Diabetes Metab J.
2012 Apr;36(2):128-135. 10.4093/dmj.2012.36.2.128.
Effect of Eplerenone, a Selective Aldosterone Blocker, on the Development of Diabetic Nephropathy in Type 2 Diabetic Rats
- Affiliations
-
- 1Division of Endocrinology and Metabolism, Department of Internal Medicine, Korea University College of Medicine, Seoul, Korea. nhkendo@gmail.com
- KMID: 2281415
- DOI: http://doi.org/10.4093/dmj.2012.36.2.128
Abstract
- BACKGROUND
Aldosterone antagonists are reported to have beneficial effects on diabetic nephropathy by effective blocking of the renin-angiotensin-aldosterone system. We investigated the renoprotective effect of the selective aldosterone receptor blocker eplerenone, the angiotensin converting enzyme inhibitor lisinopril, and combined eplerenone and lisinopril treatment in type 2 diabetic rats.
METHODS
Animals were divided into six groups as follows: Otsuka Long-Evans Tokushima Fatty (OLETF) rat control, OLETF rats treated with a low dose of eplerenone (50 mg/kg/day), OLETF rats treated with a high dose of eplerenone (200 mg/kg/day), OLETF rats treated with lisinopril (10 mg/kg/day), OLETF rats treated with a combination of both drugs (eplerenone 200 mg/kg/day and lisinopril 10 mg/kg/day), and obese non-diabetic Long-Evans Tokushima Otsuka rats for 26 weeks.
RESULTS
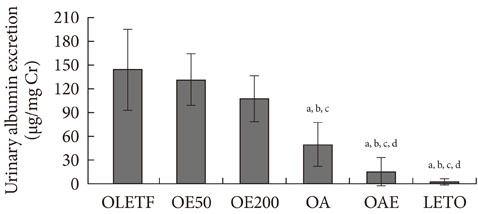
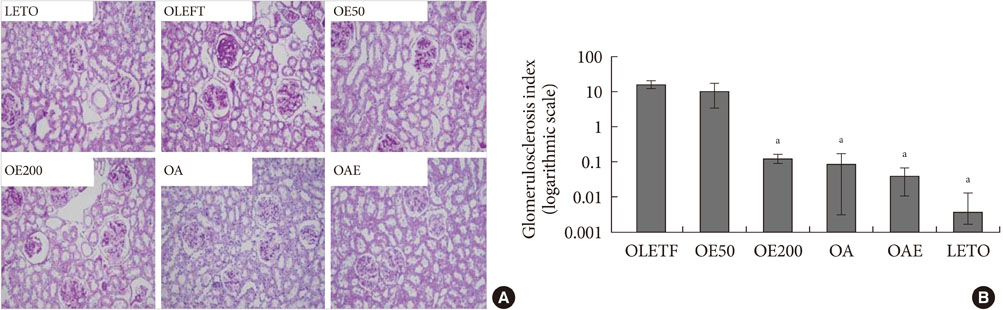
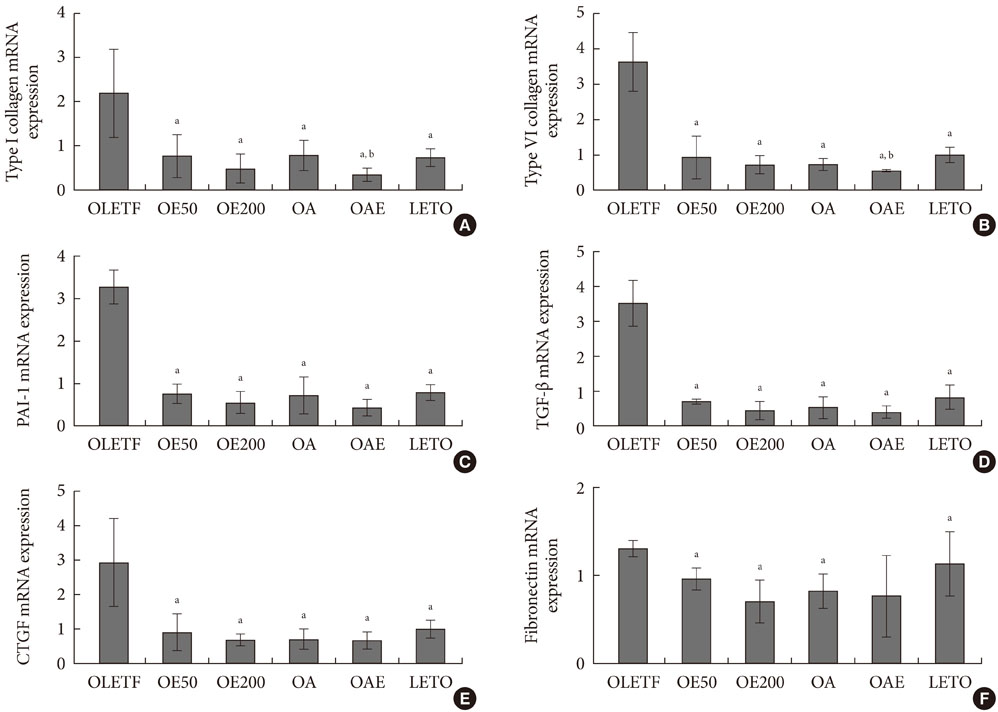
Urinary albumin excretion was significantly lower in the lisinopril group, but not in the eplerenone group. Urinary albumin excretion was decreased in the combination group than in the lisinopril group. Glomerulosclerosis and renal expression of type I and type IV collagen, plasminogen activator inhibitor-1, transforming growth factor-beta1, connective tissue growth factor, and fibronectin mRNA were markedly decreased in the lisinopril, eplerenone, and combination groups.
CONCLUSION
Eplerenone and lisinopril combination showed additional benefits on type 2 diabetic nephropathy compared to monotherapy of each drug.
Keyword
MeSH Terms
-
Aldosterone
Animals
Collagen Type IV
Connective Tissue Growth Factor
Diabetic Nephropathies
Fibronectins
Lisinopril
Mineralocorticoid Receptor Antagonists
Peptidyl-Dipeptidase A
Plasminogen Activators
Rats
Rats, Inbred OLETF
Receptors, Mineralocorticoid
Renin-Angiotensin System
RNA, Messenger
Spironolactone
Aldosterone
Collagen Type IV
Connective Tissue Growth Factor
Fibronectins
Lisinopril
Mineralocorticoid Receptor Antagonists
Peptidyl-Dipeptidase A
Plasminogen Activators
RNA, Messenger
Receptors, Mineralocorticoid
Spironolactone
Figure
Reference
-
1. Gurley SB, Coffman TM. The renin-angiotensin system and diabetic nephropathy. Semin Nephrol. 2007. 27:144–152.2. Lewis EJ, Hunsicker LG, Bain RP, Rohde RD. The effect of angiotensin-converting-enzyme inhibition on diabetic nephropathy. The Collaborative Study Group. N Engl J Med. 1993. 329:1456–1462.3. Pitt B. "Escape" of aldosterone production in patients with left ventricular dysfunction treated with an angiotensin converting enzyme inhibitor: implications for therapy. Cardiovasc Drugs Ther. 1995. 9:145–149.4. Sato A, Hayashi K, Naruse M, Saruta T. Effectiveness of aldosterone blockade in patients with diabetic nephropathy. Hypertension. 2003. 41:64–68.5. Bertocchio JP, Warnock DG, Jaisser F. Mineralocorticoid receptor activation and blockade: an emerging paradigm in chronic kidney disease. Kidney Int. 2011. 79:1051–1060.6. Jain G, Campbell RC, Warnock DG. Mineralocorticoid receptor blockers and chronic kidney disease. Clin J Am Soc Nephrol. 2009. 4:1685–1691.7. Nagase M. Activation of the aldosterone/mineralocorticoid receptor system in chronic kidney disease and metabolic syndrome. Clin Exp Nephrol. 2010. 14:303–314.8. Han SY, Kim CH, Kim HS, Jee YH, Song HK, Lee MH, Han KH, Kim HK, Kang YS, Han JY, Kim YS, Cha DR. Spironolactone prevents diabetic nephropathy through an anti-inflammatory mechanism in type 2 diabetic rats. J Am Soc Nephrol. 2006. 17:1362–1372.9. Taira M, Toba H, Murakami M, Iga I, Serizawa R, Murata S, Kobara M, Nakata T. Spironolactone exhibits direct renoprotective effects and inhibits renal renin-angiotensin-aldosterone system in diabetic rats. Eur J Pharmacol. 2008. 589:264–271.10. Fujisawa G, Okada K, Muto S, Fujita N, Itabashi N, Kusano E, Ishibashi S. Spironolactone prevents early renal injury in streptozotocin-induced diabetic rats. Kidney Int. 2004. 66:1493–1502.11. Chrysostomou A, Pedagogos E, MacGregor L, Becker GJ. Double-blind, placebo-controlled study on the effect of the aldosterone receptor antagonist spironolactone in patients who have persistent proteinuria and are on long-term angiotensin-converting enzyme inhibitor therapy, with or without an angiotensin II receptor blocker. Clin J Am Soc Nephrol. 2006. 1:256–262.12. Struthers A, Krum H, Williams GH. A comparison of the aldosterone-blocking agents eplerenone and spironolactone. Clin Cardiol. 2008. 31:153–158.13. Craft J. Eplerenone (Inspra), a new aldosterone antagonist for the treatment of systemic hypertension and heart failure. Proc (Bayl Univ Med Cent). 2004. 17:217–220.14. Goa KL, Haria M, Wilde MI. Lisinopril. A review of its pharmacology and use in the management of the complications of diabetes mellitus. Drugs. 1997. 53:1081–1105.15. Ma LJ, Nakamura S, Aldigier JC, Rossini M, Yang H, Liang X, Nakamura I, Marcantoni C, Fogo AB. Regression of glomerulosclerosis with high-dose angiotensin inhibition is linked to decreased plasminogen activator inhibitor-1. J Am Soc Nephrol. 2005. 16:966–976.16. Schjoedt KJ, Rossing K, Juhl TR, Boomsma F, Rossing P, Tarnow L, Parving HH. Beneficial impact of spironolactone in diabetic nephropathy. Kidney Int. 2005. 68:2829–2836.17. Schjoedt KJ, Rossing K, Juhl TR, Boomsma F, Tarnow L, Rossing P, Parving HH. Beneficial impact of spironolactone on nephrotic range albuminuria in diabetic nephropathy. Kidney Int. 2006. 70:536–542.18. Van den Meiracker AH, Baggen RG, Pauli S, Lindemans A, Vulto AG, Poldermans D, Boomsma F. Spironolactone in type 2 diabetic nephropathy: Effects on proteinuria, blood pressure and renal function. J Hypertens. 2006. 24:2285–2292.19. Lee MY, Shim MS, Kim BH, Hong SW, Choi R, Lee EY, Nam SM, Kim GW, Shin JY, Shin YG, Chung CH. Effects of spironolactone and losartan on diabetic nephropathy in a type 2 diabetic rat model. Diabetes Metab J. 2011. 35:130–137.20. Kang YS, Ko GJ, Lee MH, Song HK, Han SY, Han KH, Kim HK, Han JY, Cha DR. Effect of eplerenone, enalapril and their combination treatment on diabetic nephropathy in type II diabetic rats. Nephrol Dial Transplant. 2009. 24:73–84.21. Lai L, Chen J, Hao CM, Lin S, Gu Y. Aldosterone promotes fibronectin production through a Smad2-dependent TGF-beta1 pathway in mesangial cells. Biochem Biophys Res Commun. 2006. 348:70–75.22. Nagai Y, Miyata K, Sun GP, Rahman M, Kimura S, Miyatake A, Kiyomoto H, Kohno M, Abe Y, Yoshizumi M, Nishiyama A. Aldosterone stimulates collagen gene expression and synthesis via activation of ERK1/2 in rat renal fibroblasts. Hypertension. 2005. 46:1039–1045.23. Terada Y, Kuwana H, Kobayashi T, Okado T, Suzuki N, Yoshimoto T, Hirata Y, Sasaki S. Aldosterone-stimulated SGK1 activity mediates profibrotic signaling in the mesangium. J Am Soc Nephrol. 2008. 19:298–309.24. Brown NJ, Nakamura S, Ma L, Nakamura I, Donnert E, Freeman M, Vaughan DE, Fogo AB. Aldosterone modulates plasminogen activator inhibitor-1 and glomerulosclerosis in vivo. Kidney Int. 2000. 58:1219–1227.25. Gillespie EL, White CM, Kardas M, Lindberg M, Coleman CI. The impact of ACE inhibitors or angiotensin II type 1 receptor blockers on the development of new-onset type 2 diabetes. Diabetes Care. 2005. 28:2261–2266.26. Shiuchi T, Cui TX, Wu L, Nakagami H, Takeda-Matsubara Y, Iwai M, Horiuchi M. ACE inhibitor improves insulin resistance in diabetic mouse via bradykinin and NO. Hypertension. 2002. 40:329–334.27. Miana M, de Las Heras N, Rodriguez C, Sanz-Rosa D, Martin-Fernandez B, Mezzano S, Lahera V, Martinez-Gonzalez J, Cachofeiro V. Effect of eplerenone on hypertension-associated renal damage in rats: potential role of peroxisome proliferator activated receptor gamma (PPAR-gamma). J Physiol Pharmacol. 2011. 62:87–94.28. DREAM Trial Investigators. Bosch J, Yusuf S, Gerstein HC, Pogue J, Sheridan P, Dagenais G, Diaz R, Avezum A, Lanas F, Probstfield J, Fodor G, Holman RR. Effect of ramipril on the incidence of diabetes. N Engl J Med. 2006. 355:1551–1562.29. Epstein M, Williams GH, Weinberger M, Lewin A, Krause S, Mukherjee R, Patni R, Beckerman B. Selective aldosterone blockade with eplerenone reduces albuminuria in patients with type 2 diabetes. Clin J Am Soc Nephrol. 2006. 1:940–951.30. Bakris GL, Weir MR. Angiotensin-converting enzyme inhibitor-associated elevations in serum creatinine: is this a cause for concern? Arch Intern Med. 2000. 160:685–693.31. Schjoedt KJ, Christensen PK, Jorsal A, Boomsma F, Rossing P, Parving HH. Autoregulation of glomerular filtration rate during spironolactone treatment in hypertensive patients with type 1 diabetes: a randomized crossover trial. Nephrol Dial Transplant. 2009. 24:3343–3349.32. Mavrakanas TA, Cheva A, Kallaras K, Karkavelas G, Mironidou-Tzouveleki M. Effect of ramipril alone compared to ramipril with eplerenone on diabetic nephropathy in streptozocin-induced diabetic rats. Pharmacology. 2010. 86:85–91.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Effect of Spironolactone on Progression of Nephropathy in Type 2 Diabetic Rat Model
- Aldosterone Receptor Blockade Prevents Inflammatory Reaction on Type 2 Diabetic Nephropathy
- Glycemic Control in Diabetic Patients with Diabetic Nephropathy
- Recent Updates on Diabetic Nephropathy
- Aldosterone Rapidly Enhances Levels of the Striatin and Caveolin-1 Proteins in Rat Kidney: The Role of the Mineralocorticoid Receptor