J Korean Orthop Assoc.
2008 Jun;43(3):276-286. 10.4055/jkoa.2008.43.3.276.
The Effect of Human Amniotic Membrane, Epidermal Cells and Marrow Mesenchymal Stem Cells in Healing a Skin Defect
- Affiliations
-
- 1Department of Orthopaedic Surgery, College of Medicine, Dong-A University, Busan, Korea. sskim2@dau.ac.kr
- KMID: 2186442
- DOI: http://doi.org/10.4055/jkoa.2008.43.3.276
Abstract
-
PURPOSE: The purpose of this study was to investigate the transplantation results of human amniotic membrane (HAM), epidermal cells, or marrow mesenchymal stem cells (MSCs) in healing a skin defect.
MATERIALS AND METHODS
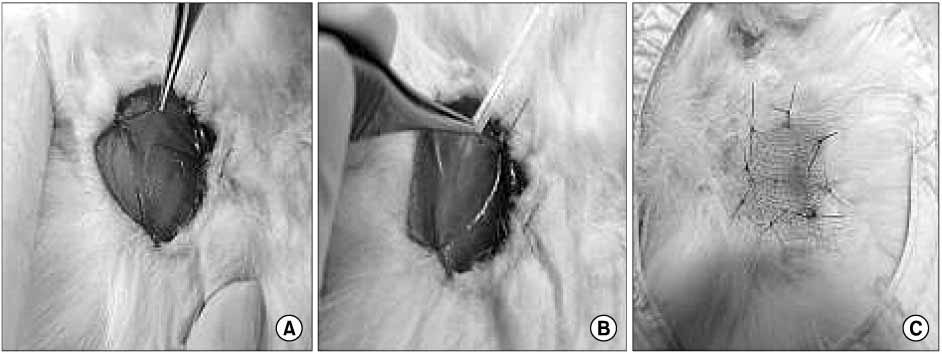
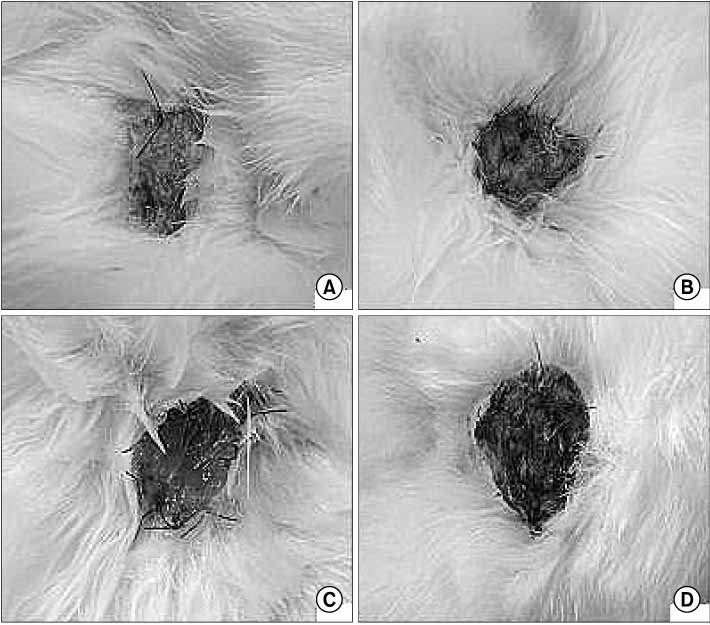
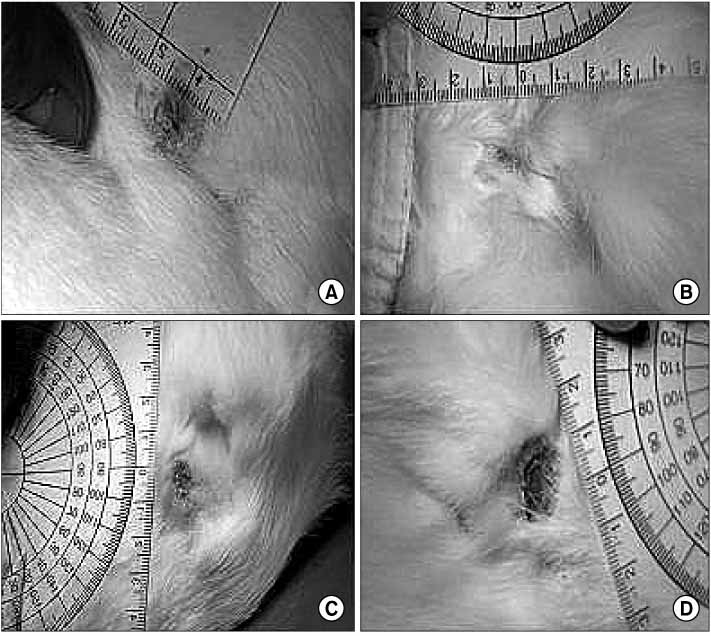
Defects (full-thickness) in rabbits were treated with HAM alone (group A), HAM injected with cultivated epidermal cells (group B), HAM injected with cultivated MSCs (group C), or Vaseline gauze (group D). Tissue granulation, regeneration, re-epithelization and healing time were measured. Defects and healed area were calculated 2 weeks after surgery.
RESULTS
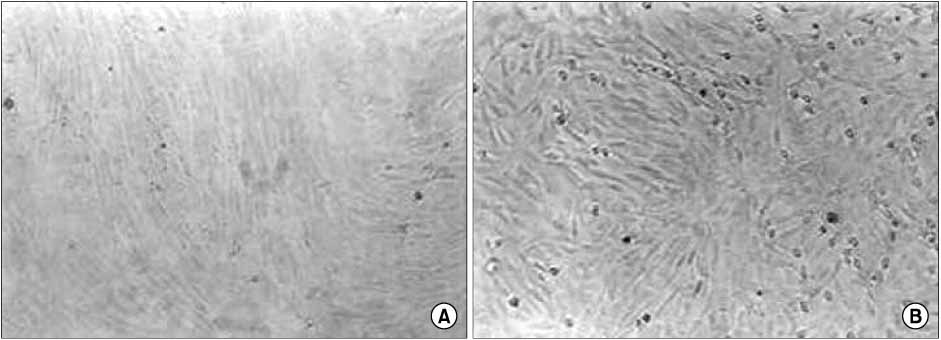
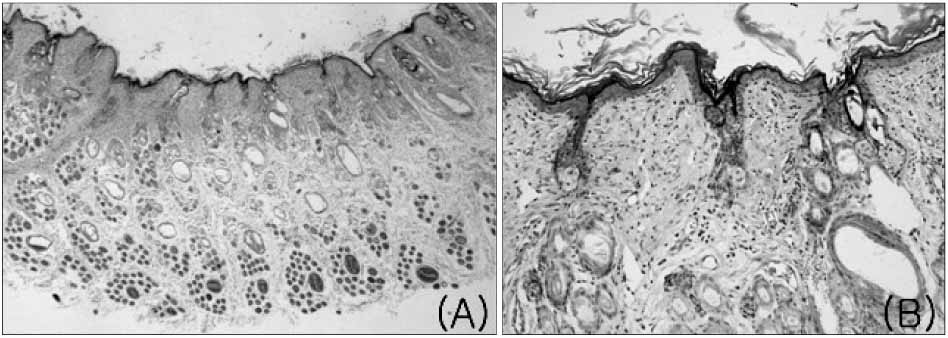
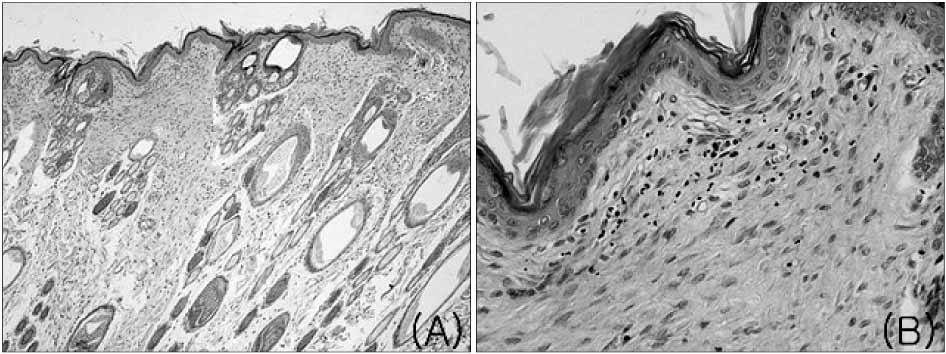
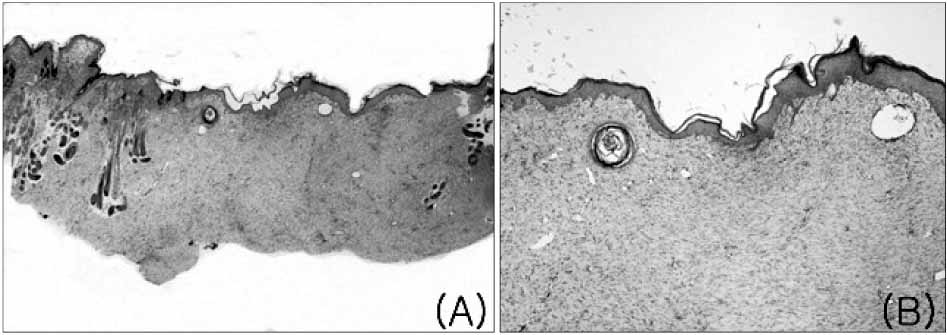
The mean healing area was 67.5%, 81.7%, 83.2% and 49.5% in each group, with all treatment groups significantly different than group D (p<0.01), and groups B and C compared higher than group A (p<0.05). The healing time of groups A, B, and C was 5.7 to 6.4 days faster than that of group D (p<0.01). Histologic analysis showed that the new epidermis covered nearly the whole wound surface in group B and C, and contained granulated tissue with fibroblasts, capillaries, and collagen.
CONCLUSION
HAM grafts injected with cultivated epidermal cells or MSCs promoted healing of skin defects.
Keyword
MeSH Terms
Figure
Reference
-
1. Akle CA, Adinofi M, Welsh KI, Leibowitz S, McColl I. Immunogenicity of human amniotic epithelial cells after transplantation into volunteers. Lancet. 1981. 7:1003–1005.
Article2. Bennett JP, Matthews R, Faulk WP. Treatment of chronic ulceration of the legs with human amnion. Lancet. 1980. 31:1153–1156.
Article3. Buultmann S, You L, Spandau U. Amniotic membrane down-regulate chemokine expression in human keratocytes. Invest Ophthalmol Vis Sci. 1999. 40:3044–3060.4. Caplan AI. Mesenchymal stem cells. J Orthop Res. 1991. 9:641–650.
Article5. Clark RA. Cutaneous tissue repair: basic biologic consideration. J Am Acad Dermatol. 1985. 13:701–725.6. Cunningham FG, Gant NF, Leveno KJ, et al. Williams obstetrics. 2001. 21th ed. New York: McGraw-Hill;101–105.7. Egan TJ, O'Driscoll J, Thaker DR. Human amnion in the management of chronic ulceration of the lower limb: a clinicopathologic study. Angiology. 1983. 34:197–203.
Article8. Faulk WP, Matthews R, Stevens PJ, Bennett JP, Burgos H, Hsi BL. Human amnion as an adjunct in wound healing. Lancet. 1980. 31:1156–1158.
Article9. Jaiswal N, Haynesworth SE, Caplan AI, Bruder SP. Osteogenic differentiation of purified, culture-expanded human mesenchymal stem cells in vitro. J Cell Biochem. 1997. 64:295–312.
Article10. Jeung WJ, Ha SJ, Park WC, Yoo KW. The effects of amniotic membrane for prevention of adhesion in strabismus surgery in rabbits. J Korean Opthalmol Soc. 2002. 43:402–410.11. Kim HS, Song KH, Park WC, Kim KH. Effectiveness of amniotic membrane patch in the treatment of chronic ulcers. Korean J Derma. 2005. 43:29–36.12. Kim JC, Tseng SC. The effects on inhibition of corneal neovascularization after human amniotic membrane transplantation in severely damaged rabbit corneas. Korean J Opthalmol. 1995. 9:32–46.
Article13. Kim JC, Tseng SC. Transplantation of preserved human amniotic membrane for surface reconstruction in severely damaged rabbit corneas. Cornea. 1995. 14:473–484.
Article14. Koizumi NJ, Inatomi TJ, Sotozona CJ, Fullwood NJ, Quantock AJ, Kinoshita S. Growth factor mRNA and protein in preserved human amniotic membrane. Curr Eye Res. 2000. 20:173–177.
Article15. Krishnan R, Coombs R, Wright N. Coombs R, Gristina A, Hungerford D, editors. Amniotic arthroplasty - a biological alternative. Joint replacement : state of art. 1990. St Louis: Mosby;31–37.16. Maximillian S. The grafting of preserved amniotic membrane to burned and ulcerated surfaces, substituting skin grafts. JAMA. 1913. 13:973–974.17. Ohgushi H, Goldberg VM, Caplan AI. Repair of bone defects with marrow cells and porous ceramic. Experiments in rats. Acta Orthop Scand. 1989. 60:334–339.
Article18. Reed BR, Clark RA. Cutaneous tissue repair: practical implications of current knowledge. II. J Am Acad Dermatol. 1985. 13:919–941.
Article19. Robson MC, Krizek TJ. The effect of human amniotic membranes on the bacterial population of infected rat burns. Ann J Surg. 1973. 177:144–149.
Article20. Ryan JM, Barry FP, Murphy JM, Mahon BP. Mesenchymal stem cells avoid allogenic rejection. J Inflamm (Lond). 2005. 2:8.21. Satoh H, Kishi K, Tanaka T, et al. Transplanted mesenchymal stem cells are effective for skin. regeneration in acute cutaneous wounds. Cell Transplant. 2004. 13:405–412.
Article22. Wakitini S, Goto T, Pineda SJ, et al. Mesenchymal cell-based repair of large, full-thickness defects of articular cartilage. J Bone Joint Surg Am. 1994. 76:579–592.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Progenitor Cells in Healing after Pterygium Excision
- Comparison of Human Bone Marrow Stromal Cells with Fibroblasts in Cell Proliferation and Collagen Synthesis
- Role of Angiogenic Stem Cells in Bone Regeneration
- Comparison with human amniotic membrane- and adipose tissue-derived mesenchymal stem cells
- The Effect of Amniotic Membrane Patch in Wound Healing of Skin Defect