J Dent Rehabil Appl Sci.
2015 Sep;31(3):212-220. 10.14368/jdras.2015.31.3.212.
Antifungal effect of electrolyzed hydrogen water on Candida albicans biofilm
- Affiliations
-
- 1Department of Oral Microbiology and Immunology, College of Dentistry, Dankook University, Cheonan, Republic of Korea. micro94@gmail.com
- 2Dental Research Institute, Dankook University, Cheonan, Republic of Korea.
- KMID: 2180022
- DOI: http://doi.org/10.14368/jdras.2015.31.3.212
Abstract
- PURPOSE
Candida albicans can cause mucosal disease in many vulnerable patients. Also they are associated with denture-related stomatitis. Electrolyzed water is generated by electric current passed via water using various metal electrodes and has antimicrobial activity. The aim of this study was to investigate antifungal activity of electrolyzed water on C. albicans biofilm.
MATERIALS AND METHODS
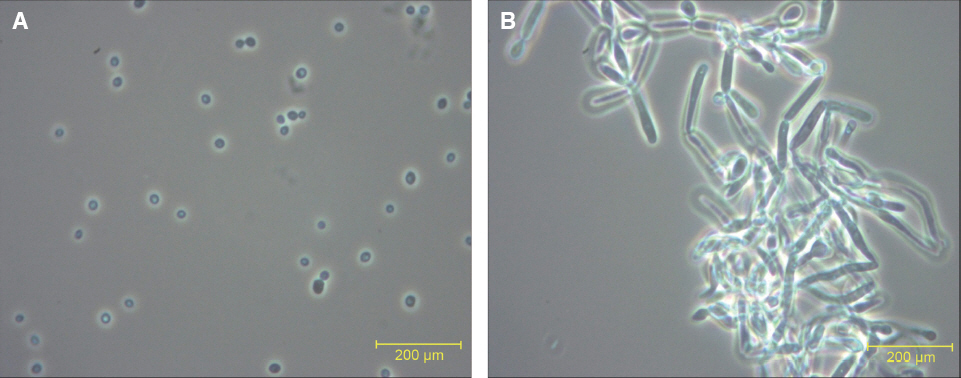
C. albicans was cultured by sabouraud dextrose broth and F-12 nutrient medium in aerobic and 5% CO2 condition to form blastoconidia (yeast) and hyphae type, respectively. For formation of C. albicans biofilm, C. albicans was cultivated on rough surface 6-well plate by using F-12 nutrient medium in CO2 incubator for 48 hr. After electrolyzing tap water using various metal electrodes, the blastoconidia and hyphal type of C. albicans were treated with electrolyzed water. C. albicans formed blastoconidia and hyphae type when they were cultured by sabouraud dextrose broth and F-12 nutrient medium, respectively.
RESULTS
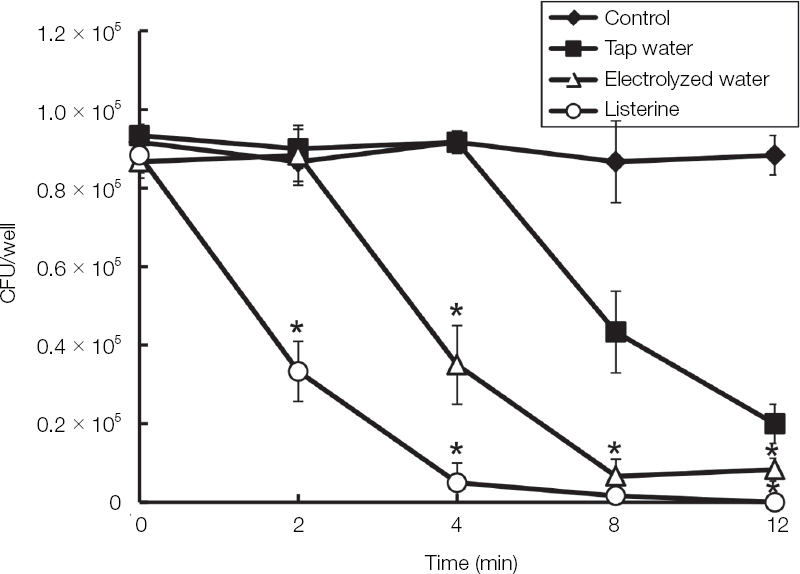
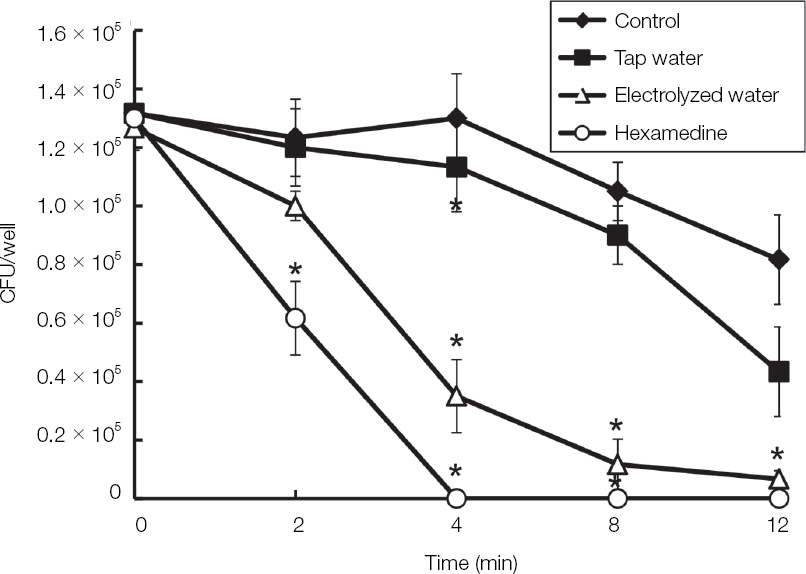
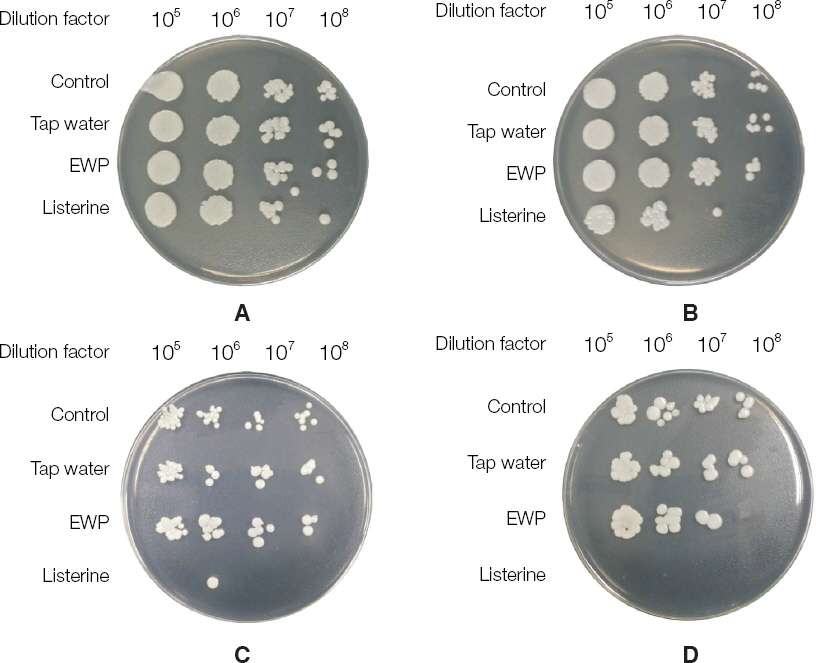
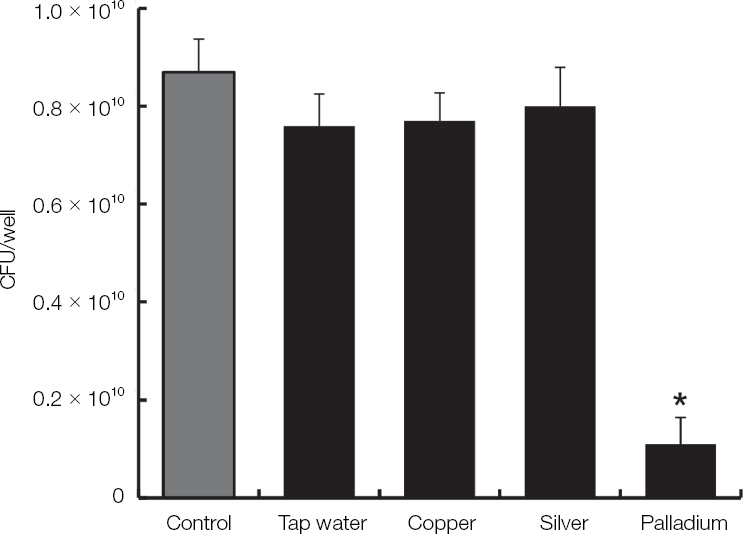
The electrolyzed water using palladium electrode (EWP) exhibited antifungal effect on blastoconidia of C. albicans. Also, the EWP significantly has antifungal activity against C. albicans biofilm and hyphae. In the electrolyzed water using various metal electrodes, only the EWP have antifungal activity.
CONCLUSION
The EWP may use a gargle solution and a soaking solution for prevention of oral candidiasis and denture-related stomatitis due to antifungal activity. (
MeSH Terms
Figure
Cited by 1 articles
-
Antifungal effects of hydrogen water on the growth of Candida albicans
Mi-Jung Park, Youn-Hee Choi, Keun-Bae Song, Eun-Kyong Kim
J Korean Acad Oral Health. 2019;43(4):173-177. doi: 10.11149/jkaoh.2019.43.4.173.
Reference
-
References
1. Coronado-Castellote L, Jimenez-Soriano Y. Clinical and microbiological diagnosis of oral candidiasis. J Clin Exp Dent. 2013; 5:e279–86. DOI: 10.4317/jced.51242. PMID: 24455095. PMCID: PMC3892259.2. Rupp S. Interactions of the fungal pathogen Candida albicans with the host. Future Microbiol. 2007; 2:141–51. DOI: 10.2217/17460913.2.2.141. PMID: 17661651.3. Sudbery P, Gow N, Berman J. The distinct morphogenic states of Candida albicans. Trends Microbiol. 2004; 12:317–24. DOI: 10.1016/j.tim.2004.05.008. PMID: 15223059.4. Sevilla MJ, Odds FC. Development of Candida albicans hyphae in different growth media--variations in growth rates, cell dimensions and timing of morphogenetic events. J Gen Microbiol. 1986; 132:30838. DOI: 10.1099/00221287-132-11-3083.5. Calderone RA, Fonzi WA. Virulence factors of Candida albicans. Trends Microbiol. 2001; 9:327–35. DOI: 10.1016/S0966-842X(01)02094-7.6. Bulad K, Taylor RL, Verran J, McCord JF. Colonization and penetration of denture soft lining materials by Candida albicans. Dent Mater. 2004; 20:16775. DOI: 10.1016/S0109-5641(03)00088-5.7. Huh JB, Lim Y, Youn HI, Chang BM, Lee JY, Shin SW. Effect of denture cleansers on Candida albicans biofilm formation over resilient liners. J Adv Prosthodont. 2014; 6:109–14. DOI: 10.4047/jap.2014.6.2.109. PMID: 24843395. PMCID: PMC4024554.8. Nikawa H, Hamada T, Yamashiro H, Kumagai H. A review of in vitro and in vivo methods to evaluate the efficacy of denture cleansers. Int J Prosthodont. 1999; 12:153–9. PMID: 10371918.9. Verran J, Maryan CJ. Retention of Candida albicans on acrylic resin and silicone of different surface topography. J Prosthet Dent. 1997; 77:535–9. DOI: 10.1016/S0022-3913(97)70148-3.10. Kitamura T, Todo H, Sugibayashi K. Effect of several electrolyzed waters on the skin permeation of lidocaine, benzoic acid, and isosorbide mononitrate. Drug Dev Ind Pharm. 2009; 35:145–53. DOI: 10.1080/03639040802005040. PMID: 19065312.11. Hricova D, Stephan R, Zweifel C. Electrolyzed water and its application in the food industry. J Food Prot. 2008; 71:1934–47. PMID: 18810883.12. Pangloli P, Hung YC. Efficacy of slightly acidic electrolyzed water in killing or reducing Escherichia coli O157: H7 on iceberg lettuce and tomatoes under simulated food service operation conditions. J Food Sci. 2011; 76:M361–6. DOI: 10.1111/j.1750-3841.2011.02219.x. PMID: 22417508.13. Ye J, Li Y, Hamasaki T, Nakamichi N, Komatsu T, Kashiwagi T, Teruya K, Nishikawa R, Kawahara T, Osada K, Toh K, Abe M, Tian H, Kabayama S, Otsubo K, Morisawa S, Katakura Y, Shirahata S. Inhibitory effect of electrolyzed reduced water on tumor angiogenesis. Biol Pharm Bull. 2008; 31:1926. DOI: 10.1248/bpb.31.19.14. Park SK, Park SK. Electrolyzed-reduced water increases resistance to oxidative stress, fertility, and lifespan via insulin/IGF-1-like signal in C. elegans. Biol Res. 2013; 46:147–52. DOI: 10.4067/S0716-97602013000200005. PMID: 23959012.15. Lee SH, Choi BK. Antibacterial effect of electrolyzed water on oral bacteria. J Microbiol. 2006; 44:417–22. PMID: 16953177.16. Komachiya M, Yamaguchi A, Hirai K, Kikuchi Y, Mizoue S, Takeda N, Ito M, Kato T, Ishihara K, Yamashita S, Akihiro K. Antiseptic effect of slightly acidic electrolyzed water on dental unit water systems. Bull Tokyo Dent Coll. 2014; 55:77–86. DOI: 10.2209/tdcpublication.55.77. PMID: 24965952.17. Mayer FL, Wilson D, Hube B. Candida albicans pathogenicity mechanisms. Virulence. 2013; 4:11928. DOI: 10.4161/viru.22913. PMID: 23302789. PMCID: PMC3654610.18. Goll G, Smith DE, Plein JB. The effect of denture cleansers on temporary soft liners. J Prosthet Dent. 1983; 50:466–72. DOI: 10.1016/0022-3913(83)90564-4.19. Sudbery PE. Growth of Candida albicans hyphae. Nat Rev Microbiol. 2011; 9:737–48. DOI: 10.1038/nrmicro2636. PMID: 21844880.20. Rautemaa R, Ramage G. Oral candidosis--clinical challenges of a biofilm disease. Crit Rev Microbiol. 2011; 37:328–36. DOI: 10.3109/1040841X.2011.585606. PMID: 21777047.21. Blankenship JR, Mitchell AP. How to build a biofilm: a fungal perspective. Curr Opin Microbiol. 2006; 9:588–94. DOI: 10.1016/j.mib.2006.10.003. PMID: 17055772.22. Tanaka H, Hirakata Y, Kaku M, Yoshida R, Takemura H, Mizukane R, Ishida K, Tomono K, Koga H, Kohno S, Kamihira S. Antimicrobial activity of superoxidized water. J Hosp Infect. 1996; 34:43–9. DOI: 10.1016/S0195-6701(96)90124-3.23. Nakagawara S, Goto T, Nara M, Ozawa Y, Hotta K, Arata Y. Spectroscopic characterization and the pH dependence of bactericidal activity of the aqueous chlorine solution. Analytical Sciences. 1998; 14:6918. DOI: 10.2116/analsci.14.691.24. Nakajima N, Nakano T, Harada F, Taniguchi H, Yokoyama I, Hirose J, Daikoku E, Sano K. Evaluation of disinfective potential of reactivated free chlorine in pooled tap water by electrolysis. J Microbiol Methods. 2004; 57:163–73. DOI: 10.1016/j.mimet.2003.12.011. PMID: 15063056.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Antifungal effects of hydrogen water on the growth of Candida albicans
- Preventive effects of shiitake mushroom extract on candida stomatitis
- Comparison of Antimicrobial Activity of Electrolyzed Water Using Various Electrodes against Biofilm of Oral Pathogens
- Candida albicans Biofilm Formation and Pathophysiology
- Studies on sensitivity and synergism of antifungal agents against candida albicans