Anat Cell Biol.
2014 Mar;47(1):55-65. 10.5115/acb.2014.47.1.55.
Topohistology of sympathetic and parasympathetic nerve fibers in branches of the pelvic plexus: an immunohistochemical study using donated elderly cadavers
- Affiliations
-
- 1Department of Urology, Kobe University Graduate School of Medicine, Kobe, Japan. hinata@med.kobe-u.ac.jp
- 2Department of Urology, Hiroshima University School of Medicine, Hiroshima, Japan.
- 3Division of Gynecology, Ishikawa Prefectural Central Hospital, Kanazawa, Japan.
- 4Division of Internal Medicine, Iwamizawa Kojin-kai Hospital, Iwamizawa, Japan.
- 5Department of Anatomy, Tokyo Dental College, Tokyo, Japan.
- KMID: 2168857
- DOI: http://doi.org/10.5115/acb.2014.47.1.55
Abstract
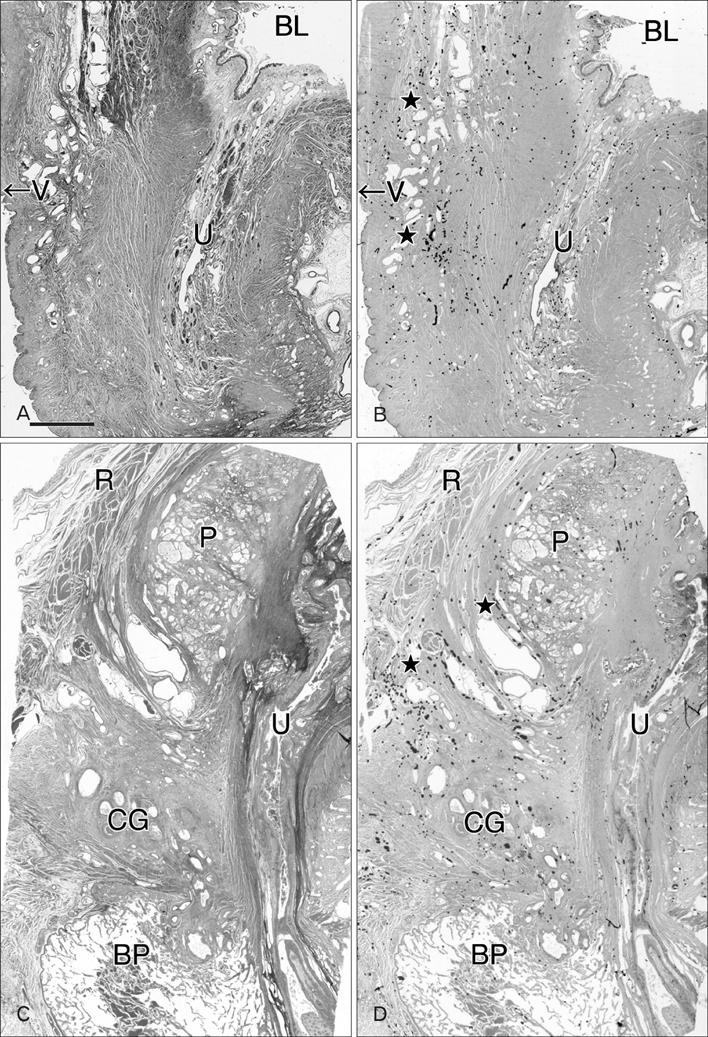
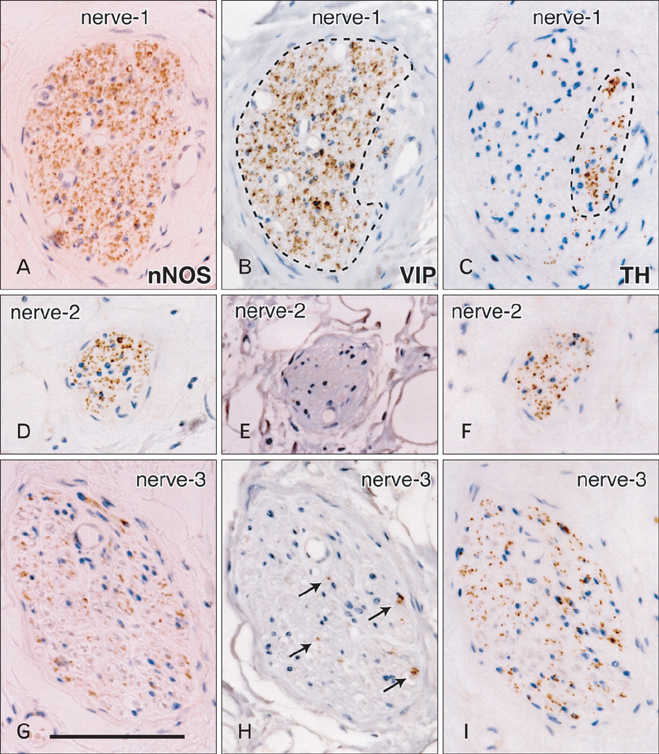
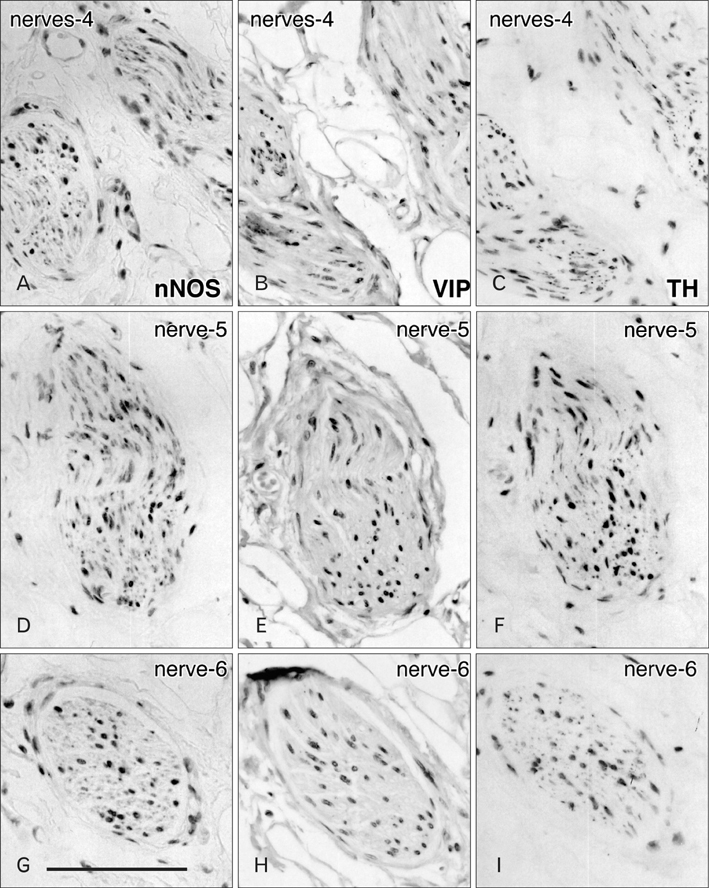
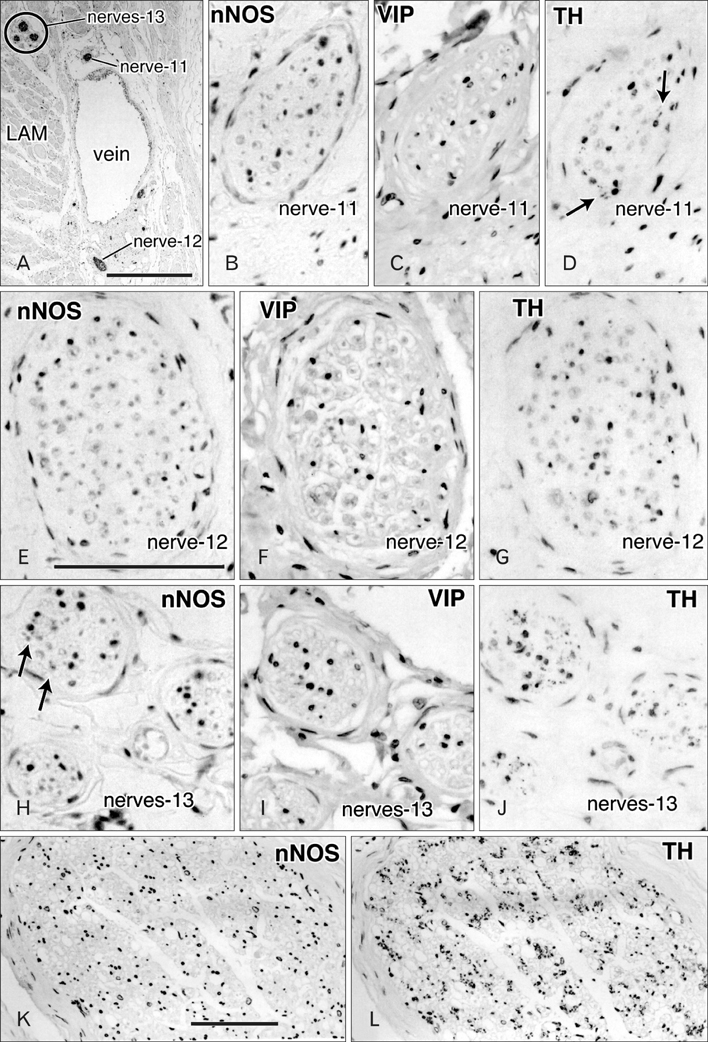
- Although the pelvic autonomic plexus may be considered a mixture of sympathetic and parasympathetic nerves, little information on its composite fibers is available. Using 10 donated elderly cadavers, we investigated in detail the topohistology of nerve fibers in the posterior part of the periprostatic region in males and the infero-anterior part of the paracolpium in females. Neuronal nitric oxide synthase (nNOS) and vasoactive intestinal polypeptide (VIP) were used as parasympathetic nerve markers, and tyrosine hydroxylase (TH) was used as a marker of sympathetic nerves. In the region examined, nNOS-positive nerves (containing nNOS-positive fibers) were consistently predominant numerically. All fibers positive for these markers appeared to be thin, unmyelinated fibers. Accordingly, the pelvic plexus branches were classified into 5 types: triple-positive mixed nerves (nNOS+, VIP+, TH+, thick myelinated fibers + or -); double-positive mixed nerves (nNOS+, VIP-, TH+, thick myelinated fibers + or -); nerves in arterial walls (nNOS-, VIP+, TH+, thick myelinated fibers-); non-parasympathetic nerves (nNOS-, VIP-, TH+, thick myelinated fibers + or -); (although rare) pure sensory nerve candidates (nNOS-, VIP-, TH-, thick myelinated fibers+). Triple-positive nerves were 5-6 times more numerous in the paracolpium than in the periprostatic region. Usually, the parasympathetic nerve fibers did not occupy a specific site in a nerve, and were intermingled with sympathetic fibers. This morphology might be the result of an "incidentally" adopted nerve fiber route, rather than a target-specific pathway.
Keyword
MeSH Terms
Figure
Reference
-
1. Hoyle CH, Stones RW, Robson T, Whitley K, Burnstock G. Innervation of vasculature and microvasculature of the human vagina by NOS and neuropeptide-containing nerves. J Anat. 1996; 188(Pt 3):633–644.2. Busacchi P, De Giorgio R, Santini D, Bellavia E, Perri T, Oliverio C, Paradisi R, Corinaldesi R, Flamigni C. A histological and immunohistochemical study of neuropeptide containing somatic nerves in the levator ani muscle of women with genitourinary prolapse. Acta Obstet Gynecol Scand. 1999; 78:2–5.3. Butler-Manuel SA, Buttery LD, A'Hern RP, Polak JM, Barton DP. Pelvic nerve plexus trauma at radical and simple hysterectomy: a quantitative study of nerve types in the uterine supporting ligaments. J Soc Gynecol Investig. 2002; 9:47–56.4. Butler-Manuel SA, Buttery LD, Polak JM, A'Hern R, Barton DP. Autonomic nerve trauma at radical hysterectomy: the nerve content and subtypes within the superficial and deep uterosacral ligaments. Reprod Sci. 2008; 15:91–96.5. Uckert S, Stanarius A, Stief CG, Wolf G, Jonas U, Machtens S. Immunocytochemical distribution of nitric oxide synthase in the human seminal vesicle: a light and electron microscopical study. Urol Res. 2003; 31:262–266.6. Hisasue S, Hashimoto K, Kobayashi K, Takeuchi M, Kyoda Y, Sato S, Masumori N, Tsukamoto T. Baseline erectile function alters the cavernous nerve quantity and distribution around the prostate. J Urol. 2010; 184:2062–2067.7. Alsaid B, Bessede T, Karam I, Abd-Alsamad I, Uhl JF, Benoît G, Droupy S, Delmas V. Coexistence of adrenergic and cholinergic nerves in the inferior hypogastric plexus: anatomical and immuno histochemical study with 3D reconstruction in human male fetus. J Anat. 2009; 214:645–654.8. Moszkowicz D, Peschaud F, Bessede T, Benoit G, Alsaid B. Internal anal sphincter parasympathetic-nitrergic and sympathetic-adrenergic innervation: a 3-dimensional morphological and functional analysis. Dis Colon Rectum. 2012; 55:473–481.9. Ali M, Johnson IP, Hobson J, Mohammadi B, Khan F. Anatomy of the pelvic plexus and innervation of the prostate gland. Clin Anat. 2004; 17:123–129.10. Takenaka A, Kawada M, Murakami G, Hisasue S, Tsukamoto T, Fujisawa M. Interindividual variation in distribution of extramural ganglion cells in the male pelvis: a semi-quantitative and immunohistochemical study concerning nerve-sparing pelvic surgery. Eur Urol. 2005; 48:46–52.11. Imai K, Furuya K, Kawada M, Kinugasa Y, Omote K, Namiki A, Uchiyama E, Murakami G. Human pelvic extramural ganglion cells: a semiquantitative and immunohistochemical study. Surg Radiol Anat. 2006; 28:596–605.12. Hieda K, Cho KH, Arakawa T, Fujimiya M, Murakami G, Matsubara A. Nerves in the intersphincteric space of the human anal canal with special reference to their continuation to the enteric nerve plexus of the rectum. Clin Anat. 2013; 03. 20. [Epub]. http://dx.doi.org/10.1002/ca.22227.13. Maas CP, DeRuiter MC, Kenter GG, Trimbos JB. The inferior hypogastric plexus in gynecologic surgery. J Gynecol Tech. 1999; 5:55–62.14. Baader B, Herrmann M. Topography of the pelvic autonomic nervous system and its potential impact on surgical intervention in the pelvis. Clin Anat. 2003; 16:119–130.15. Akita K, Sakamoto H, Sato T. Origins and courses of the nervous branches to the male urethral sphincter. Surg Radiol Anat. 2003; 25:387–392.16. Costello AJ, Brooks M, Cole OJ. Anatomical studies of the neurovascular bundle and cavernosal nerves. BJU Int. 2004; 94:1071–1076.17. Mauroy B, Demondion X, Bizet B, Claret A, Mestdagh P, Hurt C. The female inferior hypogastric (= pelvic) plexus: anatomical and radiological description of the plexus and its afferences: applications to pelvic surgery. Surg Radiol Anat. 2007; 29:55–66.18. Grozdanovic Z, Baumgarten HG. Colocalisation of NADPH-diaphorase with neuropeptides in the ureterovesical ganglia of humans. Acta Histochem. 1996; 98:245–253.19. Takenaka A, Murakami G, Matsubara A, Han SH, Fujisawa M. Variation in course of cavernous nerve with special reference to details of topographic relationships near prostatic apex: histologic study using male cadavers. Urology. 2005; 65:136–142.20. Kato M, Niikura H, Yaegashi N, Murakami G, Tatsumi H, Matsubara A. Histotopography of the female cavernous nerve: a study using donated fetuses and adult cadavers. Int Urogynecol J Pelvic Floor Dysfunct. 2008; 19:1687–1695.21. Hirata E, Koyama M, Murakami G, Ohtsuka A, Abe S, Ide Y, Fujiwara H, Kudo Y. Comparative histological study of levels 1-3 supportive tissues using pelvic floor semiserial sections from elderly nulliparous and multiparous women. J Obstet Gynaecol Res. 2011; 37:13–23.22. Motohashi O, Suzuki M, Shida N, Umezawa K, Ohtoh T, Sakurai Y, Yoshimoto T. Subarachnoid haemorrhage induced proliferation of leptomeningeal cells and deposition of extracellular matrices in the arachnoid granulations and subarachnoid space. Immunhistochemical study. Acta Neurochir (Wien). 1995; 136:88–91.23. Hayashi T, Kumasaka T, Mitani K, Yao T, Suda K, Seyama K. Loss of heterozygosity on tuberous sclerosis complex genes in multifocal micronodular pneumocyte hyperplasia. Mod Pathol. 2010; 23:1251–1260.24. Boreham MK, Wai CY, Miller RT, Schaffer JI, Word RA. Morphometric properties of the posterior vaginal wall in women with pelvic organ prolapse. Am J Obstet Gynecol. 2002; 187:1501–1508.25. Hong X, Huang L, Song Y. Role of vasoactive intestinal peptide and pituitary adenylate cyclase activating polypeptide in the vaginal wall of women with stress urinary incontinence and pelvic organ prolapse. Int Urogynecol J Pelvic Floor Dysfunct. 2008; 19:1151–1157.26. Zhu L, Lang J, Jiang X, Jiang F, Chen J, Wong F. Neuropeptide Y expression in vaginal epithelium of women with pelvic organ prolapse and stress urinary incontinence. Int J Gynaecol Obstet. 2008; 102:65–68.27. Maggi CA, Giuliani S, Santicioli P, Patacchini R, Said SI, Theodorsson E, Turini D, Barbanti G, Giachetti A, Meli A. Direct evidence for the involvement of vasoactive intestinal polypeptide in the motor response of the human isolated ileum to capsaicin. Eur J Pharmacol. 1990; 185:169–178.28. Jacobson ED, Berguer R, Pawlik WW, Hottenstein OD. Mesenteric purinergic and peptidergic vasodilators. In : Wood JD, editor. The Gastointestinal System. Moltility and Circulation, Handbook of Physiology, Section 6. Bethesda: American Physiological Society;1989. p. 1647–1667.29. Ekblad E, Bauer AJ. Role of vasoactive intestinal peptide and inflammatory mediators in enteric neuronal plasticity. Neurogastroenterol Motil. 2004; 16:Suppl 1. 123–128.30. Kuntz A, Hoffman H, Schaeffer EM. Fiber components of the splanchnic nerves. Anat Rec. 1957; 128:139–146.31. Donker PJ. A study of the myelinated fibres in the branches of the pelvic plexus. Neurourol Urodyn. 1986; 5:185–202.32. Lance-Jones C, Landmesser L. Pathway selection by embryonic chick motoneurons in an experimentally altered environment. Proc R Soc Lond B Biol Sci. 1981; 214:19–52.33. Honig MG, Lance-Jones C, Landmesser L. The development of sensory projection patterns in embryonic chick hindlimb under experimental conditions. Dev Biol. 1986; 118:532–548.34. Ferns MJ, Hollyday M. Motor innervation of dorsoventrally reversed wings in chick/quail chimeric embryos. J Neurosci. 1993; 13:2463–2476.35. Tosney KW, Hotary KB, Lance-Jones C. Specifying the target identity of motoneurons. Bioessays. 1995; 17:379–382.36. Yucel S, De Souza A Jr, Baskin LS. Neuroanatomy of the human female lower urogenital tract. J Urol. 2004; 172:191–195.37. Kiyoshima K, Yokomizo A, Yoshida T, Tomita K, Yonemasu H, Nakamura M, Oda Y, Naito S, Hasegawa Y. Anatomical features of periprostatic tissue and its surroundings: a histological analysis of 79 radical retropubic prostatectomy specimens. Jpn J Clin Oncol. 2004; 34:463–468.38. Costello AJ, Dowdle BW, Namdarian B, Pedersen J, Murphy DG. Immunohistochemical study of the cavernous nerves in the periprostatic region. BJU Int. 2011; 107:1210–1215.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Composite nerve fibers in the hypogastric and pelvic splanchnic nerves: an immunohistochemical study using elderly cadavers
- Human nasociliary nerve with special reference to its unique parasympathetic cutaneous innervation
- Nerves and fasciae in and around the paracolpium or paravaginal tissue: an immunohistochemical study using elderly donated cadavers
- Superior Hypogastric Plexus Block for Malignant Pelvic Pain
- A New Technique for Inferior Hypogastric Plexus Block: A Coccygeal Transverse Approach: A Case Report