Diabetes Metab J.
2015 Oct;39(5):439-443. 10.4093/dmj.2015.39.5.439.
Identification of Two Cases of Ciliopathy-Associated Diabetes and Their Mutation Analysis Using Whole Exome Sequencing
- Affiliations
-
- 1Department of Internal Medicine, Seoul National University Hospital, Seoul National University College of Medicine, Seoul, Korea. kspark@snu.ac.kr
- 2Department of Internal Medicine, Gangnam Severance Hospital, Yonsei University College of Medicine, Seoul, Korea.
- 3Severance Institute for Vascular and Metabolic Research, Yonsei University College of Medicine, Seoul, Korea.
- 4Department of Internal Medicine, Seoul National University College of Medicine, Seoul, Korea.
- 5Department of Molecular Medicine and Biopharmaceutical Sciences, Graduate School of Convergence Science and Technology, Seoul National University, Seoul, Korea.
- KMID: 2174018
- DOI: http://doi.org/10.4093/dmj.2015.39.5.439
Abstract
- BACKGROUND
Alstrom syndrome and Bardet-Biedl syndrome are autosomal recessively inherited ciliopathies with common characteristics of obesity, diabetes, and blindness. Alstrom syndrome is caused by a mutation in the ALMS1 gene, and Bardet-Biedl syndrome is caused by mutations in BBS1-16 genes. Herein we report genetically confirmed cases of Alstrom syndrome and Bardet-Biedl syndrome in Korea using whole exome sequencing.
METHODS
Exome capture was done using SureSelect Human All Exon Kit V4+UTRs (Agilent Technologies). HiSeq2000 system (Illumina) was used for massive parallel sequencing. Sanger sequencing was used for genotype confirmation and familial cosegregation analysis.
RESULTS
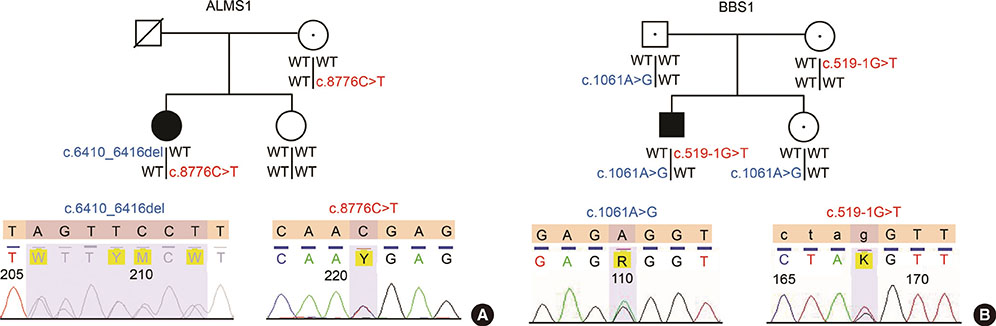
A 21-year old Korean woman was clinically diagnosed with Alstrom syndrome. She had diabetes, blindness, obesity, severe insulin resistance, and hearing loss. Whole exome sequencing revealed a nonsense mutation in exon 10 of ALMS1 (c.8776C>T, p.R2926X) and a seven base-pair deletion resulting in frameshift mutation in exon 8 (c.6410_6416del, p.2137_2139del). A 24-year-old Korean man had Bardet-Biedl syndrome with diabetes, blindness, obesity, and a history of polydactyly. Whole exome sequencing revealed a nonsynonymous mutation in exon 11 of the BBS1 gene (c.1061A>G, p.E354G) and mutation at the normal splicing recognition site of exon 7 of the BBS1 gene (c.519-1G>T).
CONCLUSION
We found novel compound heterozygous mutations of Alstrom syndrome and Bardet-Biedl syndrome using whole exome sequencing. The whole exome sequencing successfully identified novel genetic variants of ciliopathy-associated diabetes.
Keyword
MeSH Terms
Figure
Reference
-
1. Hildebrandt F, Benzing T, Katsanis N. Ciliopathies. N Engl J Med. 2011; 364:1533–1543.2. Lee JE, Gleeson JG. A systems-biology approach to understanding the ciliopathy disorders. Genome Med. 2011; 3:59.3. Girard D, Petrovsky N. Alstrom syndrome: insights into the pathogenesis of metabolic disorders. Nat Rev Endocrinol. 2011; 7:77–88.4. Forsythe E, Beales PL. Bardet-Biedl syndrome. Eur J Hum Genet. 2013; 21:8–13.5. Marshall JD, Maffei P, Beck S, Barrett TG, Paisey R, Naggert JK. Clinical utility gene card for: Alstrom syndrome update 2013. Eur J Hum Genet. 2013; 21.6. Slavotinek A, Beales P. Clinical utility gene card for: Bardet-Biedl syndrome. Eur J Hum Genet. 2011; 19.7. Aliferis K, Helle S, Gyapay G, Duchatelet S, Stoetzel C, Mandel JL, Dollfus H. Differentiating Alstrom from Bardet-Biedl syndrome (BBS) using systematic ciliopathy genes sequencing. Ophthalmic Genet. 2012; 33:18–22.8. Chen J, Smaoui N, Hammer MB, Jiao X, Riazuddin SA, Harper S, Katsanis N, Riazuddin S, Chaabouni H, Berson EL, Hejtmancik JF. Molecular analysis of Bardet-Biedl syndrome families: report of 21 novel mutations in 10 genes. Invest Ophthalmol Vis Sci. 2011; 52:5317–5324.9. Goh G, Choi M. Application of whole exome sequencing to identify disease-causing variants in inherited human diseases. Genomics Inform. 2012; 10:214–219.10. Li H, Durbin R. Fast and accurate short read alignment with Burrows-Wheeler transform. Bioinformatics. 2009; 25:1754–1760.11. McKenna A, Hanna M, Banks E, Sivachenko A, Cibulskis K, Kernytsky A, Garimella K, Altshuler D, Gabriel S, Daly M, DePristo MA. The Genome Analysis Toolkit: a MapReduce framework for analyzing next-generation DNA sequencing data. Genome Res. 2010; 20:1297–1303.12. Adzhubei IA, Schmidt S, Peshkin L, Ramensky VE, Gerasimova A, Bork P, Kondrashov AS, Sunyaev SR. A method and server for predicting damaging missense mutations. Nat Methods. 2010; 7:248–249.13. Adzhubei I, Jordan DM, Sunyaev SR. Predicting functional effect of human missense mutations using PolyPhen-2. Curr Protoc Hum Genet. 2013; Chapter 7:Unit7.20.14. Marshall JD, Hinman EG, Collin GB, Beck S, Cerqueira R, Maffei P, Milan G, Zhang W, Wilson DI, Hearn T, Tavares P, Vettor R, Veronese C, Martin M, So WV, Nishina PM, Naggert JK. Spectrum of ALMS1 variants and evaluation of genotype-phenotype correlations in Alstrom syndrome. Hum Mutat. 2007; 28:1114–1123.15. Yoon KH, Son HY, Kang SK, Choi YH, Cha BY, Cho HS, Kim KB, Kang JH, Choo YM, Bae SS. A case of Alstrom syndrome. J Korean Soc Endocrinol. 1998; 13:501–508.16. Daniels AB, Sandberg MA, Chen J, Weigel-DiFranco C, Fielding Hejtmancic J, Berson EL. Genotype-phenotype correlations in Bardet-Biedl syndrome. Arch Ophthalmol. 2012; 130:901–907.17. Yoon SC, Lee HJ, Ko JM, Kang HG, Cheong HI, Yu HG, Kim JH. Two siblings with Bardet-Biedl syndrome caused by mutations in BBS10: the first case identified in Korea. J Genet Med. 2014; 11:31–35.18. Collins FS, Hamburg MA. First FDA authorization for next-generation sequencer. N Engl J Med. 2013; 369:2369–2371.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Exome Sequencing in Mendelian Disorders
- Identification of a De Novo Heterozygous Missense FLNB Mutation in Lethal Atelosteogenesis Type I by Exome Sequencing
- Update on Monogenic Diabetes in Korea
- Whole Exome Sequencing of a Patient with Duchenne Muscular Dystrophy
- Congenital Orbital Fibrosis: Molecular Genetic Analysis by Whole-Exome and Mitochondrial Genome Sequencing