Korean J Physiol Pharmacol.
2009 Dec;13(6):455-460. 10.4196/kjpp.2009.13.6.455.
Type II and III Taste Bud Cells Preferentially Expressed Kainate Glutamate Receptors in Rats
- Affiliations
-
- 1Department of Physiology and Neuroscience, College of Dentistry, Gangneung-Wonju National University, Gangneung 210-702, Korea. knkim@gwnu.ac.kr
- 2Department of Pharmacology and Mechanism, Research Institute, Oscotec Inc., Cheonan 331-831, Korea.
- 3Research Institute of Oral Science, Gangneung-Wonju National University, Gangneung 210-702, Korea.
- KMID: 2285381
- DOI: http://doi.org/10.4196/kjpp.2009.13.6.455
Abstract
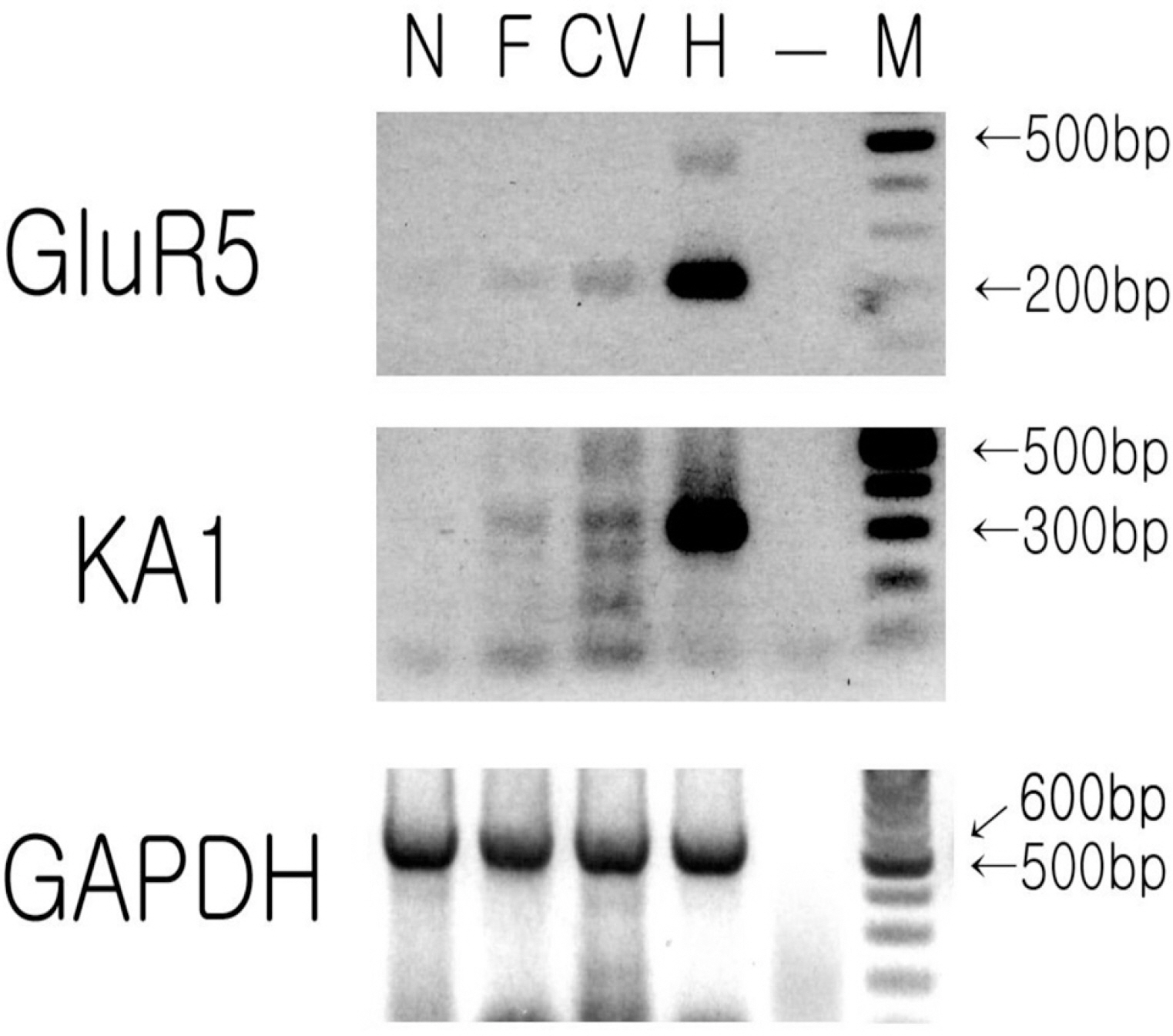
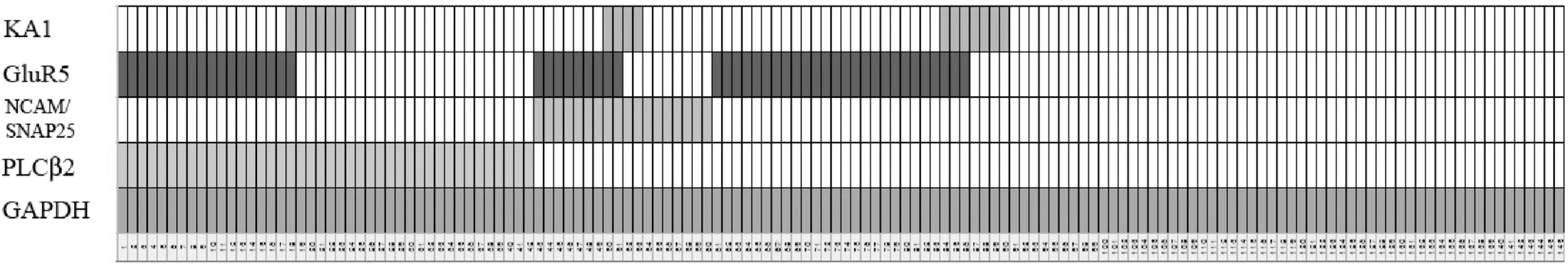
- Glutamate-induced cobalt uptake reveals that non-NMDA glutamate receptors (GluRs) are present in rat taste bud cells. Previous studies involving glutamate induced cobalt staining suggest this uptake mainly occurs via kainate type GluRs. It is not known which of the 4 types of taste bud cells express subunits of kainate GluR. Circumvallate and foliate papillae of Sprague-Dawley rats (45~60 days old) were used to search for the mRNAs of subunits of non-NMDA GluRs using RT-PCR with specific primers for GluR1-7, KA1 and KA2. We also performed RT-PCR for GluR5, KA1, PLCbeta2, and NCAM/SNAP 25 in isolated single cells from taste buds. Taste epithelium, including circumvallate or foliate papilla, express mRNAs of GluR5 and KA1. However, non-taste tongue epithelium expresses no subunits of non-NMDA GluRs. Isolated single cell RT-PCR reveals that the mRNAs of GluR5 and KA1 are preferentially expressed in Type II and Type III cells over Type I cells.
Keyword
MeSH Terms
Figure
Reference
-
References
Bigiani A. Mouse taste cells with glialike membrane properties. J Neurophysiol. 85:1552–1560. 2001.
ArticleBoughter Jr JD., Pumplin DW., Yu C., Christy RC., Smith DV. Differential expression of alpha-gustducin in taste bud populations of the rat and hamster. J Neurosci. 17:2852–2858. 1997.Bowie D. External anions and cations distinguish between AMPA and kainate receptor gating mechanisms. J Physiol (London). 539:725–733. 2002.
ArticleBradlley RM., King MS., Wang L., Shu X. Neurotransmitter and neuromodulator activity in the gustatory zone of the nucleus tractus solitarius. Chem Sens. 21:377–385. 1996.Caicedo A., Kim KN., Roper SD. Glutamate-induced cobalt uptake reveals non-NMDA receptors in rat taste cells. J Comp Neurol. 417:315–324. 2000.
ArticleCastillo PE., Malenka RC., Nicoll RA. Kainate receptors mediate a slow postsynaptic current in hippocampal CA3 neurons. Nature. 388:182–186. 1997.
ArticleChittajallu R., Vignes M., Dev KK., Barnes JM., Collingridge GL., Henley JM. Regulation of glutamate release by presynaptic kainate receptors in the hippocampus. Nature. 79:78–816. 1993.
ArticleChung KM., Lee SB., Heur R., Cho YK., Lee CH., Jung HY., Chung SH., Lee SP., Kim KN. Glutamate-induced cobalt uptake elicited by kainate receptors in rat taste bud cells. Chem Senses. 30:137–143. 2005.
ArticleDeFazio RA., Dvoryanchikov G., Maruyama Y., Kim JW., Pereira E., Roper SD., Chaudhari N. Separate populations of receptor cells and presynaptic cells in mouse taste buds. J Neurosci. 26:3971–3980. 2006.
ArticleDingledine R., Conn PJ. Peripheral Glutamate Receptors: molecular biology and role in taste sensation. J Nutr. 130:1039S–1042S. 2000.
ArticleFarbmann AI. Fine structure of taste bud. J Ultrastruct Res. 12:328–350. 1965.Finger TE., Danilova V., Barrows J., Bartel DL., Vigers AJ., Stone L., Hellekant G., Kinnamon SC. ATP signaling is crucial for communication from taste buds to gustatory nerves. Science. 310:1495–1499. 2005.
ArticleHerness MS., Chen Y. Serotonin inhibits calcium-activated K+ current in rat taste receptor cells. NeuroReport. 8:3527–3261. 1997.Herness MS., Zhao FL., Lu SG., Kaya N., Shen T., Sun XD. Adrenergic signaling between rat taste recepter cells. J Physiol (Lond). 543:601–614. 2002.Huang YJ., Maruyama Y., Lu KS., Pereira E., Plonsky I., Baur JE., Wu D., Roper SD. Mouse taste buds use serotonin as a neurotransmitter. J Neurosci. 25:843–847. 2005.
ArticleJain S., Roper SD. Immunocytochemistry of GABA, glutamate, serotonin, and histamine in Necturus taste buds. J Comp Neurol. 307:675–682. 1991.Kim KN., Caicedo A., Roper SD. Glutamate-induced cobalt uptake reveals non-NMDA receptors in developing rat taste buds. NeuroReport. 12:1715–1718. 2001.
ArticleLee SB., Lee CH., Cho YK., Chung KM., Kim KN. Expression of kainate glutamate receptors in type II cells in taste buds of rats. Int J Oral Biol. 33:83–89. 2008.Lerma J. Roles, and rules of kainate receptors in synaptic transmission. Nat Rev Neurosci. 4:481–495. 2003.
ArticleLindemann B. Taste reception. Physiol Rev. 76:719–766. 1996.
ArticleMadden DR. The structure and function of glutamate receptor ion channels. Nat Rev Neurosci. 3:91–101. 2002.
ArticleMayer ML. Crystal structure of the GluR5 and GluR6 ligand binding cores: Molecular mechanisms underlying kainate receptor selectivity. Neuron. 45:539–552. 2005.Mezei LM., Store DR. Purification of PCR products. Griffin HG, Griffin AM, editors. ed,. PCR technology: Current innovations. CRC Press;Boca Raton: p. p. 13–19. 1994.Murray RG. Mammalian taste bud type III cell: a critical analysis. J Ultrastruct Mol Struct Res. 95:175–188. 1986.Murray RG. The ultrastructure of taste buds. Friedmann I, editor. ed,. The ultrastructure of taste organs. Amsterdam, North Holland: p. p. 1–81. 1974.Nagai T., Delay RJ., Welton J., Roper SD. Uptake and release of neurotransmitter candidates, [3H]serotonin, [3H]glutamate, and [3H]GABA, in taste buds of the mudpuppy. Nectrus maculosus. J Comp Neurol. 392:199–208. 1998.Passafaro M., Nakagawa T., Sala C., Sheng M. GABAnergic neurotransmission in rat taste buds: immunocytochemical study for GABA and GABA transporter subtypes. Nature. 424:677–681. 2003.Pumplin DW., Yu C., Smith DV. Light and dark cells of rat vallate taste buds are morphologically distinct cell types. J Comp Neurol. 378:380–410. 1997.
ArticleRaphael Y., Altschuler RA. Structure and innervation of the cochlea. Brain Res Bull. 60:397–422. 2003.
ArticleRoper SD. Cell communication in taste buds. Cell Mol Life Sci. 63:1494–1500. 2006.Song I., Huganir RL. Regulation of AMPA receptors during synaptic plasticity. Trends Neurosci. 25:578–588. 2002.
ArticleYang R., Crowley HH., Rock ME., Kinnamon JC. Taste cells with synapses in rat circumvallate papillae display SNAP-25-like immunoreactivity. J Comp Neurol. 424:205–215. 2000.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Expression of Neurotrophic Factors and Their Receptors in Rat Posterior Taste Bud Cells
- Immunohistochemical study on the dopaminergic and norepinephrinergic taste cells in rat taste buds
- Immuno-electronmicroscopic study on the serotoninergic taste cells and calcitonin gene-related peptide nerve fibers in mouse taste buds
- Altered Expression of Metabotropic Glutamate Receptors mGluR1, mGluR5, and mGluR2/3 in the Diabetic Rat Retina
- Expression of Glutamate Receptors in Experimental Rat Model of Chronic Glaucoma