and its mitigation by alpha-catenin. J Biol Chem. 2002. 277:50087–50097.25. Tanaka M, Grossman HB. Connexin 26 induces growth suppression, apoptosis and increased efficacy of doxorubicin in prostate cancer cells. Oncol Rep. 2004. 11:537–541.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Effects of Phytoestrogen on Potassium Channel Activities of Smooth Muscle Cells of Rabbit Seminal Vesicle
- Effects of Fluoxetine on Potassium Channels in Cat Gastric Smooth Muscle Cells
- 17Beta-Estradiol Inhibits Calcium-Activated Potassium Channel Expressions in Rat Whole Bladder
- Characteristics of Potassium Channel in the Isolated Rat Detrusor Muscle
- Mechanical Hyperalgesia Induced by Blocking Calcium-activated Potassium Channels on Capsaicin-sensitive Afferent Fiber