Cancer Res Treat.
2011 Dec;43(4):212-216.
Phase II Study of S-1 Plus Either Irinotecan or Docetaxel for Non-small Cell Lung Cancer Patients Treated with More Than Three Lines of Treatment
- Affiliations
-
- 1Department of Oncology, Asan Medical Center, University of Ulsan College of Medicine, Seoul, Korea. leedaeho@amc.seoul.kr
Abstract
- PURPOSE
This study was designed to evaluate the efficacy of a combination treatment of S-1 plus either irinotecan or docetaxel for advanced/metastatic non-small cell lung cancer (NSCLC) patients who have already failed 3 or more lines of treatment.
MATERIALS AND METHODS
This was a prospective single center phase II study. The eligible patients received S-1 40 mg/m2 twice a day orally on days 1 though 14 combined with irinotecan 150 mg/m2on D1 only or docetaxel 35 mg/m2 on D1 and D8. The treatment was repeated every 3 weeks until disease progression, unacceptable toxicity, or patient refusal. The choice between the two regimens was made at the discretion of the treating physician.
RESULTS
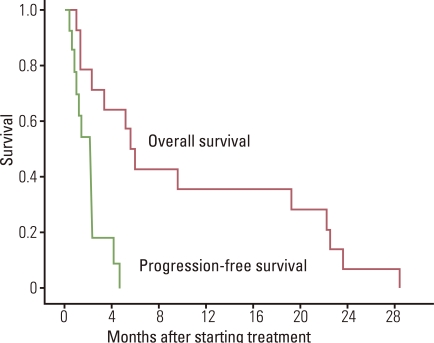
A total of 14 patients participated in the study. There were 3 patients with squamous cell carcinoma, 9 with adenocarcinoma, and 2 with NSCLC, NOS. Eight of the patients were male. There were 8 patients with an Eastern Cooperative Oncology Group (ECOG) of 1, and 6 patients with an ECOG of 2. All the patients had already been treated with platinum-based chemotherapy and epidermal growth factor receptor tyrosine kinase inhibitor therapy. Out of the 14 patients, 10 received irinotecan and S-1 and the other 4 received docetaxel and S-1. Twelve patients had also received pemetrexed. Disappointingly, there were no response from 2 patients with a stable disease, and therefore, as per the protocol, we stopped the study early. With a median follow-up time of 49 months, the median survival time was 5.6 months (95% confidence interval, 4.3 to 6.9 months).
CONCLUSION
S-1 containing doublets did not show activity in this population as a salvage treatment and further investigation cannot be recommended.
MeSH Terms
-
Adenocarcinoma
Camptothecin
Carcinoma, Non-Small-Cell Lung
Carcinoma, Squamous Cell
Disease Progression
Disulfiram
Follow-Up Studies
Glutamates
Guanine
Humans
Male
Prospective Studies
Protein-Tyrosine Kinases
Receptor, Epidermal Growth Factor
Salvage Therapy
Taxoids
Pemetrexed
Camptothecin
Disulfiram
Glutamates
Guanine
Protein-Tyrosine Kinases
Receptor, Epidermal Growth Factor
Taxoids
Figure
Reference
-
1. Schiller JH, Harrington D, Belani CP, Langer C, Sandler A, Krook J, et al. Comparison of four chemotherapy regimens for advanced non-small-cell lung cancer. N Engl J Med. 2002; 346:92–98. PMID: 11784875.
Article2. Pirker R, Pereira JR, Szczesna A, von Pawel J, Krzakowski M, Ramlau R, et al. Cetuximab plus chemotherapy in patients with advanced non-small-cell lung cancer (FLEX): an open-label randomised phase III trial. Lancet. 2009; 373:1525–1531. PMID: 19410716.
Article3. Sandler A, Gray R, Perry MC, Brahmer J, Schiller JH, Dowlati A, et al. Paclitaxel-carboplatin alone or with bevacizumab for non-small-cell lung cancer. N Engl J Med. 2006; 355:2542–2550. PMID: 17167137.
Article4. Shepherd FA, Dancey J, Ramlau R, Mattson K, Gralla R, O'Rourke M, et al. Prospective randomized trial of docetaxel versus best supportive care in patients with non-small-cell lung cancer previously treated with platinum-based chemotherapy. J Clin Oncol. 2000; 18:2095–2103. PMID: 10811675.
Article5. Hanna N, Shepherd FA, Fossella FV, Pereira JR, De Marinis F, von Pawel J, et al. Randomized phase III trial of pemetrexed versus docetaxel in patients with non-small-cell lung cancer previously treated with chemotherapy. J Clin Oncol. 2004; 22:1589–1597. PMID: 15117980.
Article6. Shepherd FA, Rodrigues Pereira J, Ciuleanu T, Tan EH, Hirsh V, Thongprasert S, et al. Erlotinib in previously treated non-small-cell lung cancer. N Engl J Med. 2005; 353:123–132. PMID: 16014882.
Article7. Kim ES, Hirsh V, Mok T, Socinski MA, Gervais R, Wu YL, et al. Gefitinib versus docetaxel in previously treated non-small-cell lung cancer (INTEREST): a randomised phase III trial. Lancet. 2008; 372:1809–1818. PMID: 19027483.
Article8. Shirasaka T, Shimamato Y, Ohshimo H, Yamaguchi M, Kato T, Yonekura K, et al. Development of a novel form of an oral 5-fluorouracil derivative (S-1) directed to the potentiation of the tumor selective cytotoxicity of 5-fluorouracil by two biochemical modulators. Anticancer Drugs. 1996; 7:548–557. PMID: 8862723.
Article9. Shirasaka T, Shimamoto Y, Fukushima M. Inhibition by oxonic acid of gastrointestinal toxicity of 5-fluorouracil without loss of its antitumor activity in rats. Cancer Res. 1993; 53:4004–4009. PMID: 7689420.10. Okamoto I, Yoshioka H, Morita S, Ando M, Takeda K, Seto T, et al. Phase III trial comparing oral S-1 plus carboplatin with paclitaxel plus carboplatin in chemotherapy-naïve patients with advanced non-small-cell lung cancer: results of a west Japan oncology group study. J Clin Oncol. 2010; 28:5240–5246. PMID: 21079147.
Article11. Govindan R, Morgensztern D, Kommor MD, Herbst RS, Schaefer P, Gandhi J, et al. Phase II trial of S-1 as second-line therapy in patients with advanced non-small cell lung cancer. J Thorac Oncol. 2011; 6:790–795. PMID: 21325974.
Article12. Ono A, Naito T, Murakami H, Takahashi T, Nakamura Y, Tsuya A, et al. Evaluation of S-1 as third- or further-line chemotherapy in advanced non-small-cell lung cancer. Int J Clin Oncol. 2010; 15:161–165. PMID: 20198400.
Article13. Han JY, Lee DH, Kim HY, Hong EK, Yoon SM, Chun JH, et al. A phase II study of weekly docetaxel plus capecitabine for patients with advanced nonsmall cell lung carcinoma. Cancer. 2003; 98:1918–1924. PMID: 14584075.
Article14. Han JY, Lee DH, Kim HY, Kim EA, Lee JJ, Ju SY, et al. A Phase II study of weekly irinotecan and capecitabine in patients with previously treated non-small cell lung cancer. Clin Cancer Res. 2003; 9(16 Pt 1):5909–5914. PMID: 14676114.15. Han JY, Lee DH, Lee SY, Park CG, Kim HY, Lee HG, et al. Phase II study of weekly irinotecan plus capecitabine for chemotherapy-naïve patients with advanced nonsmall cell lung carcinoma. Cancer. 2005; 104:2759–2765. PMID: 16294344.16. Lee JJ, Han JY, Lee DH, Kim HY, Chun JH, Lee HG, et al. A phase II trial of docetaxel plus capecitabine in patients with previously treated non-small cell lung cancer. Jpn J Clin Oncol. 2006; 36:761–767. PMID: 17043059.
Article17. Chen YM, Shih JF, Fan WC, Wu CH, Chou KT, Tsai CM, et al. Third-line or fourth-line chemotherapy in non-small-cell lung cancer patients with relatively good performance status. J Chin Med Assoc. 2011; 74:209–214. PMID: 21550007.
Article18. Fox E, Curt GA, Balis FM. Clinical trial design for target-based therapy. Oncologist. 2002; 7:401–409. PMID: 12401902.
Article19. Adjei AA, Salavaggione OE, Mandrekar SJ, Dy GK, Ziegler KL, Endo C, et al. Correlation between polymorphisms of the reduced folate carrier gene (SLC19A1) and survival after pemetrexed-based therapy in non-small cell lung cancer: a North Central Cancer Treatment Group-based exploratory study. J Thorac Oncol. 2010; 5:1346–1353. PMID: 20651609.
Article20. Kim ES, Herbst RS, Wistuba II, Lee JJ, Blumenschein GR, Tsao A, et al. The BATTLE tiral: personalizing therapy for lung cancer. Cancer Discov. 2011; 1:44–53.21. Kris MG, Johnson BE, Kwiatkowski DJ, Iafrate AJ, Wistuba II, Aronson SL, et al. Identification of driver mutations in tumor specimens from 1,000 patients with lung adenocarcinoma: The NCI's Lung Cancer Mutation Consortium (LCMC). J Clin Oncol. 2011; 29(s):CRA7506.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Cell Death Induction Mechanism of Non-small Cell Lung Cancer Cell Line, NCI-H1703 by Docetaxel
- Treatment of Small Cell Lung Cancer
- Phase II Study of Irinotecan Plus Cisplatin as First Line therapy in Extensive Small-Cell Lung Cancer
- Comparison of Gefitinib versus Docetaxel in Patients with Pre-Treated Non-Small Cell Lung Cancer (NSCLC)
- Successful Treatment of Small-Cell Lung Cancer With Irinotecan in a Hemodialysis Patient With End-Stage Renal Disease