Korean J Pediatr Infect Dis.
2012 Apr;19(1):28-36.
Prevalence of Viruses with Diarrhea among Hospitalized Children West Gyeonggi Province
- Affiliations
-
- 1Department of Pediatrics, Bucheon St Mary's Hospital, College of Medicine, The Catholic University of Korea, Seoul, Korea. leewb@catholic.ac.kr
- 2Clinical Medical Research Institute, Bucheon St Mary's Hospital, College of Medicine, The Catholic University of Korea, Seoul, Korea.
Abstract
- PURPOSE
This study was conducted to evaluate epidemiological data of the viral pathogens obtained from stool exams and provide information on the regional prevalence of infectious diarrheal disease west in Gyeonggi Province, Korea.
METHODS
We enrolled a cohort of children <10 years of age admitted for treatment of acute diarrhea at Bucheon St. Mary's Hospital, College of Medicine, The Catholic University of Korea. In total, 310 fecal specimens, documented to be free of common bacterial pathogens, were collected from pediatric patients during a 12-month period from January to December 2009 and were tested for the presence of rotavirus, parechovirus, adenovirus, astrovirus, enterovirus, and norovirus using polymerase chain reaction (PCR) and reverse transcription polymerase chain reaction (RT-PCR) assay.
RESULTS
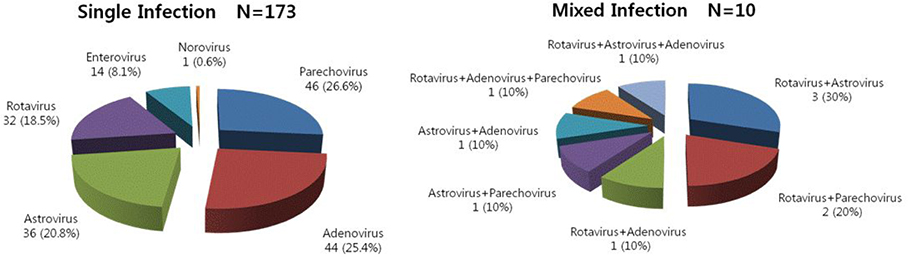
The most common virus was parechovirus (16%), followed by adenovirus (15%), astrovirus (14%), rotavirus (13%), and enterovirus (5%). Interestingly, only one of the specimens was positive for norovirus. Single infection cases were detected in 173 (55.8%) of the 310 children, whereas mixed viral infections were detected in 10 (3.2%) of the same children. Viral gastroenteritis generally showed a double peak of incidence. Parechovirus, rotavirus, and adenovirus shared a similar pattern of peak incidence with overall viruses; however, astrovirus infections occurred more frequently in the spring. Eighty-five percent of the confirmed viral gastroenteritis cases developed in under 24 months.
CONCLUSION
The results support the importance of parechovirus, adenovirus, astrovirus, and enterovirus as causative agents of diarrhea in children, which may be underestimated by current routine diagnostic testing.
Keyword
MeSH Terms
Figure
Reference
-
1. Clark B, McKendrick M. A review of viral gastroenteritis. Curr Opin Infect Dis. 2004. 17:461–469.2. Cohen MB. Etiology and mechanisms of acute infectious diarrhea in infants in the United States. J Pediatr. 1991. 118:S34–S39.
Article3. Oh DY, Gaedicke G, Schreier E. Viral agents of acute gastroenteritis in German children: prevalence and molecular diversity. J Med Virol. 2003. 71:82–93.
Article4. Glass RI, Bresee J, Jiang B, Gentsch J, Ando T, Fankhauser R, et al. Gastroenteritis viruses: an overview. Novartis Found Symp. 2001. 238:5–19. discussion -25.
Article5. Medici MC, Martinelli M, Arcangeletti MC, Pinardi F, De Conto F, Dodi I, et al. Epidemiological aspects of human rotavirus infection in children hospitalized with acute gastroenteritis in an area of northern Italy. Acta Biomed. 2004. 75:100–106.6. Iritani N, Seto Y, Kubo H, Murakami T, Haruki K, Ayata M, et al. Prevalence of Norwalk-like virus infections in cases of viral gastroenteritis among children in Osaka City, Japan. J Clin Microbiol. 2003. 41:1756–1759.
Article7. Chung JK, Song HJ, Kim SH, Seo JJ, Kee HY, Kim ES, et al. Epidemiological study of viral diarrhea in Gwangju area during 2000-2002. J Bacteriol Virol. 2006. 36:195–203.
Article8. Korea Centers for Disease Control and Prevention. 2008 Disease Control White Paper. 2009. 306.9. Bon F, Fascia P, Dauvergne M, Tenenbaum D, Planson H, Petion AM, et al. Prevalence of group A rotavirus, human calicivirus, astrovirus, and adenovirus type 40 and 41 infections among children with acute gastroenteritis in Dijon, France. J Clin Microbiol. 1999. 37:3055–3058.
Article10. Persing DH, Smith TF, Tenover FC, White TJ. Diagnostic Molecular Microbiology: Principles and Applications. 1993. Washington, D.C.: American Society for Microbiology.11. Zhang Z, Mitchell DK, Afflerbach C, Jakab F, Walter J, Zhang YJ, et al. Quantitation of human astrovirus by real-time reverse-transcription-polymerase chain reaction to examine correlation with clinical illness. J Virol Methods. 2006. 134:190–196.
Article12. Noordhoek GT, Weel JF, Poelstra E, Hooghiemstra M, Brandenburg AH. Clinical validation of a new real-time PCR assay for detection of enteroviruses and parechoviruses, and implications for diagnostic procedures. J Clin Virol. 2008. 41:75–80.
Article13. Wolf S, Williamson WM, Hewitt J, Rivera-Aban M, Lin S, Ball A, et al. Sensitive multiplex real-time reverse transcription-PCR assay for the detection of human and animal noroviruses in clinical and environmental samples. Appl Environ Microbiol. 2007. 73:5464–5470.
Article14. Koo HL, Ajami N, Atmar RL, DuPont HL. Noroviruses: The leading cause of gastroenteritis worldwide. Discov Med. 2010. 10:61–70.15. Atmar RL, Estes MK. The epidemiologic and clinical importance of norovirus infection. Gastroenterol Clin North Am. 2006. 35:275–290. viii.
Article16. Stanway G, Joki-Korpela P, Hyypiä T. Human parechoviruses-- biology and clinical significance. Rev Med Virol. 2000. 10:57–69.17. Phan TG, Nguyen TA, Shimizu H, Yagyu F, Okitsu S, Müller WE, et al. Identification of enteroviral infection among infants and children admitted to hospital with acute gastroentritis in Ho Chi Minh City, Vietnam. J Med Virol. 2005. 77:257–264.
Article18. Pham NT, Trinh QD, Chan-It W, Khamrin P, Shimizu H, Okitsu S, et al. A novel RT-multiplex PCR for detection of Aichi virus, human parechovirus, enteroviruses, and human bocavirus among infants and children with acute gastroenteritis. J Virol Methods. 2010. 169:193–197.
Article19. Kim KH, Yang JM, Joo SI, Cho YG, Glass RI, Cho YJ. Importance of rotavirus and adenovirus types 40 and 41 in acute gastroenteritis in Korean children. J Clin Microbiol. 1990. 28:2279–2284.
Article20. Patel MM, Widdowson MA, Glass RI, Akazawa K, Vinjé J, Parashar UD. Systematic literature review of role of noroviruses in sporadic gastroenteritis. Emerg Infect Dis. 2008. 14:1224–1231.
Article21. Harada S, Okada M, Yahiro S, Nishimura K, Matsuo S, Miyasaka J, et al. Surveillance of pathogens in outpatients with gastroenteritis and characterization of sapovirus strains between 2002 and 2007 in Kumamoto Prefecture, Japan. J Med Virol. 2009. 81:1117–1127.
Article22. Moreno-Espinosa S, Farkas T, Jiang X. Human caliciviruses and pediatric gastroenteritis. Semin Pediatr Infect Dis. 2004. 15:237–245.
Article23. Medici MC, Martinelli M, Abelli LA, Ruggeri FM, Di Bartolo I, Arcangeletti MC, et al. Molecular epidemiology of norovirus infections in sporadic cases of viral gastroenteritis among children in Northern Italy. J Med Virol. 2006. 78:1486–1492.
Article24. McIver CJ, Hansman G, White P, Doultree JC, Catton M, Rawlinson WD. Diagnosis of enteric pathogens in children with gastroenteritis. Pathology (Phila). 2001. 33:353–358.
Article25. Colomba C, Saporito L, Giammanco GM, De Grazia S, Ramirez S, Arista S, et al. Norovirus and gastroenteritis in hospitalized children, Italy. Emerg Infect Dis. 2007. 13:1389–1391.
Article26. Junquera CG, de Baranda CS, Mialdea OG, Serrano EB, Sánchez-Fauquier A. Prevalence and clinical characteristics of norovirus gastroenteritis among hospitalized children in Spain. Pediatr Infect Dis J. 2009. 28:604–607.
Article27. Levidiotou S, Gartzonika C, Papaventsis D, Christaki C, Priavali E, Zotos N, et al. Viral agents of acute gastroenteritis in hospitalized children in Greece. Clin Microbiol Infect. 2009. 15:596–598.
Article28. Zheng DP, Ando T, Fankhauser RL, Beard RS, Glass RI, Monroe SS. Norovirus classification and proposed strain nomenclature. Virology. 2006. 346:312–323.
Article29. Wang QH, Han MG, Cheetham S, Souza M, Funk JA, Saif LJ. Porcine noroviruses related to human noroviruses. Emerg Infect Dis. 2005. 11:1874–1881.
Article30. Siebenga JJ, Vennema H, Vinjé J, Lee BE, Pang XL, et al. Norovirus illness is a global problem: emergence and spread of norovirus GII.4 variants, 2001-2007. J Infect Dis. 2009. 200:802–812.
Article31. Kim KH. Small, round-structured virus in Seoul: Determination of its importance and direct sequencing of a genome. J Korean Soc Virol. 1995. 25:23–30.32. Chang MY, Yang JM, Kim KH. Expression and antigenicity of replicase protein from snow mountain-like caliciviruses, Korean isolates. J Korean Soc Virol. 1997. 27:151–160.33. Jee Y, Kim KS, Cheon DS, Park JK, Kang YH, Chung YS, et al. Sequence analysis of small round structured viruses (SRSV) isolated from a diarrheal patient in Wonju. J Korean Soc Virol. 1999. 29:247–259.34. Marie-Cardine A, Gourlain K, Mouterde O, Castignolles N, Hellot MF, Mallet E, et al. Epidemiology of acute viral gastroenteritis in children hospitalized in Rouen, France. Clin Infect Dis. 2002. 34:1170–1178.
Article35. Román E, Wilhelmi I, Colomina J, Villar J, Cilleruelo ML, Nebreda V, et al. Acute viral gastroenteritis: proportion and clinical relevance of multiple infections in Spanish children. J Med Microbiol. 2003. 52:435–440.
Article36. Mitchell DK, Monroe SS, Jiang X, Matson DO, Glass RI, Pickering LK. Virologic features of an astrovirus diarrhea outbreak in a day care center revealed by reverse transcriptase-polymerase chain reaction. J Infect Dis. 1995. 172:1437–1444.
Article37. Chiba S, Sakuma Y, Kogasaka R, Akihara M, Horino K, Nakao T, et al. An outbreak of gastroenteritis associated with calicivirus in an infant home. J Med Virol. 1979. 4:249–254.
Article38. Cunliffe NA, Booth JA, Elliot C, Lowe SJ, Sopwith W, Kitchin N, et al. Healthcare-associated viral gastroenteritis among children in a large pediatric hospital, United Kingdom. Emerg Infect Dis. 2010. 16:55–62.
Article39. Baumgarte S, de Souza Luna LK, Grywna K, Panning M, Drexler JF, Karsten C, et al. Prevalence, types, and RNA concentrations of human parechoviruses, including a sixth parechovirus type, in stool samples from patients with acute enteritis. J Clin Microbiol. 2008. 46:242–248.
Article40. Abed Y, Boivin G. Human parechovirus types 1, 2 and 3 infections in Canada. Emerg Infect Dis. 2006. 12:969–975.41. Benschop KS, Schinkel J, Minnaar RP, Pajkrt D, Spanjerberg L, Kraakman HC, et al. Human parechovirus infections in Dutch children and the association between serotype and disease severity. Clin Infect Dis. 2006. 42:204–210.
Article42. Rimoldi SG, Stefani F, Pagani C, Chenal LL, Zanchetta N, Di Bartolo I, et al. Epidemiological and clinical characteristics of pediatric gastroenteritis associated with new viral agents. Arch Virol. 2011. 156:1583–1589.
Article43. Brandt CD, Kim HW, Rodriguez WJ, Arrobio JO, Jeffries BC, Parrot RH. Rotavirus gastroenteritis and weather. J Clin Microbiol. 1982. 16:478–482.
Article44. Parashar UD, Bresee JS, Gentsch JR, Glass RI. Rotavirus. Emerg Infect Dis. 1998. 4:561–570.
Article45. Lee JI, Park SH, Kim MS, Oh YH, Yu IS, Choi BH, et al. Surveillance of acute gastroenteritis in Seoul, Korea, during May 2004 and June 2007. J Bacteriol Virol. 2009. 39:363–371.
Article46. Suzuki H, Sakai T, Tanabe N, Okabe N. Peak rotavirus activity shifted from winter to early spring in Japan. Pediatr Infect Dis J. 2005. 24:257–260.
Article47. Iturriza-Gómara M, Dallman T, Bányai K, Böttiger B, Buesa J, Diedrich S, et al. Rotavirus surveillance in Europe, 2005-2008: web-enabled reporting and real-time analysis of genotyping and epidemiological data. J Infect Dis. 2009. 11. 1:200 Suppl 1. S215–S221.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Acute viral gastroenteritis in children hospitalized in Iksan, Korea during December 2010-June 2011
- Genetic Typing of Bovine Viral Diarrhea Viruses (BVDV) Circulating in Korea
- The Prevalence of Respiratory Viral Infection in Exacerbation of Asthma in Hospitalized Children
- Different Age Distribution between Campylobacteriosis and Nontyphoidal Salmonellosis in Hospitalized Korean Children with Acute Inflammatory Diarrhea
- Association between Sociodemographic Factors and Diarrhea in Children Under 5 Years in Rwanda