Diabetes Metab J.
2011 Apr;35(2):101-106. 10.4093/dmj.2011.35.2.101.
The Causes and Consequences of Low Levels of High Density Lipoproteins in Patients with Diabetes
- Affiliations
-
- 1The Heart Research Institute, Sydney, Australia. barterp@hri.org.au
- KMID: 2281707
- DOI: http://doi.org/10.4093/dmj.2011.35.2.101
Abstract
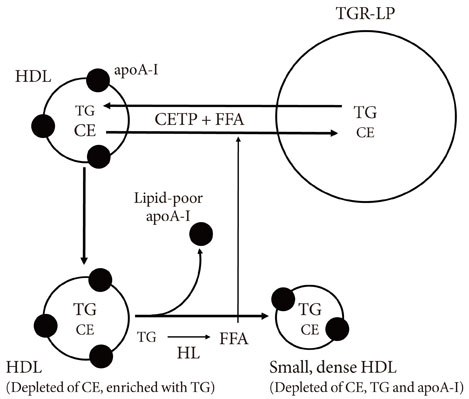
- Type 2 diabetes is commonly accompanied by a low level of high density lipoprotein cholesterol (HDL-C) that contributes to the increased cardiovascular risk associated with this condition. Given that HDLs have the ability to improve increase the uptake of glucose by skeletal muscle and to stimulate the secretion of insulin from pancreatic beta cells the possibility arises that a low HDL concentration in type 2 diabetes may also contribute to a worsening of diabetic control. Thus, there is a double case for raising the level of HDL-C in patients with type 2 diabetes: to reduce cardiovascular risk and to improve glycemic control. Approaches to raising HDL-C include lifestyle factors such as weight reduction, increased physical activity and stopping smoking. Of currently available drugs, the most effective is niacin. Newer formulations of niacin are reasonably well tolerated and have the ability to increase HDL-C by up to 30%. The effect of niacin on cardiovascular events in type 2 diabetes is currently being tested in a large-scale clinical outcome trial.
Keyword
MeSH Terms
Figure
Cited by 1 articles
-
Higher High Density Lipoprotein 2 (HDL2) to Total HDL Cholesterol Ratio Is Associated with a Lower Risk for Incident Hypertension
You-Cheol Hwang, Wilfred Y. Fujimoto, Steven E. Kahn, Donna L. Leonetti, Edward J. Boyko
Diabetes Metab J. 2019;43(1):114-122. doi: 10.4093/dmj.2018.0053.
Reference
-
1. Drew BG, Duffy SJ, Formosa MF, Natoli AK, Henstridge DC, Penfold SA, Thomas WG, Mukhamedova N, de Courten B, Forbes JM, Yap FY, Kaye DM, van Hall G, Febbraio MA, Kemp BE, Sviridov D, Steinberg GR, Kingwell BA. High-density lipoprotein modulates glucose metabolism in patients with type 2 diabetes mellitus. Circulation. 2009. 119:2103–2111.2. Fryirs MA, Barter PJ, Appavoo M, Tuch BE, Tabet F, Heather AK, Rye KA. Effects of high-density lipoproteins on pancreatic beta-cell insulin secretion. Arterioscler Thromb Vasc Biol. 2010. 30:1642–1648.3. Mooradian AD. Dyslipidemia in type 2 diabetes mellitus. Nat Clin Pract Endocrinol Metab. 2009. 5:150–159.4. Barter PJ. Hugh sinclair lecture: the regulation and remodelling of HDL by plasma factors. Atheroscler Suppl. 2002. 3:39–47.5. Barter PJ, Nicholls S, Rye KA, Anantharamaiah GM, Navab M, Fogelman AM. Antiinflammatory properties of HDL. Circ Res. 2004. 95:764–772.6. Hoang A, Murphy AJ, Coughlan MT, Thomas MC, Forbes JM, O'Brien R, Cooper ME, Chin-Dusting JP, Sviridov D. Advanced glycation of apolipoprotein A-I impairs its anti-atherogenic properties. Diabetologia. 2007. 50:1770–1779.7. Nobecourt E, Tabet F, Lambert G, Puranik R, Bao S, Yan L, Davies MJ, Brown BE, Jenkins AJ, Dusting GJ, Bonnet DJ, Curtiss LK, Barter PJ, Rye KA. Nonenzymatic glycation impairs the antiinflammatory properties of apolipoprotein A-I. Arterioscler Thromb Vasc Biol. 2010. 30:766–772.8. Sorrentino SA, Besler C, Rohrer L, Meyer M, Heinrich K, Bahlmann FH, Mueller M, Horvath T, Doerries C, Heinemann M, Flemmer S, Markowski A, Manes C, Bahr MJ, Haller H, von Eckardstein A, Drexler H, Landmesser U. Endothelial-vasoprotective effects of high-density lipoprotein are impaired in patients with type 2 diabetes mellitus but are improved after extended-release niacin therapy. Circulation. 2010. 121:110–122.9. Kruit JK, Kremer PH, Dai L, Tang R, Ruddle P, de Haan W, Brunham LR, Verchere CB, Hayden MR. Cholesterol efflux via ATP-binding cassette transporter A1 (ABCA1) and cholesterol uptake via the LDL receptor influences cholesterol-induced impairment of beta cell function in mice. Diabetologia. 2010. 53:1110–1119.10. Kruit JK, Brunham LR, Verchere CB, Hayden MR. HDL and LDL cholesterol significantly influence beta-cell function in type 2 diabetes mellitus. Curr Opin Lipidol. 2010. 21:178–185.11. Kodama S, Tanaka S, Saito K, Shu M, Sone Y, Onitake F, Suzuki E, Shimano H, Yamamoto S, Kondo K, Ohashi Y, Yamada N, Sone H. Effect of aerobic exercise training on serum levels of high-density lipoprotein cholesterol: a meta-analysis. Arch Intern Med. 2007. 167:999–1008.12. Keech A, Simes RJ, Barter P, Best J, Scott R, Taskinen MR, Forder P, Pillai A, Davis T, Glasziou P, Drury P, Kesaniemi YA, Sullivan D, Hunt D, Colman P, d'Emden M, Whiting M, Ehnholm C, Laakso M. FIELD study investigators. Effects of long-term fenofibrate therapy on cardiovascular events in 9795 people with type 2 diabetes mellitus (the FIELD study): randomised controlled trial. Lancet. 2005. 366:1849–1861.13. ACCORD Study Group. Ginsberg HN, Elam MB, Lovato LC, Crouse JR 3rd, Leiter LA, Linz P, Friedewald WT, Buse JB, Gerstein HC, Probstfield J, Grimm RH, Ismail-Beigi F, Bigger JT, Goff DC Jr, Cushman WC, Simons-Morton DG, Byington RP. Effects of combination lipid therapy in type 2 diabetes mellitus. N Engl J Med. 2010. 362:1563–1574.14. Barter PJ, Rye KA. Is there a role for fibrates in the management of dyslipidemia in the metabolic syndrome? Arterioscler Thromb Vasc Biol. 2008. 28:39–46.15. Barter PJ, Brandrup-Wognsen G, Palmer MK, Nicholls SJ. Effect of statins on HDL-C: a complex process unrelated to changes in LDL-C: analysis of the VOYAGER Database. J Lipid Res. 2010. 51:1546–1553.16. Sattar N, Preiss D, Murray HM, Welsh P, Buckley BM, de Craen AJ, Seshasai SR, McMurray JJ, Freeman DJ, Jukema JW, Macfarlane PW, Packard CJ, Stott DJ, Westendorp RG, Shepherd J, Davis BR, Pressel SL, Marchioli R, Marfisi RM, Maggioni AP, Tavazzi L, Tognoni G, Kjekshus J, Pedersen TR, Cook TJ, Gotto AM, Clearfield MB, Downs JR, Nakamura H, Ohashi Y, Mizuno K, Ray KK, Ford I. Statins and risk of incident diabetes: a collaborative meta-analysis of randomised statin trials. Lancet. 2010. 375:735–742.17. Hao M, Bogan JS. Cholesterol regulates glucose-stimulated insulin secretion through phosphatidylinositol 4,5-bisphosphate. J Biol Chem. 2009. 284:29489–29498.18. Cholesterol Treatment Trialists' (CTT) Collaborators. Kearney PM, Blackwell L, Collins R, Keech A, Simes J, Peto R, Armitage J, Baigent C. Efficacy of cholesterol-lowering therapy in 18,686 people with diabetes in 14 randomised trials of statins: a meta-analysis. Lancet. 2008. 371:117–125.19. Duggal JK, Singh M, Attri N, Singh PP, Ahmed N, Pahwa S, Molnar J, Singh S, Khosla S, Arora R. Effect of niacin therapy on cardiovascular outcomes in patients with coronary artery disease. J Cardiovasc Pharmacol Ther. 2010. 15:158–166.20. Kuvin JT, Dave DM, Sliney KA, Mooney P, Patel AR, Kimmelstiel CD, Karas RH. Effects of extended-release niacin on lipoprotein particle size, distribution, and inflammatory markers in patients with coronary artery disease. Am J Cardiol. 2006. 98:743–745.21. Yvan-Charvet L, Kling J, Pagler T, Li H, Hubbard B, Fisher T, Sparrow CP, Taggart AK, Tall AR. Cholesterol efflux potential and antiinflammatory properties of high-density lipoprotein after treatment with niacin or anacetrapib. Arterioscler Thromb Vasc Biol. 2010. 30:1430–1438.22. Wu BJ, Yan L, Charlton F, Witting P, Barter PJ, Rye KA. Evidence that niacin inhibits acute vascular inflammation and improves endothelial dysfunction independent of changes in plasma lipids. Arterioscler Thromb Vasc Biol. 2010. 30:968–975.23. Bruckert E, Labreuche J, Amarenco P. Meta-analysis of the effect of nicotinic acid alone or in combination on cardiovascular events and atherosclerosis. Atherosclerosis. 2010. 210:353–361.24. Guyton JR. Extended-release niacin for modifying the lipoprotein profile. Expert Opin Pharmacother. 2004. 5:1385–1398.25. Maccubbin D, Koren MJ, Davidson M, Gavish D, Pasternak RC, Macdonell G, Mallick M, Sisk CM, Paolini JF, Mitchel Y. Flushing profile of extended-release niacin/laropiprant versus gradually titrated niacin extended-release in patients with dyslipidemia with and without ischemic cardiovascular disease. Am J Cardiol. 2009. 104:74–81.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- The Role of Oxidized Low Density Lipoprotein in Atherogenesis
- Mechanism, clinical consequences, and management of dyslipidemia in children with nephrotic syndrome
- High Density Lipoprotein: A Therapeutic Target in Type 2 Diabetes
- Low Density Lipoprotein Cholesterol Target Goal Attainment Rate in Korean Patients with Diabetes
- A Case of Familial Hypercholesterolemia with Diabetes Mellitus