J Korean Ophthalmol Soc.
2009 Sep;50(9):1334-1340. 10.3341/jkos.2009.50.9.1334.
Short-Term Effect of Intravitreal Bevacizumab Injection for Choroidal Neovascularization Associated With Degenerative Myopia
- Affiliations
-
- 1Department of Ophthalmology and Visual Science, College of Medicine, The Catholic University of Korea, Seoul, Korea. wklee@catholic.ac.kr
- KMID: 2212616
- DOI: http://doi.org/10.3341/jkos.2009.50.9.1334
Abstract
- PURPOSE
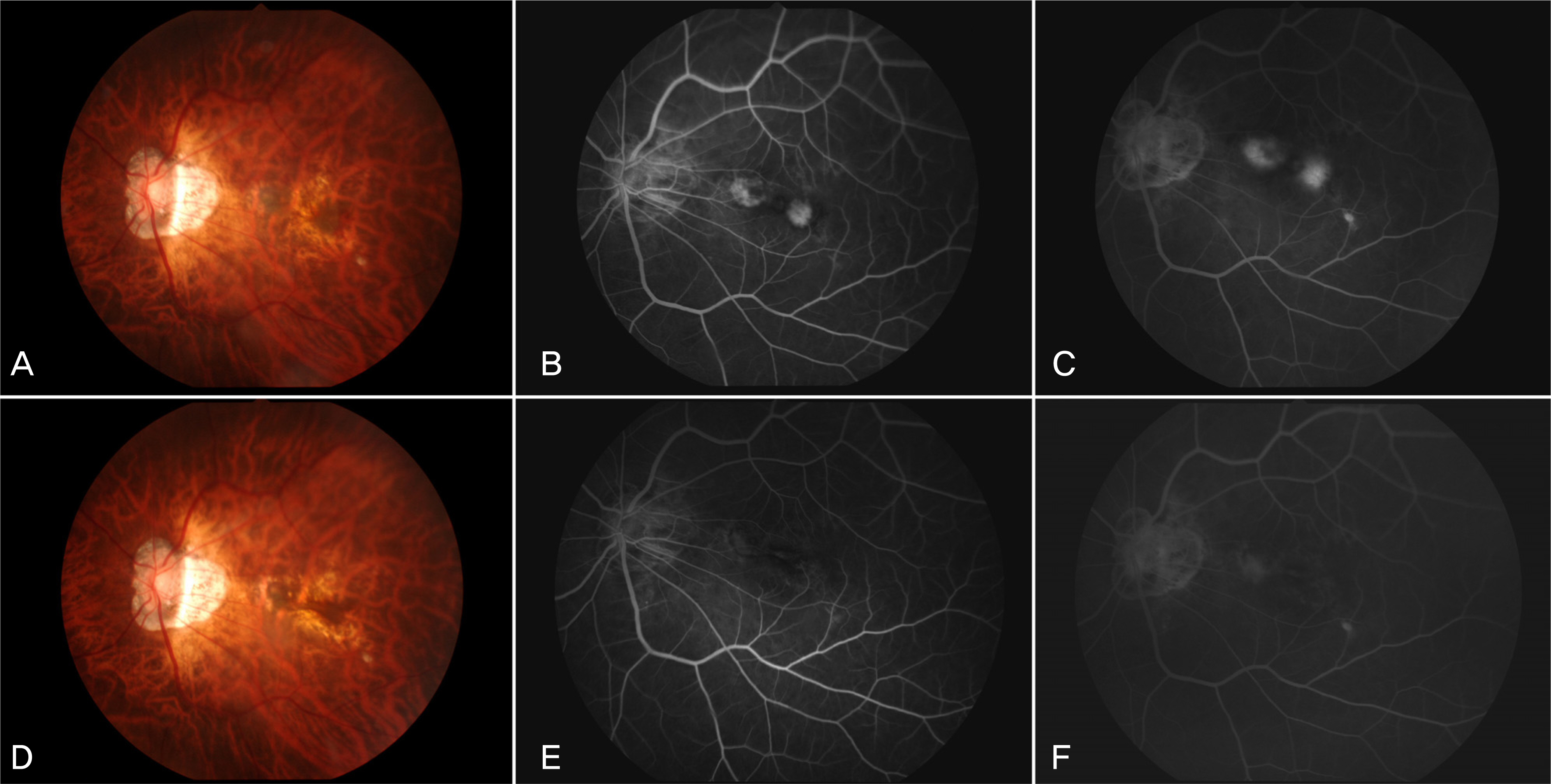
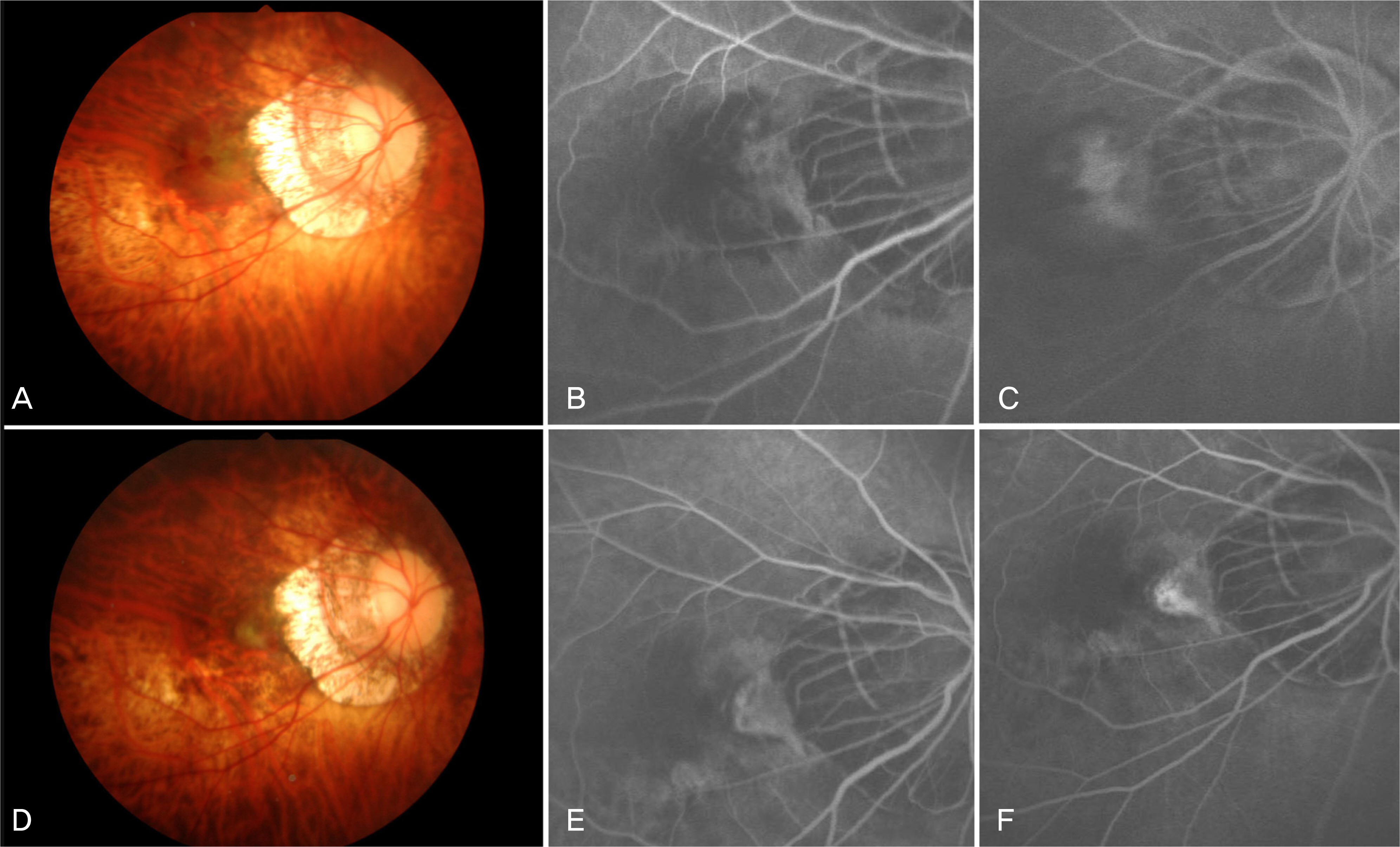
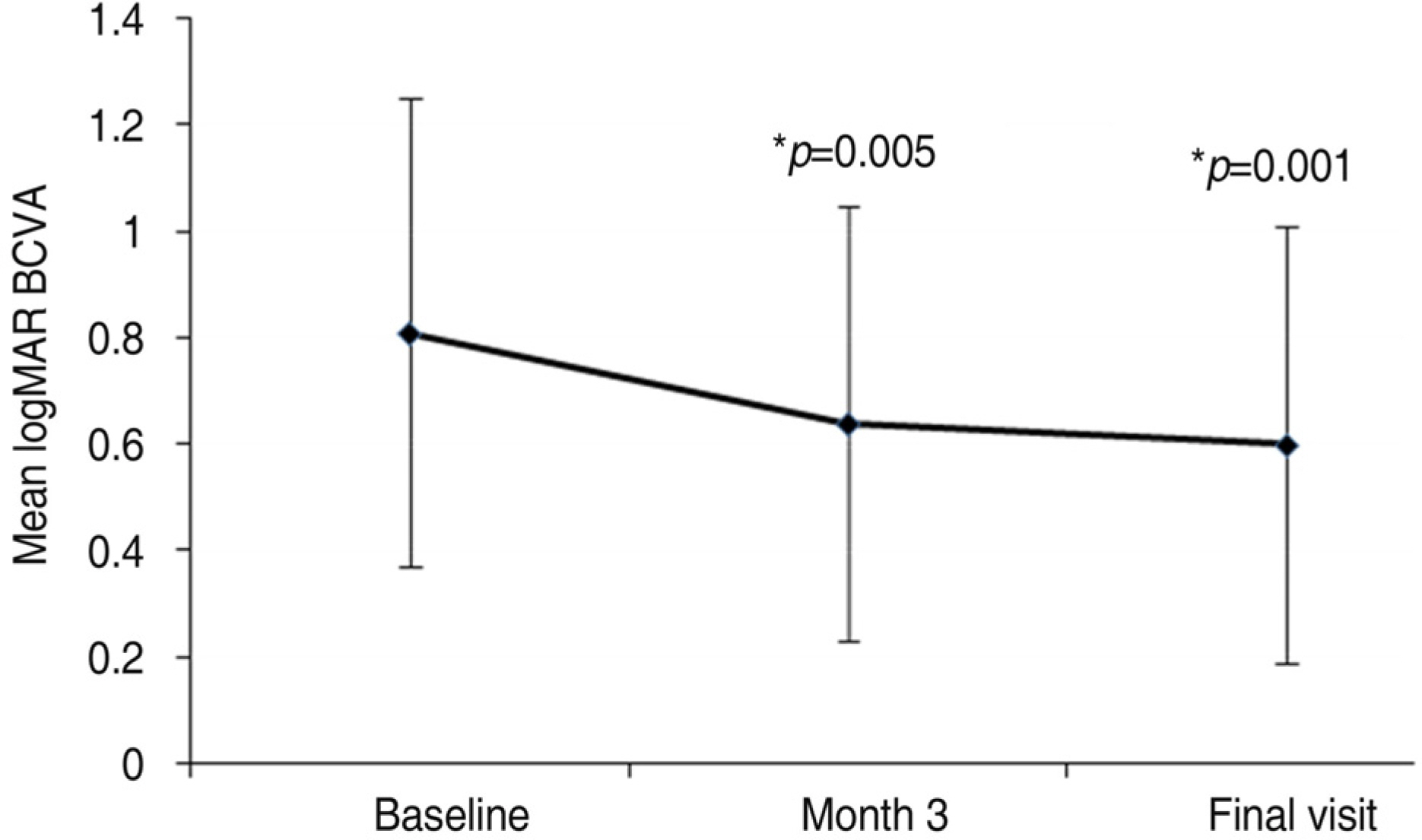
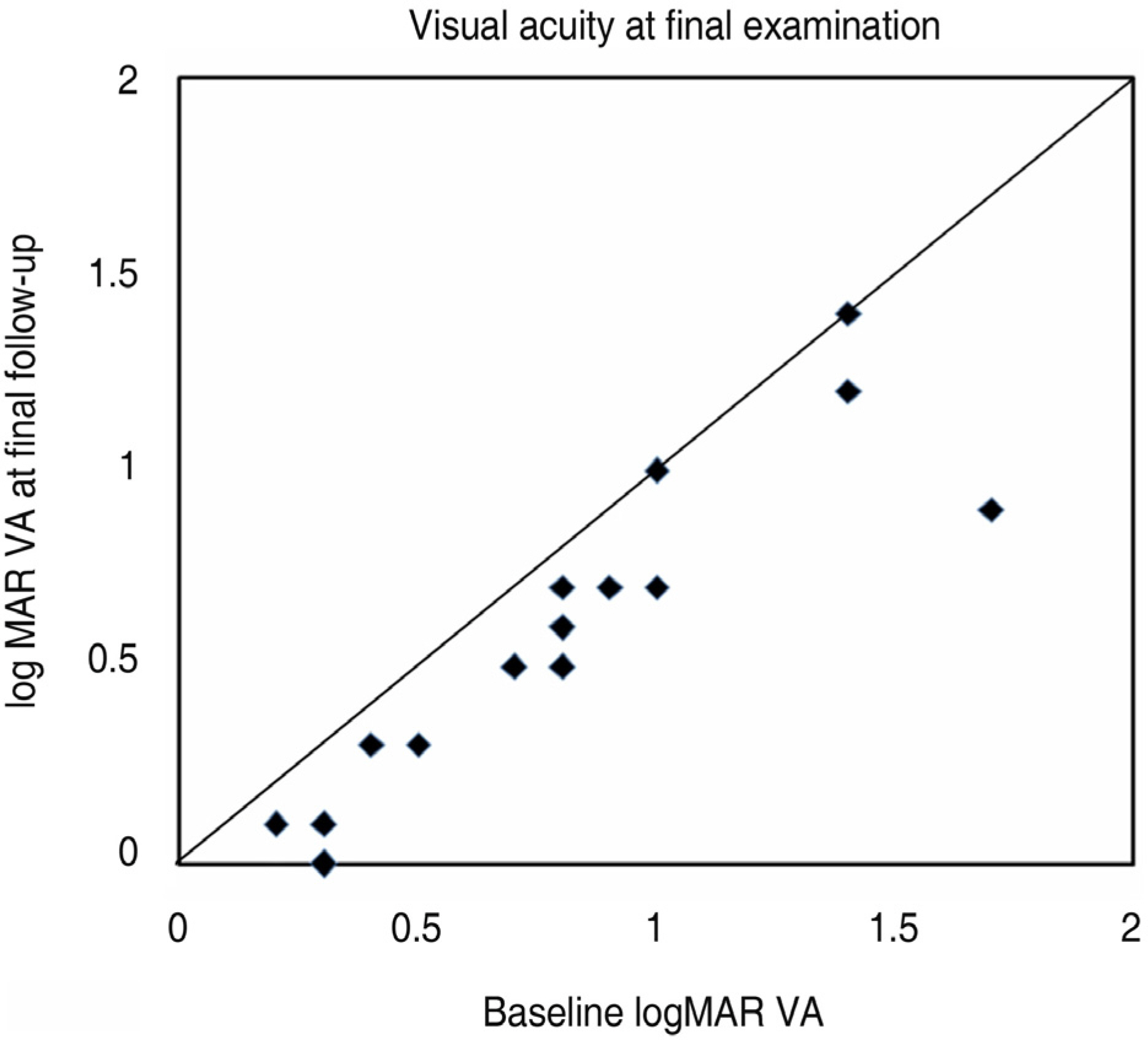
To investigate the short-term effect of intravitreal bevacizumab injection for choroidal neovascularization associated with degenerative myopia. METHODS: In 15 eyes of 15 patients, one or two consecutive intravitreal bevacizumab injections were given. The best-corrected visual acuity (BCVA) and fundus examination were evaluated at baseline and monthly thereafter. Fluorescence angiography (FA) was performed at baseline, 1 month and 3 months after treatment. When the angiographic leakage persisted 1 month after the first injection, a second injection was administered. RESULTS: The mean follow-up period was 9.7 months. The mean logarithm of the minimum angle of resolution (LogMAR) BCVA was 0.81+/-0.44 at baseline, 0.64+/-0.41 at 3 months (p=0.005), and 0.60+/-0.41 (p=0.001) at the final examination. Five eyes received a single injection, while the other ten eyes had two consecutive injections. Three months after the first injection, 14 eyes (93.3%) had no angiographic leakage, and 1 eye (6.7%) showed a decrease in leakage. The mean lines of visual improvement at 3 months and at the final examination were 1.7 and 2.1 lines, respectively. No case of vision loss was observed throughout the follow-up period. CONCLUSIONS: One or two consecutive intravitreal bevacizumab injections had favorable short-term effects on visual acuity stabilization and the regression of choroidal neovascularization associated with neovascular degenerative myopia.
MeSH Terms
Figure
Reference
-
References
1. Grossniklaus HE, Green WR. Pathologic finding in pathologic myopia. Retina. 1992; 12:127–33.2. Hotchkiss ML, Fine SL. Pathologic myopia and choroidal neovascularization. Am J Ophthalmol. 1981; 91:177–83.
Article3. Ohno-Matsui K, Yoshida T, Futagami S, et al. Patchy atrophy and lacquer cracks predispose to the development of choroidal neovascularization in pathological myopia. Br J Ophthalmol. 2003; 87:570–3.4. Hampton GR, Kohen D, Bird AC. Visual prognosis of disciform degeneration in myopia. Ophthalmology. 1983; 90:923–6.
Article5. Yoshida T, Ohno-Matsui K, Yasuzumi K, et al. Myopic choroidal neovascularization. A 10-year followup. Ophthalmology. 2003; 110:1297–305.6. Secretan M, Kuhn D, Soubrane G, Coscas G. Long-term visual outcome of choroidal neovascularization in pathologic myopia; natural history and laser treatment. Eur J Ophthalmol. 1997; 7:307–16.7. Johnson DA, Yannuzzi LA, Shakin JL, Lightman DA. Lacquer cracks following laser treatment of choroidal neovascularization in pathologic myopia. Retina. 1998; 18:118–24.
Article8. Bottoni F, Perego E, Airaghi P, et al. Surgical removal of subfoveal choraoidal neovascular membranes in high myopia. Graefes Arch Clin Exp Ophthalmol. 1999; 237:573–82.9. Uemura A, Thomas MA. Subretinal surgery for choroidal neovascularization in patients with high myopia. Arch Ophthalmol. 2000; 118:344–50.
Article10. Glacet-Bernard A, Simon P, Hamelin N, et al. Translocation of the macula for management of subfoveal choroidal neovascularization: comparision of results in age-related macular degeneration and degenerative myopia. Am J Ophthalmol. 2001; 131:78–89.11. Ichibe M, Imai K, Ohta M, et al. Foveal translocation with scleral imbrication in patients with myopic neovascular maculopathy. Am J Ophthalmol. 2001; 132:164–71.
Article12. Verteporfin in Photodynamic Therapy (VIP) Study Group. Photodynamic therapy of subfoveal choroidal neovascularization in pathologic myopia with verteporfin: 1-year results of a randomized clinical trial-VIP report no. 1. Ophthalmology. 2001; 108:841–52.13. Verteporfin in Photodynamic Therapy (VIP) Study Group. Verteporfin therapy of subfoveal choroidal neovascularization in pathologic myopia: 2-year results of a randomizedclinical trial-VIP report no. 3. Ophtalmology. 2003; 110:667–73.14. Ryu IH, Kim BG, Lee SC. Photodynamic Therapy of Subfoveal Choroidal Neovascularization in Pathologic Myopia. J Korean Ophthalmol Soc. 2003; 44:1991–5.15. Chung EJ, Oh HS, Koh HJ, et al. Photodynamic therapy in practice: A review of experiences with myopic CNV in Korean patients. J Korean Ophthalmol Soc. 2005; 46:664–70.16. Schnurrbusch UE, Jochmann C, Wiedemann P, Wolf S. Quan-titative assessment of the long-term effect of photodynamic therapy in patients with pathologic myopia. Graefes Arch Clin Exp Opththalmol. 2005; 243:829–33.
Article17. Lam DS, Liu DT, Fan DS, et al. Photodynamic therapy with verteporfin for juxtafoveal choroidal neovascularization secondary to pathologic myopia-1-year results of a prospective series. Eye. 2005; 19:834–40.
Article18. Pece A, Isola V, Vadalà M, Matranga D. Photodynamic therapy with verteporfin for subfoveal choroidal neovascularization secondary to pathologic myopia: long-term study. Retina. 2006; 26:746–51.19. Pece A, Vadalà M, Isola V, Matranga D. Photodynamic therapy with verteporfin for juxtafoveal choroidal neovascularization in pathologic myopia: a long-term followup study. Am J Ophthalmol. 2007; 143:449–54.
Article20. Degenring RF, Jonas JB. Photodynamic therapy in combination with intravitreal triamcinolone for myopic choroidal neovascularization. Acta Ophthalmol Scand. 2005; 83:621.
Article21. Chan WM, Lai TY, Wong AL, et al. Combined photodynamic therapy and intravitreal triamcinolone injection for the treatment of choroidal neovascularization secondary to pathological myopia: a pilot study. Br J Ophthalmol. 2007; 91:174–9.22. Sakaguchi H, Ikuno Y, Gomi F, et al. Intravitreal injection of bevacizumab for choroidal neovascularization associated with pathological myopia. Br J Ophthalmol. 2007; 91:161–5.23. Yamamoto I, Rogers AH, Reichel E, et al. Intravitreal bevacizumab (Avastin) as treatment for subfoveal choroidal neovascularization secondary to pathological myopia. Br J Ophthalmol. 2007; 91:157–60.24. Chan WM, Lai TY, Liu DT, Lam DS. Intravitreal bevacizumab (Avastin) for myopic choroidal neovascularization: six-month results of a prospective pilot study. Ophthalmology. 2007; 114:2190–6.25. Ruiz-Moreno JM, Gomez-Ulla F, Montero JA, et al. Intravitreous bevacizumab to treat subfoveal choroidal neovascularization in highly myopic eyes: short-term results. Eye. 2009; 23:334–8.
Article26. Mandal S, Venkatesh P, Sampangi R, Garg S. Intravitreal bevacizumab (Avastin) as primary treatment for myopic choroidal neovascularization. Eur J Ophthalmol. 2007; 17:620–6.
Article27. Hernandez-Rojas ML, Quiroz-Mercado H, Dalma-Weiszhausz J, et al. Short-term effects of intravitreal bevacizumab for subfoveal choroidal neovascularization in pathologic myopia. Retina. 2007; 27:707–12.28. Steidl SM, Pruett RC. Macular complication associated with posterior staphyloma. Am J Ophthalmol. 1997; 123:181–7.29. Brancato R, Trabucchi G, Introini U, et al. Indocyanine green angiography (ICGA) in pathologic myopia. Eur J Ophthalmol. 1996; 16:39–43.30. Avila MP, Weitter JJ, Jalkh AE, et al. Natural history of choroidal neovascularization in degenerative myopia. Ophthalmology. 1984; 91:1573–81.
Article31. Song KY, Kim HK, Kim HC. Clinical features of Choroidal Neovascularization in Patient with High Myopia. J Korean Ophthalmol Soc. 2001; 42:983–90.32. Shahar J, Avery RL, Heilweil G, et al. Electrophysiologic and retinal penetration studies following intravitreal injection of bevacizumab (Avastin). Retina. 2006; 26:262–9.
Article33. Manzano RP, Peyman GA, Khan P, et al. Testing intravitreal toxicity of bevacizumab (Avastin). Retina. 2006; 26:257–61.
Article34. Avery RL, Pieramici DJ, Rabena MD, et al. Intravitreal bevacizumab (Avastin) for neovascular age-related macular degeneration. Ophthalmology. 2006; 113:363–72.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Long-term Therapeutic Effect of Intravitreal Bevacizumab (Avastin) on Myopic Choroidal Neovascularization
- Effect of High-dose Intravitreal Bevacizumab Injection on Refractory Idiopathic Choroidal Neovasculariz
- A Case of Intravitreal Bevacizumab Injection for the Treatment of Choroidal Neovascularization in Morning Glory Syndrome
- Multifocal Electroretinogram Findings after Intravitreal Bevacizumab Injection in Choroidal Neovascularization of Age-Related Macular Degeneration
- Choroidal Neovascularization in a Patient with Best Disease