J Gynecol Oncol.
2012 Apr;23(2):103-109. 10.3802/jgo.2012.23.2.103.
Changes in biologic markers of oxidative stress and plasma endotoxin levels in gynecologic cancer patients treated with pelvic radiotherapy: a pilot study
- Affiliations
-
- 1Department of Radiation Oncology, Seoul National University College of Medicine, Seoul, Korea.
- 2Cancer Research Institute, Seoul National University College of Medicine, Seoul, Korea.
- 3Institute of Radiation Medicine, Medical Research Center, Seoul National University, Seoul, Korea.
- 4Department of Radiation Oncology, Chung-Ang University Hospital, Seoul, Korea.
- 5Institute of Complementary and Integrative Medicine, Medical Research Center, Seoul National University, Seoul, Korea. drdemian@snu.ac.kr
- KMID: 2177583
- DOI: http://doi.org/10.3802/jgo.2012.23.2.103
Abstract
OBJECTIVE
We conducted a pilot study to evaluate the effects of pelvic radiotherapy on biologic markers of oxidative stress and plasma endotoxin levels, and to assess the relationship between the changes of such factors and radiotherapy-related complications.
METHODS
Twelve gynecologic cancer patients who were treated via pelvic radiotherapy with or without concurrent chemotherapy were enrolled in this study. Biologic markers of oxidative stress, such as glutathione (GSH) and oxidized glutathione (GSSG), as well as endotoxin levels, were measured weekly during treatment. Subjective symptoms were assessed using the Korean version of the EORTC QLQ-C30 at the baseline and on the 5th week of radiotherapy.
RESULTS
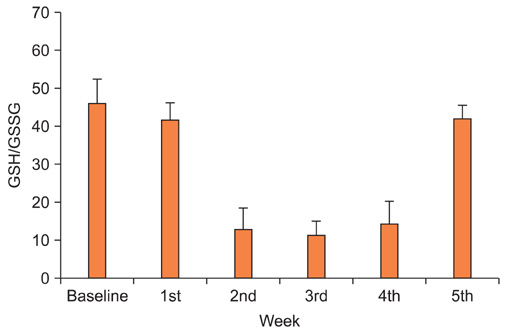
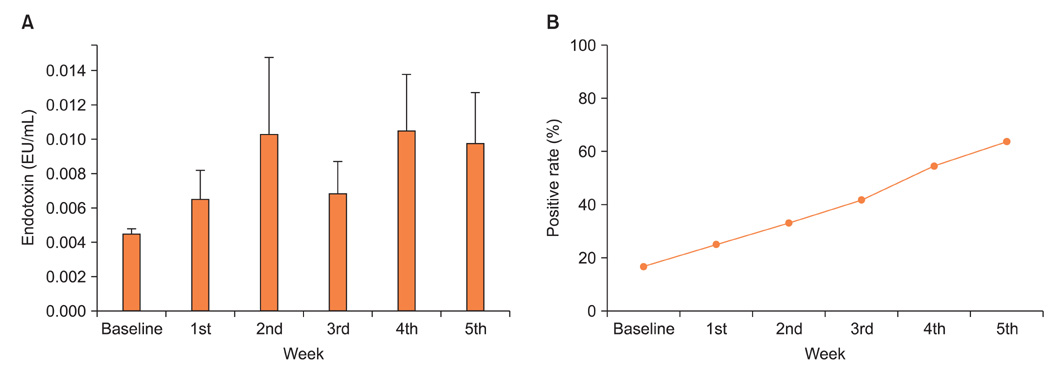
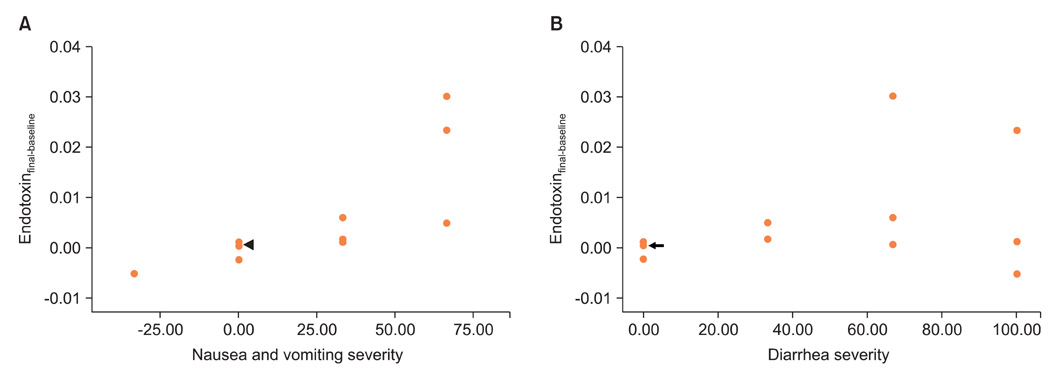
No changes were noted in the level of GSH in whole blood, but the GSH/GSSG ratio was reduced dramatically after the initiation of radiotherapy. The mean plasma endotoxin for all patients tended to increase and persisted during radiotherapy, and the number of patients who evidenced clinically significant endotoxin levels (defined as >0.005 EU/mL) also increased. Nausea/vomiting and diarrhea were significantly changed (p=0.019 and p<0.001, respectively). A significant relationship was noted to exist between the changes in the endotoxin level and nausea/vomiting (p=0.001). However, such symptoms did not correlate with the changes of oxidative stress markers.
CONCLUSION
Pelvic radiotherapy oxidized the GSH redox system and increased plasma endotoxin. Further investigations containing interventional and longitudinal studies will be required to assess the effects of the changes in oxidative stress markers and endotoxin on radiotherapy-related adverse events.
Keyword
MeSH Terms
Figure
Reference
-
1. Kim HJ, Ha SW, Wu HG. Treatment outcomes and prognostic factors in uterine cervical cancer patients treated with postoperative extended field radiation therapy. J Gynecol Oncol. 2009. 20:227–231.2. Kim YH, Chung HH, Kim JW, Park NH, Song YS, Kang SB. Prognostic significance of neutropenia during adjuvant concurrent chemoradiotherapy in early cervical cancer. J Gynecol Oncol. 2009. 20:146–150.3. Aaronson NK, Ahmedzai S, Bergman B, Bullinger M, Cull A, Duez NJ, et al. The European Organization for Research and Treatment of Cancer QLQ-C30: a quality-of-life instrument for use in international clinical trials in oncology. J Natl Cancer Inst. 1993. 85:365–376.4. Trask PC, Hsu MA, McQuellon R. Other paradigms: health-related quality of life as a measure in cancer treatment: its importance and relevance. Cancer J. 2009. 15:435–440.5. Andreyev J. Gastrointestinal complications of pelvic radiotherapy: are they of any importance? Gut. 2005. 54:1051–1054.6. Hurley JC. Endotoxemia: methods of detection and clinical correlates. Clin Microbiol Rev. 1995. 8:268–292.7. Maxwell A, Gaffin SL, Wells MT. Radiotherapy, endotoxaemia, and nausea. Lancet. 1986. 1:1148–1149.8. Richie JP Jr, Skowronski L, Abraham P, Leutzinger Y. Blood glutathione concentrations in a large-scale human study. Clin Chem. 1996. 42:64–70.9. Pastore A, Federici G, Bertini E, Piemonte F. Analysis of glutathione: implication in redox and detoxification. Clin Chim Acta. 2003. 333:19–39.10. Araujo AR, Saraiva ML, Lima JL. Determination of total and oxidized glutathione in human whole blood with a sequential injection analysis system. Talanta. 2008. 74:1511–1519.11. Kim JH, Kim HJ, Hong S, Wu HG, Ha SW. Post-hysterectomy radiotherapy in FIGO stage IB-IIB uterine cervical carcinoma. Gynecol Oncol. 2005. 96:407–414.12. Tietze F. Enzymic method for quantitative determination of nanogram amounts of total and oxidized glutathione: applications to mammalian blood and other tissues. Anal Biochem. 1969. 27:502–522.13. Yun YH, Park YS, Lee ES, Bang SM, Heo DS, Park SY, et al. Validation of the Korean version of the EORTC QLQ-C30. Qual Life Res. 2004. 13:863–868.14. Dalle-Donne I, Rossi R, Colombo R, Giustarini D, Milzani A. Biomarkers of oxidative damage in human disease. Clin Chem. 2006. 52:601–623.15. Benzie IF, Wachtel-Galor S. Vegetarian diets and public health: biomarker and redox connections. Antioxid Redox Signal. 2010. 13:1575–1591.16. Rossi R, Dalle-Donne I, Milzani A, Giustarini D. Oxidized forms of glutathione in peripheral blood as biomarkers of oxidative stress. Clin Chem. 2006. 52:1406–1414.17. Chen Y, Jungsuwadee P, Vore M, Butterfield DA, St Clair DK. Collateral damage in cancer chemotherapy: oxidative stress in nontargeted tissues. Mol Interv. 2007. 7:147–156.18. McGough C, Baldwin C, Frost G, Andreyev HJ. Role of nutritional intervention in patients treated with radiotherapy for pelvic malignancy. Br J Cancer. 2004. 90:2278–2287.19. Mossman KL, Henkin RI. Radiation-induced changes in taste acuity in cancer patients. Int J Radiat Oncol Biol Phys. 1978. 4:663–670.20. Afzal M, Afzal A, Jones A, Armstrong D. A rapid method for the quantification of GSH and GSSG in biological samples. Methods Mol Biol. 2002. 186:117–122.21. Ladas E, Kelly KM. The antioxidant debate. Explore (NY). 2010. 6:75–85.22. Pan H, Mukhopadhyay P, Rajesh M, Patel V, Mukhopadhyay B, Gao B, et al. Cannabidiol attenuates cisplatin-induced nephrotoxicity by decreasing oxidative/nitrosative stress, inflammation, and cell death. J Pharmacol Exp Ther. 2009. 328:708–714.23. Ladas EJ, Jacobson JS, Kennedy DD, Teel K, Fleischauer A, Kelly KM. Antioxidants and cancer therapy: a systematic review. J Clin Oncol. 2004. 22:517–528.24. DellaGioia N, Hannestad J. A critical review of human endotoxin administration as an experimental paradigm of depression. Neurosci Biobehav Rev. 2010. 34:130–143.25. Seal M, Naito Y, Barreto R, Lorenzetti A, Safran P, Marotta F. Experimental radiotherapy-induced enteritis: a probiotic interventional study. J Dig Dis. 2007. 8:143–147.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- The maternal change of malondialdehye levels in plasma and superoxide dismutase levels in plasma and erythrocyte as biologic markers of oxidative stress in pregnancy with preeclampsia
- Relationships of Plasma Homocysteine Concentration and Oxidative Stress Markers in Korean Collage Students
- The Correlation of Plasma Endotoxin and Components of Systemic Inflammatory Response Syndrome in Terminally Ill Cancer Patients
- Total radical TRapping antioxidant parameter, calculated
- Effect of the inhibition of PLA2 on the oxidative stress in the lungs of glutathione depleted rats given endotoxin intratracheally