J Korean Ophthalmol Soc.
2011 Jul;52(7):800-806. 10.3341/jkos.2011.52.7.800.
Effect of 0.1% Sodium Hyaluronate and 0.05% Cyclosporine on Tear Film Parameters after Cataract Surgery
- Affiliations
-
- 1Department of Ophthalmology, Chonnam National University Medical School, Gwangju, Korea. kcyoon@chonnam.ac.kr
- KMID: 2214704
- DOI: http://doi.org/10.3341/jkos.2011.52.7.800
Abstract
- PURPOSE
To evaluate the efficacy of topical 0.1% sodium hyaluronate (HA) and 0.05% cyclosporine A (CsA) on tear film parameters after cataract surgery.
METHODS
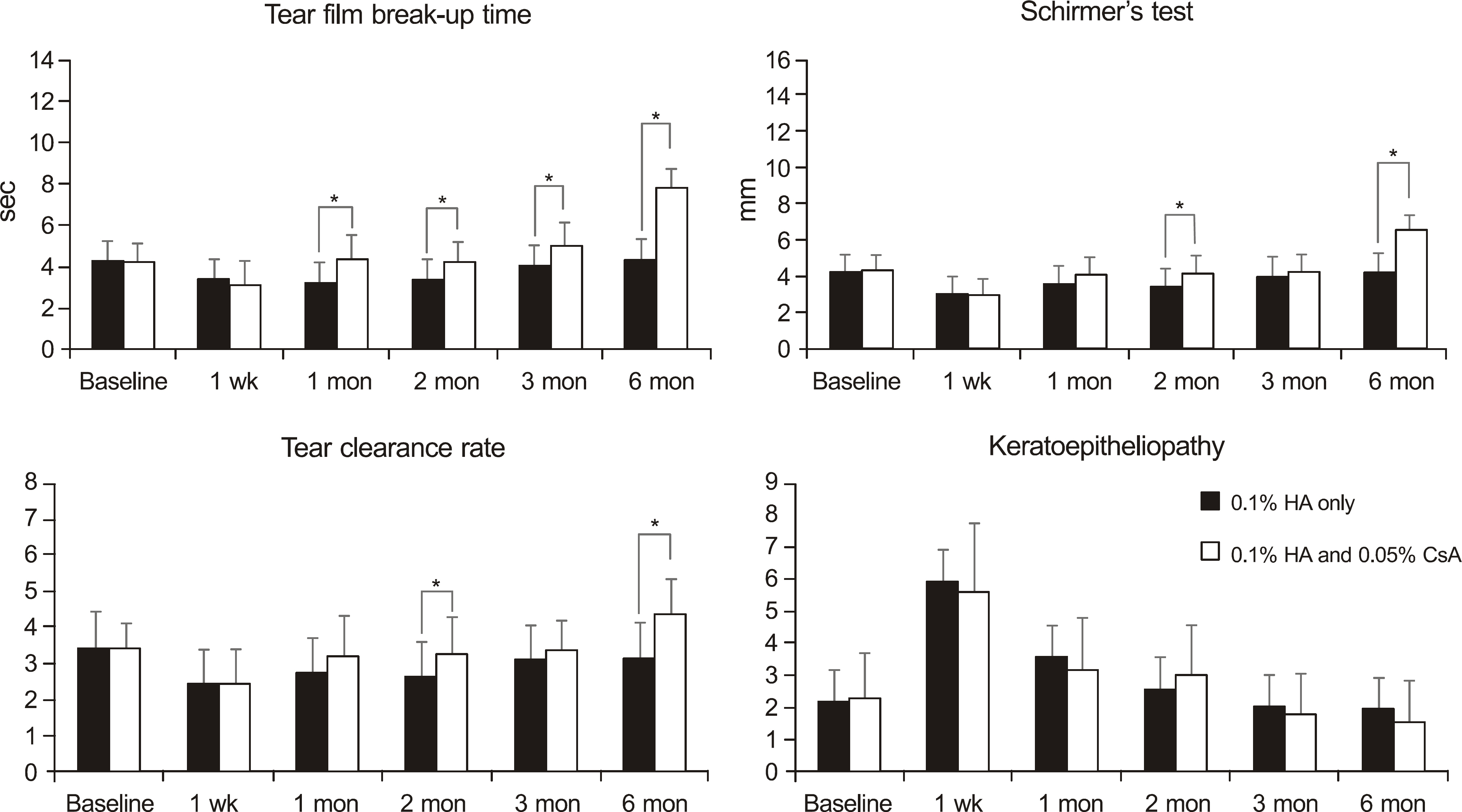
A total of 109 eyes of 86 patients who underwent cataract surgery were divided into four groups: 74 eyes without dry eye syndrome (groups 1 and 2) and 35 eyes with dry eye syndrome (groups 3 and 4). Groups 1 (37 eyes) and 3 (17 eyes) used 0.1% HA only, while Groups 2 (37 eyes) and 4 (18 eyes) used both 0.1% HA and 0.05% CsA. Tear film break-up time (BUT), Schirmer's test, tear clearance rate (TCR) and keratoepitheliopathy were evaluated before surgery and 1 week, 1 month, 2, 3 and 6 months after surgery.
RESULTS
But, Schirmer's test and TCR recovered at postoperative 3 months in groups 1 and 3, after 2 months in group 2 and after 1 month in group 4. Keratoepitheliopathy recovered at postoperative 1 month in groups 1, 2 and 4, after 2 months in group 3.
CONCLUSIONS
The combined use of topical 0.1% HA with 0.05% CsA is helpful in restoring tear film parameters after cataract surgery especially in patients with dry eye syndrome.
MeSH Terms
Figure
Cited by 3 articles
-
Effectiveness of Cyclosporine-steroid Treatment after Cataract Surgery according to Dry Eye Severity
Jae Yeong Park, Sang Cheol Yang, Young Min Park, Ji Eun Lee, Choul Yong Park, Jong Soo Lee
J Korean Ophthalmol Soc. 2019;60(9):821-828. doi: 10.3341/jkos.2019.60.9.821.Effect of 0.05% Cyclosporine A on the Ocular Surface after Photorefractive Keratectomy
Yong Il Kim, Sung Hyun Koo, Sang Won Ha, Gwang Ja Lee, Kyoo Won Lee, Young Jeung Park
J Korean Ophthalmol Soc. 2016;57(5):710-717. doi: 10.3341/jkos.2016.57.5.710.Efficacy of Topical Cyclosporine in Mild Dry Eye Patients Having Refractive Surgery
Kun Wook Kang, Hong Kyun Kim
J Korean Ophthalmol Soc. 2014;55(12):1752-1757. doi: 10.3341/jkos.2014.55.12.1752.
Reference
-
References
1. The definition of classification of dry eye diseases: report of the Definition and Classification Subcommittee of the International Dry Eye WorkShop (2007). Ocul Surf. 2007; 5:75–92.2. Horwath-Winter J, Vidic B, Schwantzer G, Schmut O. Early changes in corneal sensation, ocular surface integrity, and tear-film function after laser-assisted subepithelial keratectomy. J Cataract Refract Surg. 2004; 30:2316–21.
Article3. Li XM, Hu L, Hu J, Wang W. Investigation of dry eye disease and analysis of the pathogenic factors in patients after cataract surgery. Cornea. 2007; 26(9 Suppl 1):S16–20.
Article4. Pflugfelder SC, Solomon A, Stern ME. The diagnosis and management of dry eye: a twenty-five-year review. Cornea. 2000; 19:644–9.5. Nussenblatt RB, Palestine AG. Cyclosporine: immunology, pharmacology and therapeutic uses. Surv Ophthalmol. 1986; 31:159–69.
Article6. Hemady R, Tauber J, Foster CS. Immunosuppressive drugs in immune and inflammatory ocular disease. Surv Ophthalmol. 1991; 35:369–85.
Article7. Jabs DA, Wingard J, Green WR, et al. The eye in bone marrow transplantation. III. Conjunctival graft-vs-host disease. Arch Ophthalmol. 1989; 107:1343–8.8. Bhan AK, Fujikawa LS, Foster CS. T-cell subsets and Langerhans cells in normal and diseased conjunctiva. Am J Ophthalmol. 1982; 94:205–12.
Article9. Gilbard JP, Rossi SR, Azar DT, Heyda KG. Effect of punctal occlusion by Freeman silicone plug insertion on tear osmolarity in dry eye disorders. CLAO J. 1989; 15:216–8.10. Lee EH, Jang JW, Lew HM. The changes of tear osmolarity and protein after silicone punctal plug insertion in dry eye. J Korean Ophthalmol Soc. 2001; 42:1509–14.11. Yen MT, Pflugfelder SC, Feuer WJ. The effect of punctal occlusion on tear production, tear clearance, and ocular surface sensation in normal subjects. Am J Ophthalmol. 2001; 131:314–23.
Article12. Pflugfelder SC, De Paiva CS, Villarreal AL, Stern ME. Effects of sequential artificial tear and cyclosporine emulsion therapy on conjunctival goblet cell density and transforming growth factor-beta2 production. Cornea. 2008; 27:64–9.13. Wilson SE, Perry HD. Long-term resolution of chronic dry eye symptoms and signs after topical cyclosporine treatment. Ophthalmology. 2007; 114:76–9.
Article14. Roberts CW, Carniglia PE, Brazzo BG. Comparison of topical cyclosporine, punctal occlusion, and a combination for the treatment of dry eye. Cornea. 2007; 26:805–9.
Article15. Raphael M, Bellefqih S, Piette JC, et al. Conjunctival biopsy in Sjögren's syndrome: correlations between histological and immunohistochemical features. Histopathology. 1988; 13:191–202.
Article16. Kunert KS, Tisdale AS, Stern ME, et al. Analysis of topical cyclosporine treatment of patients with dry eye syndrome: effect on conjunctival lymphocytes. Arch Ophthalmol. 2000; 118:1489–96.17. O'Brien PD, Collum LM. Dry eye: diagnosis and current treatment strategies. Curr Allergy Asthma Rep. 2004; 4:314–9.18. Small DS, Acheampong A, Reis B, et al. Blood concentrations of cyclosporin a during long-term treatment with cyclosporin a ophthalmic emulsions in patients with moderate to severe dry eye disease. J Ocul Pharmacol Ther. 2002; 18:411–8.
Article19. Laibovitz RA, Solch S, Andriano K, et al. Pilot trial of cyclosporine 1% ophthalmic ointment in the treatment of keratoconjunctivitis sicca. Cornea. 1993; 12:315–23.
Article20. Turner K, Pflugfelder SC, Ji Z, et al. Interleukin-6 levels in the conjunctival epithelium of patients with dry eye disease treated with cyclosporine ophthalmic emulsion. Cornea. 2000; 19:492–6.
Article21. Stevenson D, Tauber J, Reis BL. Efficacy and safety of cyclosporin A ophthalmic emulsion in the treatment of moderate-to-severe dry eye disease: a dose-ranging, randomized trial. The Cyclosporin A Phase 2 Study Group. Ophthalmology. 2000; 107:967–74.22. Moon JW, Lee HJ, Shin KC, et al. Short term effects of topical cyclosporine and viscoelastic on the ocular surfaces in patients with dry eye. Korean J Ophthalmol. 2007; 21:189–94.
Article23. Xu KP, Yagi Y, Tsubota K. Decrease in corneal sensitivity and change in tear function in dry eye. Cornea. 1996; 15:235–9.
Article24. Hiroko BM. Cataract surgery in the presence of other ocular comorbidities. Steinert RF, editor. Cataract Surgery: Technique, Complication and Management. 2nd ed.Philadelphia: Saunders;2004. chap. 32.25. Walker TD. Benzalkonium toxicity. Clin Experiment Ophthalmol. 2004; 32:657.26. Cho YK, Kim MS. Dry eye after cataract surgery and associated intraoperative risk factors. Korean J Ophthalmol. 2009; 23:65–73.
Article27. Donnenfeld ED, Solomon R, Roberts CW, et al. Cyclosporine 0.05% to improve visual outcomes after multifocal intraocular lens implantation. J Cataract Refract Surg. 2010; 36:1095–100.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Effect of Sodium Hyaluronate and Cyclosporine A on Tear Film in Dry Eye Syndrome
- Effect of Topical 0.05% Cyclosporine A in Dry Eye Associated With Thyroid Ophthalmopathy
- Effect of Cyclosporin A on Tear Film and Corneal Aberration after Cataract Surgery
- Comparison of the Effects of 0.05% and 0.1% Cyclosporine for Dry Eye Syndrome Patients after Cataract Surgery
- Effects of Cyclosporine 0.05% Ophthalmic Emulsion to Improve Reduction of Tear Production after Cataract Surgery