Korean J Hematol.
2011 Dec;46(4):229-238. 10.5045/kjh.2011.46.4.229.
Adenovirus as a new agent for multiple myeloma therapies: Opportunities and restrictions
- Affiliations
-
- 1Department of Pathology and Diagnostics, University of Verona Medical School, Verona, Italy. vladia.monsurro@univr.it
- 2Azienda Ospedaliera Universitaria Integrata, Verona, Italy.
- KMID: 2251983
- DOI: http://doi.org/10.5045/kjh.2011.46.4.229
Abstract
- Multiple myeloma is a malignancy of B-cells that is characterized by the clonal expansion and accumulation of malignant plasma cells in the bone marrow. This disease remains incurable, and a median survival of 3-5 years has been reported with the use of current treatments. Viral-based therapies offer promising alternatives or possible integration with current therapeutic regimens. Among several gene therapy vectors and oncolytic agents, adenovirus has emerged as a promising agent, and it is already being used for the treatment of solid tumors in humans. The main concern with the clinical use of this vector has been its high immunogenicity; adenovirus is often able to induce a strong immune response in the host. Furthermore, new limitations in the efficacy of this therapy, intrinsic to the nature of tumor cells, have been recently observed. For example, our group showed a strong antiviral phenotype in vitro and in vivo in a subset of tumors, shedding new insights that may explain the partial failure of clinical trials based on this promising new therapy. In this review, we describe novel therapeutic approaches that implement viral-based treatments in hematological malignancies and address the novelty as well as the possible limitations of these new therapies, especially in the context of the use of adenoviral vectors for treating multiple myeloma.
MeSH Terms
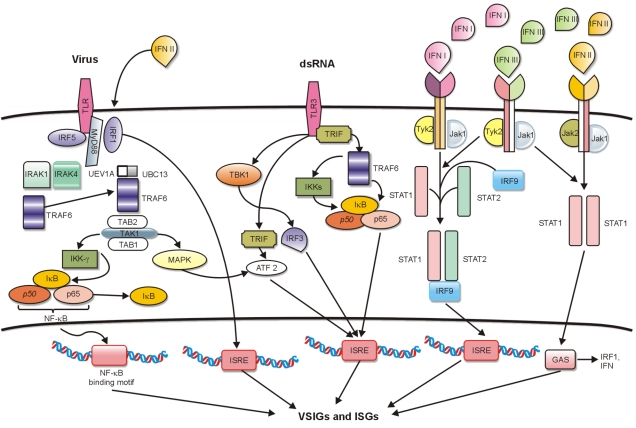
Figure
Reference
-
1. Carli PM, Coebergh JW, Verdecchia A. Variation in survival of adult patients with haematological malignancies in Europe since 1978. Eur J Cancer. 1998; 34:2253–2263. PMID: 10070296.
Article2. Barlogie B, Epstein J, Selvanayagam P, Alexanian R. Plasma cell myeloma-new biological insights and advances in therapy. Blood. 1989; 73:865–879. PMID: 2465790.
Article4. Bhardwaj A, Sethi G, Vadhan-Raj S, et al. Resveratrol inhibits proliferation, induces apoptosis, and overcomes chemoresistance through down-regulation of STAT3 and nuclear factor-kappaB-regulated antiapoptotic and cell survival gene products in human multiple myeloma cells. Blood. 2007; 109:2293–2302. PMID: 17164350.5. Delforge M, Blade J, Dimopoulos MA, et al. Treatment-related peripheral neuropathy in multiple myeloma: the challenge continues. Lancet Oncol. 2010; 11:1086–1095. PMID: 20932799.
Article6. Greipp P. Treatment paradigms for the newly diagnosed patient with multiple myeloma. Semin Hematol. 2005; 42(4 Suppl 4):S16–S21. PMID: 16344097.
Article7. Bluming AZ, Ziegler JL. Regression of Burkitt's lymphoma in association with measles infection. Lancet. 1971; 2:105–106. PMID: 4103972.
Article8. Naschberger E, Werner T, Vicente AB, et al. Nuclear factor-kappaB motif and interferon-alpha-stimulated response element co-operate in the activation of guanylate-binding protein-1 expression by inflammatory cytokines in endothelial cells. Biochem J. 2004; 379:409–420. PMID: 14741045.
Article9. Der SD, Zhou A, Williams BR, Silverman RH. Identification of genes differentially regulated by interferon alpha, beta, or gamma using oligonucleotide arrays. Proc Natl Acad Sci U S A. 1998; 95:15623–15628. PMID: 9861020.10. Thirukkumaran CM, Nodwell MJ, Hirasawa K, et al. Oncolytic viral therapy for prostate cancer: efficacy of reovirus as a biological therapeutic. Cancer Res. 2010; 70:2435–2444. PMID: 20215509.
Article11. Shirakawa T. The current status of adenovirus-based cancer gene therapy. Mol Cells. 2008; 25:462–466. PMID: 18460898.12. Pesonen S, Kangasniemi L, Hemminki A. Oncolytic adenoviruses for the treatment of human cancer: focus on translational and clinical data. Mol Pharm. 2011; 8:12–28. PMID: 21126047.
Article13. Liikanen I, Monsurro V, Ahtiainen L, et al. Induction of interferon pathways mediates in vivo resistance to oncolytic adenovirus. Mol Ther. 2011; 19:1858–1866. PMID: 21792178.
Article14. Bergelson JM, Cunningham JA, Droguett G, et al. Isolation of a common receptor for Coxsackie B viruses and adenoviruses 2 and 5. Science. 1997; 275:1320–1323. PMID: 9036860.
Article15. Turturro F. Recombinant adenovirus-mediated cytotoxic gene therapy of lymphoproliferative disorders: is CAR important for the vector to ride? Gene Ther. 2003; 10:100–104. PMID: 12571638.
Article16. Liu J, Li J, Su C, Huang B, Luo S. Soluble Fms-like tyrosine kinase-1 expression inhibits the growth of multiple myeloma in nude mice. Acta Biochim Biophys Sin (Shanghai). 2007; 39:499–506. PMID: 17622469.
Article17. Arendt BK, Ramirez-Alvarado M, Sikkink LA, et al. Biologic and genetic characterization of the novel amyloidogenic lambda light chain-secreting human cell lines, ALMC-1 and ALMC-2. Blood. 2008; 112:1931–1941. PMID: 18567838.
Article18. Senac JS, Doronin K, Russell SJ, Jelinek DF, Greipp PR, Barry MA. Infection and killing of multiple myeloma by adenoviruses. Hum Gene Ther. 2010; 21:179–190. PMID: 19788385.
Article19. Teoh G, Chen L, Urashima M, et al. Adenovirus vector-based purging of multiple myeloma cells. Blood. 1998; 92:4591–4601. PMID: 9845525.
Article20. Fernandes MS, Gomes EM, Butcher LD, et al. Growth inhibition of human multiple myeloma cells by an oncolytic adenovirus carrying the CD40 ligand transgene. Clin Cancer Res. 2009; 15:4847–4856. PMID: 19622582.
Article21. Ni H, Ergin M, Huang Q, et al. Analysis of expression of nuclear factor kappa B (NF-kappa B) in multiple myeloma: downregulation of NF-kappa B induces apoptosis. Br J Haematol. 2001; 115:279–286. PMID: 11703322.22. Meeker TC, Lay LT, Wroblewski JM, Turturro F, Li Z, Seth P. Adenoviral vectors efficiently target cell lines derived from selected lymphocytic malignancies, including anaplastic large cell lymphoma and Hodgkin's disease. Clin Cancer Res. 1997; 3:357–364. PMID: 9815692.23. Tong AW, Seamour B, Chen J, et al. CD40 ligand-induced apoptosis is Fas-independent in human multiple myeloma cells. Leuk Lymphoma. 2000; 36:543–558. PMID: 10784400.
Article24. Ren SP, Wang LS, Guo Q, et al. Genetically modified myeloma cell vaccine inducing antitumor immune response in vivo. Zhongguo Shi Yan Xue Ye Xue Za Zhi. 2006; 14:54–60. PMID: 16584592.25. Thirukkumaran CM, Russell JA, Stewart DA, Morris DG. Viral purging of haematological autografts: should we sneeze on the graft? Bone Marrow Transplant. 2007; 40:1–12. PMID: 17450184.
Article26. Kawa A, Arakawa S. The effect of attenuated vaccinia virus AS strain on multiple myeloma; a case report. Jpn J Exp Med. 1987; 57:79–81. PMID: 3626096.27. Park BH, Hwang T, Liu TC, et al. Use of a targeted oncolytic poxvirus, JX-594, in patients with refractory primary or metastatic liver cancer: a phase I trial. Lancet Oncol. 2008; 9:533–542. PMID: 18495536.
Article28. Goel A, Carlson SK, Classic KL, et al. Radioiodide imaging and radiovirotherapy of multiple myeloma using VSV(Delta51)-NIS, an attenuated vesicular stomatitis virus encoding the sodium iodide symporter gene. Blood. 2007; 110:2342–2350. PMID: 17515401.29. Forsyth P, Roldán G, George D, et al. A phase I trial of intratumoral administration of reovirus in patients with histologically confirmed recurrent malignant gliomas. Mol Ther. 2008; 16:627–632. PMID: 18253152.
Article30. Gollamudi R, Ghalib MH, Desai KK, et al. Intravenous administration of Reolysin, a live replication competent RNA virus is safe in patients with advanced solid tumors. Invest New Drugs. 2010; 28:641–649. PMID: 19572105.
Article31. Vidal L, Pandha HS, Yap TA, et al. A phase I study of intravenous oncolytic reovirus type 3 Dearing in patients with advanced cancer. Clin Cancer Res. 2008; 14:7127–7137. PMID: 18981012.
Article32. Harrington KJ, Karapanagiotou EM, Roulstone V, et al. Two-stage phase I dose-escalation study of intratumoral reovirus type 3 dearing and palliative radiotherapy in patients with advanced cancers. Clin Cancer Res. 2010; 16:3067–3077. PMID: 20484020.
Article33. Myers RM, Greiner SM, Harvey ME, et al. Preclinical pharmacology and toxicology of intravenous MV-NIS, an oncolytic measles virus administered with or without cyclophosphamide. Clin Pharmacol Ther. 2007; 82:700–710. PMID: 17971816.
Article34. Dingli D, Peng KW, Harvey ME, et al. Image-guided radiovirotherapy for multiple myeloma using a recombinant measles virus expressing the thyroidal sodium iodide symporter. Blood. 2004; 103:1641–1646. PMID: 14604966.
Article35. Msaouel P, Dispenzieri A, Galanis E. Clinical testing of engineered oncolytic measles virus strains in the treatment of cancer: an overview. Curr Opin Mol Ther. 2009; 11:43–53. PMID: 19169959.36. Peng KW, Donovan KA, Schneider U, Cattaneo R, Lust JA, Russell SJ. Oncolytic measles viruses displaying a single-chain antibody against CD38, a myeloma cell marker. Blood. 2003; 101:2557–2562. PMID: 12433686.
Article37. Au GG, Lincz LF, Enno A, Shafren DR. Oncolytic coxsackievirus A21 as a novel therapy for multiple myeloma. Br J Haematol. 2007; 137:133–141. PMID: 17391493.
Article38. Rogers JM, Diana GD, McKinlay MA. Pleconaril. A broad spectrum antipicornaviral agent. Adv Exp Med Biol. 1999; 458:69–76. PMID: 10549380.39. Rotbart HA. Antiviral therapy for enteroviral infections. Pediatr Infect Dis J. 1999; 18:632–633. PMID: 10440441.
Article40. Surah S, O'Shea S, Dunn H, et al. Utilization of HIV point-of-care testing clinics in general practice and genitourinary medicine services in south-east London. Int J STD AIDS. 2009; 20:168–169. PMID: 19255263.
Article41. Nayak S, Herzog RW. Progress and prospects: immune responses to viral vectors. Gene Ther. 2010; 17:295–304. PMID: 19907498.
Article42. Seiler MP, Cerullo V, Lee B. Immune response to helper dependent adenoviral mediated liver gene therapy: challenges and prospects. Curr Gene Ther. 2007; 7:297–305. PMID: 17979676.
Article43. Yamaguchi T, Kawabata K, Koizumi N, et al. Role of MyD88 and TLR9 in the innate immune response elicited by serotype 5 adenoviral vectors. Hum Gene Ther. 2007; 18:753–762. PMID: 17685831.
Article44. Appledorn DM, McBride A, Seregin S, et al. Complex interactions with several arms of the complement system dictate innate and humoral immunity to adenoviral vectors. Gene Ther. 2008; 15:1606–1617. PMID: 18615115.
Article45. Liu JM, Liu ZB, Zhu GH, et al. Determination of trace glucose and forecast of human diseases by affinity adsorption solid substrateroom temperature phosphorimetry based on triticum vulgaris lectin labeled with dendrimers-porphyrin dual luminescence molecule. Talanta. 2008; 74:625–631. PMID: 18371685.
Article46. Muruve DA, Petrilli V, Zaiss AK, et al. The inflammasome recognizes cytosolic microbial and host DNA and triggers an innate immune response. Nature. 2008; 452:103–107. PMID: 18288107.
Article47. Nociari M, Ocheretina O, Schoggins JW, Falck-Pedersen E. Sensing infection by adenovirus: Toll-like receptor-independent viral DNA recognition signals activation of the interferon regulatory factor 3 master regulator. J Virol. 2007; 81:4145–4157. PMID: 17251283.
Article48. Mastrangeli A, Harvey BG, Yao J, et al. "Sero-switch" adenovirus-mediated in vivo gene transfer: circumvention of anti-adenovirus humoral immune defenses against repeat adenovirus vector administration by changing the adenovirus serotype. Hum Gene Ther. 1996; 7:79–87. PMID: 8825871.49. Dharmapuri S, Peruzzi D, Aurisicchio L. Engineered adenovirus serotypes for overcoming anti-vector immunity. Expert Opin Biol Ther. 2009; 9:1279–1287. PMID: 19645630.
Article50. Boo KH, Yang JS. Intrinsic cellular defenses against virus infection by antiviral type I interferon. Yonsei Med J. 2010; 51:9–17. PMID: 20046508.
Article51. Sarkar SN, Sen GC. Novel functions of proteins encoded by viral stress-inducible genes. Pharmacol Ther. 2004; 103:245–259. PMID: 15464592.
Article52. zum Büschenfelde CM, Metzger J, Hermann C, Nicklisch N, Peschel C, Bernhard H. The generation of both T killer and Th cell clones specific for the tumor-associated antigen HER2 using retrovirally transduced dendritic cells. J Immunol. 2001; 167:1712–1719. PMID: 11466395.
Article53. Sadler AJ, Williams BR. Interferon-inducible antiviral effectors. Nat Rev Immunol. 2008; 8:559–568. PMID: 18575461.
Article54. Sung RS, Qin L, Bromberg JS. TNFalpha and IFNgamma induced by innate anti-adenoviral immune responses inhibit adenovirus-mediated transgene expression. Mol Ther. 2001; 3:757–767. PMID: 11356080.55. Wacher C, Muller M, Hofer MJ, et al. Coordinated regulation and widespread cellular expression of interferon-stimulated genes (ISG) ISG-49, ISG-54, and ISG-56 in the central nervous system after infection with distinct viruses. J Virol. 2007; 81:860–871. PMID: 17079283.
Article56. Staeheli P, Pavlovic J. Inhibition of vesicular stomatitis virus mRNA synthesis by human MxA protein. J Virol. 1991; 65:4498–4501. PMID: 1649344.
Article57. Krug RM, Shaw M, Broni B, Shapiro G, Haller O. Inhibition of influenza viral mRNA synthesis in cells expressing the interferon-induced Mx gene product. J Virol. 1985; 56:201–206. PMID: 2411949.
Article58. Monsurrò V, Beghelli S, Wang R, et al. Anti-viral state segregates two molecular phenotypes of pancreatic adenocarcinoma: potential relevance for adenoviral gene therapy. J Transl Med. 2010; 8:10. PMID: 20113473.
Article59. Marincola FM, Wang E, Herlyn M, Seliger B, Ferrone S. Tumors as elusive targets of T-cell-based active immunotherapy. Trends Immunol. 2003; 24:335–342. PMID: 12810110.
Article60. Tsai MH, Cook JA, Chandramouli GV, et al. Gene expression profiling of breast, prostate, and glioma cells following single versus fractionated doses of radiation. Cancer Res. 2007; 67:3845–3852. PMID: 17440099.
Article61. Weichselbaum RR, Ishwaran H, Yoon T, et al. An interferon-related gene signature for DNA damage resistance is a predictive marker for chemotherapy and radiation for breast cancer. Proc Natl Acad Sci U S A. 2008; 105:18490–18495. PMID: 19001271.
Article62. Martin DN, Boersma BJ, Yi M, et al. Differences in the tumor microenvironment between African-American and European-American breast cancer patients. PLoS One. 2009; 4:e4531. PMID: 19225562.
Article63. Khodarev NN, Minn AJ, Efimova EV, et al. Signal transducer and activator of transcription 1 regulates both cytotoxic and prosurvival functions in tumor cells. Cancer Res. 2007; 67:9214–9220. PMID: 17909027.
Article64. Konstantinopoulos PA, Spentzos D, Cannistra SA. Gene-expression profiling in epithelial ovarian cancer. Nat Clin Pract Oncol. 2008; 5:577–587. PMID: 18648354.
Article65. Andreu P, Colnot S, Godard C, et al. Identification of the IFITM family as a new molecular marker in human colorectal tumors. Cancer Res. 2006; 66:1949–1955. PMID: 16488993.
Article