Immune Netw.
2011 Oct;11(5):245-252. 10.4110/in.2011.11.5.245.
Antimicrobial Peptides in Innate Immunity against Mycobacteria
- Affiliations
-
- 1Department of Microbiology and Infection Signaling Network Research Center, Chungnam National University, School of Medicine, Daejeon 301-747, Korea. hayoungj@cnu.ac.kr
- KMID: 2167996
- DOI: http://doi.org/10.4110/in.2011.11.5.245
Abstract
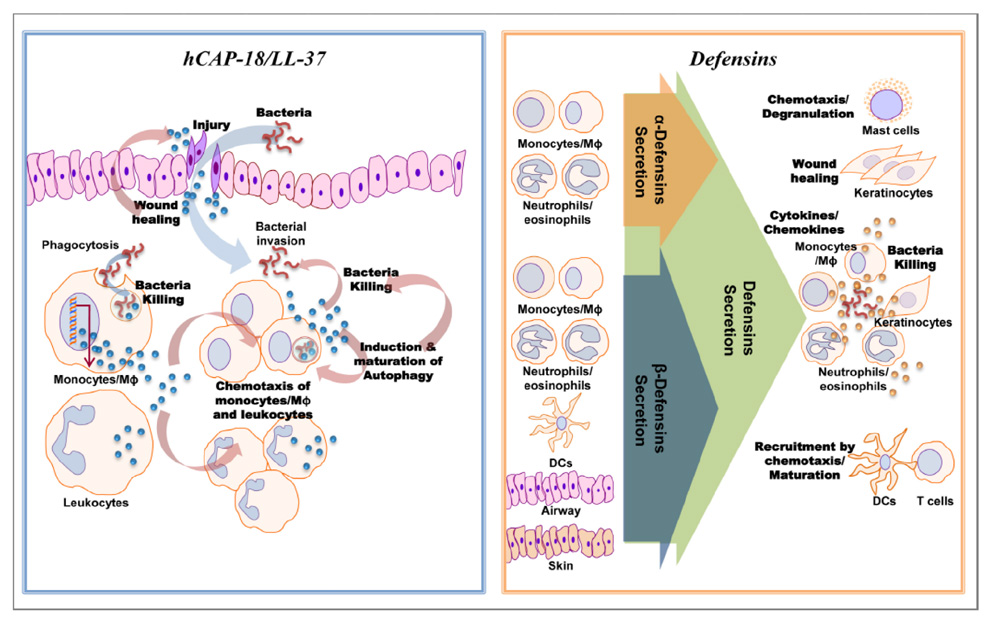
- Antimicrobial peptides/proteins are ancient and naturallyoccurring antibiotics in innate immune responses in a variety of organisms. Additionally, these peptides have been recognized as important signaling molecules in regulation of both innate and adaptive immunity. During mycobacterial infection, antimicrobial peptides including cathelicidin, defensin, and hepcidin have antimicrobial activities against mycobacteria, making them promising candidates for future drug development. Additionally, antimicrobial peptides act as immunomodulators in infectious and inflammatory conditions. Multiple crucial functions of cathelicidins in antimycobacterial immune defense have been characterized not only in terms of direct killing of mycobacteria but also as innate immune regulators, i.e., in secretion of cytokines and chemokines, and mediating autophagy activation. Defensin families are also important during mycobacterial infection and contribute to antimycobacterial defense and inhibition of mycobacterial growth both in vitro and in vivo. Hepcidin, although its role in mycobacterial infection has not yet been characterized, exerts antimycobacterial effects in activated macrophages. The present review focuses on recent efforts to elucidate the roles of host defense peptides in innate immunity to mycobacteria.
MeSH Terms
-
Adaptive Immunity
Anti-Bacterial Agents
Antimicrobial Cationic Peptides
Autophagy
Cathelicidins
Chemokines
Cytokines
Homicide
Humans
Immunity, Innate
Immunologic Factors
Macrophages
Negotiating
Peptides
Anti-Bacterial Agents
Antimicrobial Cationic Peptides
Cathelicidins
Chemokines
Cytokines
Immunologic Factors
Peptides
Figure
Reference
-
1. WHO. Global Tubercalosis Control 2010. 2010. 07. Geneva: WHO.2. Pieters J. Mycobacterium tuberculosis and the macrophage: maintaining a balance. Cell Host Microbe. 2008. 3:399–407.
Article3. Flynn JL, Chan J. Tuberculosis: latency and reactivation. Infect Immun. 2001. 69:4195–4201.
Article4. Zaiou M. Multifunctional antimicrobial peptides: therapeutic targets in several human diseases. J Mol Med. 2007. 85:317–329.
Article5. Liu PT, Modlin RL. Human macrophage host defense against Mycobacterium tuberculosis. Curr Opin Immunol. 2008. 20:371–376.
Article6. Liu PT, Stenger S, Li H, Wenzel L, Tan BH, Krutzik SR, Ochoa MT, Schauber J, Wu K, Meinken C, Kamen DL, Wagner M, Bals R, Steinmeyer A, Zügel U, Gallo RL, Eisenberg D, Hewison M, Hollis BW, Adams JS, Bloom BR, Modlin RL. Toll-like receptor triggering of a vitamin D-mediated human antimicrobial response. Science. 2006. 311:1770–1773.
Article7. Hansdottir S, Monick MM, Hinde SL, Lovan N, Look DC, Hunninghake GW. Respiratory epithelial cells convert inactive vitamin D to its active form: potential effects on host defense. J Immunol. 2008. 181:7090–7099.
Article8. Cole AM, Waring AJ. The role of defensins in lung biology and therapy. Am J Respir Med. 2002. 1:249–259.
Article9. Liu PT, Schenk M, Walker VP, Dempsey PW, Kanchanapoomi M, Wheelwright M, Vazirnia A, Zhang X, Steinmeyer A, Zugel U, Hollis BW, Cheng G, Modlin RL. Convergence of IL-1beta and VDR activation pathways in human TLR2/1-induced antimicrobial responses. PLoS One. 2009. 4:e5810.10. Sow FB, Florence WC, Satoskar AR, Schlesinger LS, Zwilling BS, Lafuse WP. Expression and localization of hepcidin in macrophages: a role in host defense against tuberculosis. J Leukoc Biol. 2007. 82:934–945.
Article11. Beisswenger C, Bals R. Functions of antimicrobial peptides in host defense and immunity. Curr Protein Pept Sci. 2005. 6:255–264.
Article12. Lehrer RI, Ganz T. Cathelicidins: a family of endogenous antimicrobial peptides. Curr Opin Hematol. 2002. 9:18–22.
Article13. Zanetti M. The role of cathelicidins in the innate host defenses of mammals. Curr Issues Mol Biol. 2005. 7:179–196.14. Fahy RJ, Wewers MD. Pulmonary defense and the human cathelicidin hCAP-18/LL-37. Immunol Res. 2005. 31:75–89.
Article15. Zanetti M. Cathelicidins, multifunctional peptides of the innate immunity. J Leukoc Biol. 2004. 75:39–48.
Article16. Lai Y, Gallo RL. AMPed up immunity: how antimicrobial peptides have multiple roles in immune defense. Trends Immunol. 2009. 30:131–141.
Article17. Yuk JM, Shin DM, Lee HM, Yang CS, Jin HS, Kim KK, Lee ZW, Lee SH, Kim JM, Jo EK. Vitamin D3 induces autophagy in human monocytes/macrophages via cathelicidin. Cell Host Microbe. 2009. 6:231–243.
Article18. Lehrer RI, Lichtenstein AK, Ganz T. Defensins: antimicrobial and cytotoxic peptides of mammalian cells. Annu Rev Immunol. 1993. 11:105–128.
Article19. Driss V, Legrand F, Hermann E, Loiseau S, Guerardel Y, Kremer L, Adam E, Woerly G, Dombrowicz D, Capron M. TLR2-dependent eosinophil interactions with mycobacteria: role of alpha-defensins. Blood. 2009. 113:3235–3244.
Article20. Kisich KO, Heifets L, Higgins M, Diamond G. Antimycobacterial agent based on mRNA encoding human beta-defensin 2 enables primary macrophages to restrict growth of Mycobacterium tuberculosis. Infect Immun. 2001. 69:2692–2699.
Article21. Yang D, Chertov O, Bykovskaia SN, Chen Q, Buffo MJ, Shogan J, Anderson M, Schröder JM, Wang JM, Howard OM, Oppenheim JJ. Beta-defensins: linking innate and adaptive immunity through dendritic and T cell CCR6. Science. 1999. 286:525–528.
Article22. De Smet K, Contreras R. Human antimicrobial peptides: defensins, cathelicidins and histatins. Biotechnol Lett. 2005. 27:1337–1347.
Article23. O'Neil DA, Porter EM, Elewaut D, Anderson GM, Eckmann L, Ganz T, Kagnoff MF. Expression and regulation of the human beta-defensins hBD-1 and hBD-2 in intestinal epithelium. J Immunol. 1999. 163:6718–6724.24. Becker MN, Diamond G, Verghese MW, Randell SH. CD14-dependent lipopolysaccharide-induced beta-defensin-2 expression in human tracheobronchial epithelium. J Biol Chem. 2000. 275:29731–29736.
Article25. Oppenheim JJ, Biragyn A, Kwak LW, Yang D. Roles of antimicrobial peptides such as defensins in innate and adaptive immunity. Ann Rheum Dis. 2003. 62:Suppl 2. ii17–ii21.
Article26. Kai-Larsen Y, Agerberth B. The role of the multifunctional peptide LL-37 in host defense. Front Biosci. 2008. 13:3760–3767.
Article27. Rivas-Santiago B, Hernandez-Pando R, Carranza C, Juarez E, Contreras JL, Aguilar-Leon D, Torres M, Sada E. Expression of cathelicidin LL-37 during Mycobacterium tuberculosis infection in human alveolar macrophages, monocytes, neutrophils, and epithelial cells. Infect Immun. 2008. 76:935–941.
Article28. Martineau AR, Newton SM, Wilkinson KA, Kampmann B, Hall BM, Nawroly N, Packe GE, Davidson RN, Griffiths CJ, Wilkinson RJ. Neutrophil-mediated innate immune resistance to mycobacteria. J Clin Invest. 2007. 117:1988–1994.
Article29. Bals R, Wang X, Zasloff M, Wilson JM. The peptide antibiotic LL-37/hCAP-18 is expressed in epithelia of the human lung where it has broad antimicrobial activity at the airway surface. Proc Natl Acad Sci U S A. 1998. 95:9541–9546.
Article30. Agerberth B, Grunewald J, Castaños-Velez E, Olsson B, Jörnvall H, Wigzell H, Eklund A, Gudmundsson GH. Antibacterial components in bronchoalveolar lavage fluid from healthy individuals and sarcoidosis patients. Am J Respir Crit Care Med. 1999. 160:283–290.
Article31. Lee HM, Shin DM, Choi DK, Lee ZW, Kim KH, Yuk JM, Kim CD, Lee JH, Jo EK. Innate immune responses to Mycobacterium ulcerans via toll-like receptors and dectin-1 in human keratinocytes. Cell Microbiol. 2009. 11:678–692.
Article32. Yang CS, Shin DM, Kim KH, Lee ZW, Lee CH, Park SG, Bae YS, Jo EK. NADPH oxidase 2 interaction with TLR2 is required for efficient innate immune responses to mycobacteria via cathelicidin expression. J Immunol. 2009. 182:3696–3705.
Article33. Méndez-Samperio P, Pérez A, Torres L. Role of reactive oxygen species (ROS) in Mycobacterium bovis bacillus Calmette Guerin-mediated up-regulation of the human cathelicidin LL-37 in A549 cells. Microb Pathog. 2009. 47:252–257.
Article34. Jo EK. Innate immunity to mycobacteria: vitamin D and autophagy. Cell Microbiol. 2010. 12:1026–1035.
Article35. Gutierrez MG, Master SS, Singh SB, Taylor GA, Colombo MI, Deretic V. Autophagy is a defense mechanism inhibiting BCG and Mycobacterium tuberculosis survival in infected macrophages. Cell. 2004. 119:753–766.
Article36. Shin DM, Yuk JM, Lee HM, Lee SH, Son JW, Harding CV, Kim JM, Modlin RL, Jo EK. Mycobacterial lipoprotein activates autophagy via TLR2/1/CD14 and a functional vitamin D receptor signalling. Cell Microbiol. 2010. 12:1648–1665.
Article37. De Yang , Chen Q, Schmidt AP, Anderson GM, Wang JM, Wooters J, Oppenheim JJ, Chertov O. LL-37, the neutrophil granule- and epithelial cell-derived cathelicidin, utilizes formyl peptide receptor-like 1 (FPRL1) as a receptor to chemoattract human peripheral blood neutrophils, monocytes, and T cells. J Exp Med. 2000. 192:1069–1074.38. Kurosaka K, Chen Q, Yarovinsky F, Oppenheim JJ, Yang D. Mouse cathelin-related antimicrobial peptide chemoattracts leukocytes using formyl peptide receptor-like 1/mouse formyl peptide receptor-like 2 as the receptor and acts as an immune adjuvant. J Immunol. 2005. 174:6257–6265.
Article39. Edfeldt K, Liu PT, Chun R, Fabri M, Schenk M, Wheelwright M, Keegan C, Krutzik SR, Adams JS, Hewison M, Modlin RL. T-cell cytokines differentially control human monocyte antimicrobial responses by regulating vitamin D metabolism. Proc Natl Acad Sci U S A. 2010. 107:22593–22598.
Article40. Ganz T. Antimicrobial polypeptides in host defense of the respiratory tract. J Clin Invest. 2002. 109:693–697.
Article41. Wilson CL, Ouellette AJ, Satchell DP, Ayabe T, López-Boado YS, Stratman JL, Hultgren SJ, Matrisian LM, Parks WC. Regulation of intestinal alpha-defensin activation by the metalloproteinase matrilysin in innate host defense. Science. 1999. 286:113–117.
Article42. Schutte BC, Mitros JP, Bartlett JA, Walters JD, Jia HP, Welsh MJ, Casavant TL, McCray PB Jr. Discovery of five conserved beta -defensin gene clusters using a computational search strategy. Proc Natl Acad Sci U S A. 2002. 99:2129–2133.
Article43. Jacobsen M, Repsilber D, Gutschmidt A, Neher A, Feldmann K, Mollenkopf HJ, Ziegler A, Kaufmann SH. Candidate biomarkers for discrimination between infection and disease caused by Mycobacterium tuberculosis. J Mol Med (Berl). 2007. 85:613–621.
Article44. Kalita A, Verma I, Khuller GK. Role of human neutrophil peptide-1 as a possible adjunct to antituberculosis chemotherapy. J Infect Dis. 2004. 190:1476–1480.
Article45. Harder J, Meyer-Hoffert U, Teran LM, Schwichtenberg L, Bartels J, Maune S, Schroder JM. Mucoid Pseudomonas aer uginosa, TNF-alpha, and IL-1beta, but not IL-6, induce human beta-defensin-2 in respiratory epithelia. Am J Respir Cell Mol Biol. 2000. 22:714–721.
Article46. Méndez-Samperio P, Miranda E, Trejo A. Mycobacterium bovis Bacillus Calmette-Guérin (BCG) stimulates human beta-defensin-2 gene transcription in human epithelial cells. Cell Immunol. 2006. 239:61–66.
Article47. Rivas-Santiago CE, Rivas-Santiago B, León DA, Castañeda-Delgado J, Hernández Pando R. Induction of β-defensins by l-isoleucine as novel immunotherapy in experimental murine tuberculosis. Clin Exp Immunol. 2011. 164:80–89.
Article48. Aguilar León D, Zumárraga MJ, Jiménez Oropeza R, Gioffré AK, Bernardelli A, Orozco Estévez H, Cataldi AA, Hernández Pando R. Mycobacterium bovis with different genotypes and from different hosts induce dissimilar immunopathological lesions in a mouse model of tuberculosis. Clin Exp Immunol. 2009. 157:139–147.
Article49. Rivas-Santiago B, Sada E, Tsutsumi V, Aguilar-Leon D, Contreras JL, Hernandez-Pando R. beta-Defensin gene expression during the course of experimental tuberculosis infection. J Infect Dis. 2006. 194:697–701.
Article50. Cole AM, Wang W, Waring AJ, Lehrer RI. Retrocyclins: using past as prologue. Curr Protein Pept Sci. 2004. 5:373–381.
Article51. Tang YQ, Yuan J, Osapay G, Osapay K, Tran D, Miller CJ, Ouellette AJ, Selsted ME. A cyclic antimicrobial peptide produced in primate leukocytes by the ligation of two truncated alpha-defensins. Science. 1999. 286:498–502.
Article52. Leonova L, Kokryakov VN, Aleshina G, Hong T, Nguyen T, Zhao C, Waring AJ, Lehrer RI. Circular minidefensins and posttranslational generation of molecular diversity. J Leukoc Biol. 2001. 70:461–464.53. Tran D, Tran PA, Tang YQ, Yuan J, Cole T, Selsted ME. Homodimeric theta-defensins from rhesus macaque leukocytes: isolation, synthesis, antimicrobial activities, and bacterial binding properties of the cyclic peptides. J Biol Chem. 2002. 277:3079–3084.54. Brandt CR, Akkarawongsa R, Altmann S, Jose G, Kolb AW, Waring AJ, Lehrer RI. Evaluation of a theta-defensin in a Murine model of herpes simplex virus type 1 keratitis. Invest Ophthalmol Vis Sci. 2007. 48:5118–5124.
Article55. Wang W, Cole AM, Hong T, Waring AJ, Lehrer RI. Retrocyclin, an antiretroviral theta-defensin, is a lectin. J Immunol. 2003. 170:4708–4716.56. Yang C, Boone L, Nguyen TX, Rudolph D, Limpakarnjanarat K, Mastro TD, Tappero J, Cole AM, Lal RB. Theta-Defensin pseudogenes in HIV-1-exposed, persistently seronegative female sex-workers from Thailand. Infect Genet Evol. 2005. 5:11–15.
Article57. Ganz T. Hepcidin and its role in regulating systemic iron metabolism. Hematology Am Soc Hematol Educ Program. 2006. 29–35.
Article58. Ganz T. Hepcidin--a peptide hormone at the interface of innate immunity and iron metabolism. Curr Top Microbiol Immunol. 2006. 306:183–198.
Article59. Ganz T, Nemeth E. Iron sequestration and anemia of inflammation. Semin Hematol. 2009. 46:387–393.
Article60. Nemeth E, Ganz T. The role of hepcidin in iron metabolism. Acta Haematol. 2009. 122:78–86.
Article61. Atanasiu V, Manolescu B, Stoian I. Hepcidin the link between inflammation and anemia in chronic renal failure. Rom J Intern Med. 2006. 44:25–33.62. Sow FB, Alvarez GR, Gross RP, Satoskar AR, Schlesinger LS, Zwilling BS, Lafuse WP. Role of STAT1, NF-kappaB, and C/EBPbeta in the macrophage transcriptional regulation of hepcidin by mycobacterial infection and IFN-gamma. J Leukoc Biol. 2009. 86:1247–1258.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Sphingolipids and Antimicrobial Peptides: Function and Roles in Atopic Dermatitis
- Cathelicidin LL-37: An Antimicrobial Peptide with a Role in Inflammatory Skin Disease
- Immunopathogenesis of Non-Tuberculous Mycobacteria Lung Disease
- Molecular Defense Mechanisms during Urinary Tract Infection
- Neonatal innate immunity and Toll-like receptor