Chonnam Med J.
2015 Apr;51(1):1-7. 10.4068/cmj.2015.51.1.1.
Dendritic Cell-Based Cancer Immunotherapy against Multiple Myeloma: From Bench to Clinic
- Affiliations
-
- 1Research Center for Cancer Immunotherapy, Chonnam National University Hwasun Hospital, Hwasun, Korea. drjejung@chonnam.ac.kr
- 2Department of Hematology-Oncology, Chonnam National University Hwasun Hospital, Hwasun, Korea.
- 3Vaxcell-Bio Therapeutics, Hwasun, Korea.
- KMID: 2172141
- DOI: http://doi.org/10.4068/cmj.2015.51.1.1
Abstract
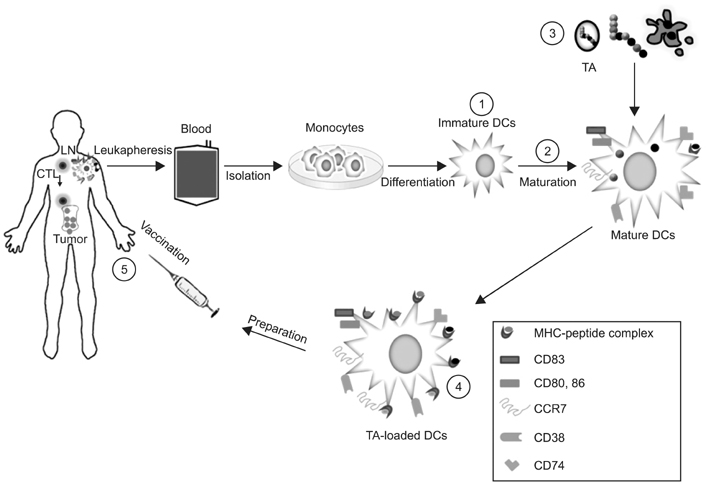
- Although the introduction of stem cell transplantation and novel agents has improved survival, multiple myeloma (MM) is still difficult to cure. Alternative approaches are clearly needed to prolong the survival of patients with MM. Dendritic cell (DC) therapy is a very promising tool immunologically in MM. We developed a method to generate potent DCs with increased Th1 polarization and migration ability for inducing strong myeloma-specific cytotoxic T lymphocytes. In this review, we discuss how the efficacy of cancer immunotherapy using DCs can be improved in MM.
Keyword
MeSH Terms
Figure
Cited by 1 articles
-
Cellular immunotherapy as a beacon of hope for hematological malignancies
Hyun-Ju Lee, Sang-Ki Kim, Duck Cho, Je-Jung Lee
Blood Res. 2015;50(3):126-128. doi: 10.5045/br.2015.50.3.126.
Reference
-
1. Mellman I, Coukos G, Dranoff G. Cancer immunotherapy comes of age. Nature. 2011; 480:480–489.
Article2. Banchereau J, Steinman RM. Dendritic cells and the control of immunity. Nature. 1998; 392:245–252.
Article3. Palucka K, Banchereau J. Dendritic-cell-based therapeutic cancer vaccines. Immunity. 2013; 39:38–48.
Article4. Tel J, Anguille S, Waterborg CE, Smits EL, Figdor CG, de Vries IJ. Tumoricidal activity of human dendritic cells. Trends Immunol. 2014; 35:38–46.
Article5. Brimnes MK, Svane IM, Johnsen HE. Impaired functionality and phenotypic profile of dendritic cells from patients with multiple myeloma. Clin Exp Immunol. 2006; 144:76–84.
Article6. Lee HJ, Choi NR, Vo MC, Hoang MD, Lee YK, Lee JJ. Generation of multiple peptide cocktail-pulsed dendritic cells as a cancer vaccine. Methods Mol Biol. 2014; 1139:17–26.
Article7. Tel J, Aarntzen EH, Baba T, Schreibelt G, Schulte BM, Benitez-Ribas D, et al. Natural human plasmacytoid dendritic cells induce antigen-specific T-cell responses in melanoma patients. Cancer Res. 2013; 73:1063–1075.
Article8. Palumbo A, Anderson K. Multiple myeloma. N Engl J Med. 2011; 364:1046–1060.
Article9. Pratt G, Goodyear O, Moss P. Immunodeficiency and immunotherapy in multiple myeloma. Br J Haematol. 2007; 138:563–579.
Article10. Li Y, Bendandi M, Deng Y, Dunbar C, Munshi N, Jagannath S, et al. Tumor-specific recognition of human myeloma cells by idiotype-induced CD8(+) T cells. Blood. 2000; 96:2828–2833.
Article11. Wen YJ, Barlogie B, Yi Q. Idiotype-specific cytotoxic T lymphocytes in multiple myeloma: evidence for their capacity to lyse autologous primary tumor cells. Blood. 2001; 97:1750–1755.
Article12. Hong S, Qian J, Yang J, Li H, Kwak LW, Yi Q. Roles of idiotype-specific t cells in myeloma cell growth and survival: Th1 and CTL cells are tumoricidal while Th2 cells promote tumor growth. Cancer Res. 2008; 68:8456–8464.
Article13. Nguyen-Pham TN, Lee YK, Lee HJ, Kim MH, Yang DH, Kim HJ, et al. Cellular immunotherapy using dendritic cells against multiple myeloma. Korean J Hematol. 2012; 47:17–27.
Article14. Takahashi T, Makiguchi Y, Hinoda Y, Kakiuchi H, Nakagawa N, Imai K, et al. Expression of MUC1 on myeloma cells and induction of HLA-unrestricted CTL against MUC1 from a multiple myeloma patient. J Immunol. 1994; 153:2102–2109.15. Pellat-Deceunynck C, Mellerin MP, Labarrière N, Jego G, Moreau-Aubry A, Harousseau JL, et al. The cancer germ-line genes MAGE-1, MAGE-3 and PRAME are commonly expressed by human myeloma cells. Eur J Immunol. 2000; 30:803–809.
Article16. Lim SH, Wang Z, Chiriva-Internati M, Xue Y. Sperm protein 17 is a novel cancer-testis antigen in multiple myeloma. Blood. 2001; 97:1508–1510.
Article17. Qian J, Wang S, Yang J, Xie J, Lin P, Freeman ME 3rd, et al. Targeting heat shock proteins for immunotherapy in multiple myeloma: generation of myeloma-specific CTLs using dendritic cells pulsed with tumor-derived gp96. Clin Cancer Res. 2005; 11:8808–8815.
Article18. Qian J, Xie J, Hong S, Yang J, Zhang L, Han X, et al. Dickkopf-1 (DKK1) is a widely expressed and potent tumor-associated antigen in multiple myeloma. Blood. 2007; 110:1587–1594.
Article19. Atanackovic D, Arfsten J, Cao Y, Gnjatic S, Schnieders F, Bartels K, et al. Cancer-testis antigens are commonly expressed in multiple myeloma and induce systemic immunity following allogeneic stem cell transplantation. Blood. 2007; 109:1103–1112.
Article20. Anderson LD Jr, Cook DR, Yamamoto TN, Berger C, Maloney DG, Riddell SR. Identification of MAGE-C1 (CT-7) epitopes for T-cell therapy of multiple myeloma. Cancer Immunol Immunother. 2011; 60:985–997.
Article21. Bae J, Tai YT, Anderson KC, Munshi NC. Novel epitope evoking CD138 antigen-specific cytotoxic T lymphocytes targeting multiple myeloma and other plasma cell disorders. Br J Haematol. 2011; 155:349–361.
Article22. Bae J, Smith R, Daley J, Mimura N, Tai YT, Anderson KC, et al. Myeloma-specific multiple peptides able to generate cytotoxic T lymphocytes: a potential therapeutic application in multiple myeloma and other plasma cell disorders. Clin Cancer Res. 2012; 18:4850–4860.
Article23. Bae J, Song W, Smith R, Daley J, Tai YT, Anderson KC, et al. A novel immunogenic CS1-specific peptide inducing antigen-specific cytotoxic T lymphocytes targeting multiple myeloma. Br J Haematol. 2012; 157:687–701.
Article24. Schmidt SM, Schag K, Müller MR, Weck MM, Appel S, Kanz L, et al. Survivin is a shared tumor-associated antigen expressed in a broad variety of malignancies and recognized by specific cytotoxic T cells. Blood. 2003; 102:571–576.
Article25. Ocadlikova D, Kryukov F, Mollova K, Kovarova L, Buresdova I, Matejkova E, et al. Generation of myeloma-specific T cells using dendritic cells loaded with MUC1- and hTERT-drived nonapeptides or myeloma cell apoptotic bodies. Neoplasma. 2010; 57:455–464.
Article26. Goodyear OC, Pearce H, Pratt G, Moss P. Dominant responses with conservation of T-cell receptor usage in the CD8+ T-cell recognition of a cancer testis antigen peptide presented through HLA-Cw7 in patients with multiple myeloma. Cancer Immunol Immunother. 2011; 60:1751–1761.
Article27. Batchu RB, Moreno AM, Szmania SM, Bennett G, Spagnoli GC, Ponnazhagan S, et al. Protein transduction of dendritic cells for NY-ESO-1-based immunotherapy of myeloma. Cancer Res. 2005; 65:10041–10049.
Article28. Hobo W, Strobbe L, Maas F, Fredrix H, Greupink-Draaisma A, Esendam B, et al. Immunogenicity of dendritic cells pulsed with MAGE3, Survivin and B-cell maturation antigen mRNA for vaccination of multiple myeloma patients. Cancer Immunol Immunother. 2013; 62:1381–1392.
Article29. Lee JJ, Choi BH, Kang HK, Park MS, Park JS, Kim SK, et al. Induction of multiple myeloma-specific cytotoxic T lymphocyte stimulation by dendritic cell pulsing with purified and optimized myeloma cell lysates. Leuk Lymphoma. 2007; 48:2022–2031.
Article30. Qian J, Hong S, Wang S, Zhang L, Sun L, Wang M, et al. Myeloma cell line-derived, pooled heat shock proteins as a universal vaccine for immunotherapy of multiple myeloma. Blood. 2009; 114:3880–3889.
Article31. Yang DH, Kim MH, Lee YK, Hong CY, Lee HJ, Nguyen-Pham TN, et al. Successful cross-presentation of allogeneic myeloma cells by autologous alpha-type 1-polarized dendritic cells as an effective tumor antigen in myeloma patients with matched monoclonal immunoglobulins. Ann Hematol. 2011; 90:1419–1426.
Article32. Jung SH, Lee YK, Lee HJ, Choi NR, Vo MC, Hoang MD, et al. Dendritic cells loaded with myeloma cells pretreated with a combination of JSI-124 and bortezomib generate potent myeloma-specific cytotoxic T lymphocytes in vitro. Exp Hematol. 2014; 42:274–281.
Article33. Milazzo C, Reichardt VL, Müller MR, Grünebach F, Brossart P. Induction of myeloma-specific cytotoxic T cells using dendritic cells transfected with tumor-derived RNA. Blood. 2003; 101:977–982.
Article34. Rosenblatt J, Vasir B, Uhl L, Blotta S, Macnamara C, Somaiya P, et al. Vaccination with dendritic cell/tumor fusion cells results in cellular and humoral antitumor immune responses in patients with multiple myeloma. Blood. 2011; 117:393–402.
Article35. Wen YJ, Min R, Tricot G, Barlogie B, Yi Q. Tumor lysate-specific cytotoxic T lymphocytes in multiple myeloma: promising effector cells for immunotherapy. Blood. 2002; 99:3280–3285.
Article36. Singh-Jasuja H, Scherer HU, Hilf N, Arnold-Schild D, Rammensee HG, Toes RE, et al. The heat shock protein gp96 induces maturation of dendritic cells and down-regulation of its receptor. Eur J Immunol. 2000; 30:2211–2215.
Article37. Nestle FO, Alijagic S, Gilliet M, Sun Y, Grabbe S, Dummer R, et al. Vaccination of melanoma patients with peptide- or tumor lysate-pulsed dendritic cells. Nat Med. 1998; 4:328–332.
Article38. de Vries IJ, Lesterhuis WJ, Scharenborg NM, Engelen LP, Ruiter DJ, Gerritsen MJ, et al. Maturation of dendritic cells is a prerequisite for inducing immune responses in advanced melanoma patients. Clin Cancer Res. 2003; 9:5091–5100.39. Jonuleit H, Kühn U, Müller G, Steinbrink K, Paragnik L, Schmitt E, et al. Pro-inflammatory cytokines and prostaglandins induce maturation of potent immunostimulatory dendritic cells under fetal calf serum-free conditions. Eur J Immunol. 1997; 27:3135–3142.
Article40. Hansen M, Hjortø GM, Donia M, Met Ö, Larsen NB, Andersen MH, et al. Comparison of clinical grade type 1 polarized and standard matured dendritic cells for cancer immunotherapy. Vaccine. 2013; 31:639–646.
Article41. Lee JJ, Foon KA, Mailliard RB, Muthuswamy R, Kalinski P. Type 1-polarized dendritic cells loaded with autologous tumor are a potent immunogen against chronic lymphocytic leukemia. J Leukoc Biol. 2008; 84:319–325.
Article42. Yang DH, Kim MH, Hong CY, Lee YK, Jin CJ, Pham TN, et al. Alpha-type 1-polarized dendritic cells loaded with apoptotic allogeneic myeloma cell line induce strong CTL responses against autologous myeloma cells. Ann Hematol. 2010; 89:795–801.
Article43. Park MH, Yang DH, Kim MH, Jang JH, Jang YY, Lee YK, et al. Alpha-type 1 polarized dendritic cells loaded with apoptotic allogeneic breast cancer cells can induce potent cytotoxic t lymphocytes against breast cancer. Cancer Res Treat. 2011; 43:56–66.
Article44. Lee HJ, Hong CY, Kim MH, Lee YK, Nguyen-Pham TN, Park BC, et al. vitro induction of anterior gradient-2-specific cytotoxic T lymphocytes by dendritic cells transduced with recombinant adenoviruses as a potential therapy for colorectal cancer. Exp Mol Med. 2012; 44:60–67.
Article45. Hwang EC, Lim MS, Im CM, Kwon DD, Lee HJ, Nguyen-Pham TN, et al. Generation of potent cytotoxic T lymphocytes against castration-resistant prostate cancer cells by dendritic cells loaded with dying allogeneic prostate cancer cells. Scand J Immunol. 2013; 77:117–124.
Article46. Bae WK, Umeyama A, Chung IJ, Lee JJ, Takei M. Uncarinic acid C plus IFN-γ generates monocyte-derived dendritic cells and induces a potent Th1 polarization with capacity to migrate. Cell Immunol. 2010; 266:104–110.
Article47. Jung TY, Pham TN, Umeyama A, Shoji N, Hashimoto T, Lee JJ, et al. Ursolic acid isolated from Uncaria rhynchophylla activates human dendritic cells via TLR2 and/or TLR4 and induces the production of IFN-gamma by CD4+ naïve T cells. Eur J Pharmacol. 2010; 643:297–303.
Article48. Takei M, Umeyama A, Lee JJ, Shoji N, Hashimoto T. Cryptomerione induces Th1 cell polarization via influencing IL-10 production by cholera toxin-primed dendritic cells. Eur J Pharmacol. 2010; 628:233–239.
Article49. Hong CY, Kim SY, Lee HJ, Lee SE, Lim SC, Rhee JH, et al. A bacterial flagellin in combination with proinflammatory cytokines activates human monocyte-derived dendritic cells to generate cytotoxic T lymphocytes having increased homing signals to cancer. J Immunother. 2014; 37:16–25.
Article50. Kim KS, Pham TN, Jin CJ, Umeyama A, Shoji N, Hashimoto T, et al. Uncarinic Acid C Isolated from Uncaria rhynchophylla Induces Differentiation of Th1-Promoting Dendritic Cells Through TLR4 Signaling. Biomark Insights. 2011; 6:27–38.51. Nguyen-Pham TN, Lim MS, Nguyen TA, Lee YK, Jin CJ, Lee HJ, et al. Type I and II interferons enhance dendritic cell maturation and migration capacity by regulating CD38 and CD74 that have synergistic effects with TLR agonists. Cell Mol Immunol. 2011; 8:341–347.
Article52. Kalinski P, Nakamura Y, Watchmaker P, Giermasz A, Muthuswamy R, Mailliard RB. Helper roles of NK and CD8+ T cells in the induction of tumor immunity. Polarized dendritic cells as cancer vaccines. Immunol Res. 2006; 36:137–146.
Article53. Pham TN, Hong CY, Min JJ, Rhee JH, Nguyen TA, Park BC, et al. Enhancement of antitumor effect using dendritic cells activated with natural killer cells in the presence of Toll-like receptor agonist. Exp Mol Med. 2010; 42:407–419.
Article54. Nguyen-Pham TN, Yang DH, Nguyen TA, Lim MS, Hong CY, Kim MH, et al. Optimal culture conditions for the generation of natural killer cell-induced dendritic cells for cancer immunotherapy. Cell Mol Immunol. 2012; 9:45–53.
Article55. Mailliard RB, Son YI, Redlinger R, Coates PT, Giermasz A, Morel PA, et al. Dendritic cells mediate NK cell help for Th1 and CTL responses: two-signal requirement for the induction of NK cell helper function. J Immunol. 2003; 171:2366–2373.
Article56. Mantia-Smaldone GM, Chu CS. A review of dendritic cell therapy for cancer: progress and challenges. BioDrugs. 2013; 27:453–468.
Article57. Yang DH, Park JS, Jin CJ, Kang HK, Nam JH, Rhee JH, et al. The dysfunction and abnormal signaling pathway of dendritic cells loaded by tumor antigen can be overcome by neutralizing VEGF in multiple myeloma. Leuk Res. 2009; 33:665–670.
Article58. Spisek R, Charalambous A, Mazumder A, Vesole DH, Jagannath S, Dhodapkar MV. Bortezomib enhances dendritic cell (DC)-mediated induction of immunity to human myeloma via exposure of cell surface heat shock protein 90 on dying tumor cells: therapeutic implications. Blood. 2007; 109:4839–4845.
Article59. Luptakova K, Rosenblatt J, Glotzbecker B, Mills H, Stroopinsky D, Kufe T, et al. Lenalidomide enhances anti-myeloma cellular immunity. Cancer Immunol Immunother. 2013; 62:39–49.
Article60. Neuber B, Herth I, Tolliver C, Schoenland S, Hegenbart U, Hose D, et al. Lenalidomide enhances antigen-specific activity and decreases CD45RA expression of T cells from patients with multiple myeloma. J Immunol. 2011; 187:1047–1056.
Article61. Henry JY, Labarthe MC, Meyer B, Dasgupta P, Dalgleish AG, Galustian C. Enhanced cross-priming of naive CD8+ T cells by dendritic cells treated by the IMiDs® immunomodulatory compounds lenalidomide and pomalidomide. Immunology. 2013; 139:377–385.
Article62. De Keersmaecker B, Fostier K, Corthals J, Wilgenhof S, Heirman C, Aerts JL, et al. Immunomodulatory drugs improve the immune environment for dendritic cell-based immunotherapy in multiple myeloma patients after autologous stem cell transplantation. Cancer Immunol Immunother. 2014; 63:1023–1036.
Article63. Fu Q, Wu Y, Yan F, Wang N, Wang W, Cao X, et al. Efficient induction of a Her2-specific anti-tumor response by dendritic cells pulsed with a Hsp70L1-Her2(341-456) fusion protein. Cell Mol Immunol. 2011; 8:424–432.
Article64. Kim BR, Yang EK, Kim DY, Kim SH, Moon DC, Lee JH, et al. Generation of anti-tumour immune response using dendritic cells pulsed with carbonic anhydrase IX-Acinetobacter baumannii outer membrane protein A fusion proteins against renal cell carcinoma. Clin Exp Immunol. 2012; 167:73–83.
Article65. Tanaka Y, Dowdy SF, Linehan DC, Eberlein TJ, Goedegebuure PS. Induction of antigen-specific CTL by recombinant HIV trans-activating fusion protein-pulsed human monocyte-derived dendritic cells. J Immunol. 2003; 170:1291–1298.
Article66. White KL, Rades T, Furneaux RH, Tyler PC, Hook S. Mannosylated liposomes as antigen delivery vehicles for targeting to dendritic cells. J Pharm Pharmacol. 2006; 58:729–737.
Article67. Zhao L, Seth A, Wibowo N, Zhao CX, Mitter N, Yu C, et al. Nanoparticle vaccines. Vaccine. 2014; 32:327–337.
Article68. Boudreau JE, Bonehill A, Thielemans K, Wan Y. Engineering dendritic cells to enhance cancer immunotherapy. Mol Ther. 2011; 19:841–853.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Cellular immunotherapy using dendritic cells against multiple myeloma
- Cellular immunotherapy in multiple myeloma
- Distinct features of dendritic cell-based immunotherapy as cancer vaccines
- Current Approaches in Development of Immunotherapeutic Vaccines for Breast Cancer
- Dendritic Cell-Based Cancer Immunotherapy