Cancer Res Treat.
2011 Jun;43(2):117-123.
Predictive Value of In Vitro Adenosine Triphosphate-Based Chemotherapy Response Assay in Advanced Gastric Cancer Patients Who Received Oral 5-Fluorouracil after Curative Resection
- Affiliations
-
- 1Department of Internal Medicine, Dong-A University College of Medicine, Busan, Korea.
- 2Department of Surgery, Dong-A University College of Medicine, Busan, Korea.
- 3Department of Pathology and Medical Research Center for Cancer Molecular Therapy, Dong-A University College of Medicine, Busan, Korea.
- 4Section of Gastroenterology, Dong-A University College of Medicine, Busan, Korea. sychoi@dau.ac.kr
Abstract
- PURPOSE
To assess the usefulness of adenosine triphosphate-based chemotherapy response assay (ATP-CRA) results in advanced gastric cancer patients receiving adjuvant chemotherapy.
MATERIALS AND METHODS
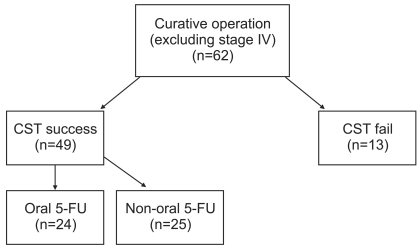
Sixty-two patients underwent curative surgical resection between January, 2006 and December, 2008. Their highly purified surgical specimens were evaluated by ATP-CRAs. Of the 62, 49 had successful assay results and they received either oral 5-fluorouracil or other chemotherapies. We retrospectively analyzed data for 24 patients who were treated with oral 5-fluorouracil and whose assays were successful.
RESULTS
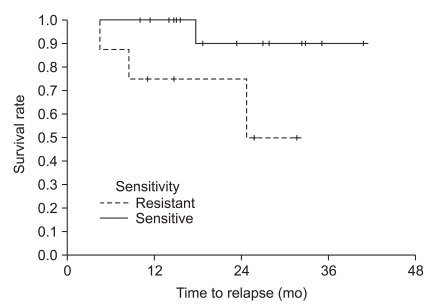
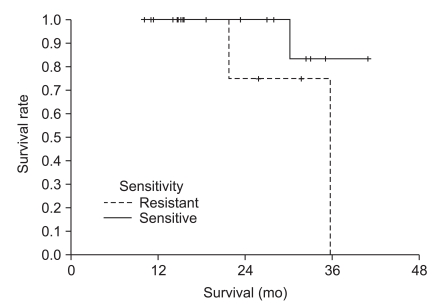
The median observation time was 24.6 months (range, 10.1 to 40.9 months). The median treatment time was 11.2 months (range, 1.2 to 17.7 months). The median age was 66 years (range, 30 to 81 years). Patients were grouped into sensitive- and resistant-groups according to adenosine triphosphate-based chemotherapy response results for fluorouracil. The sensitive-group showed a significantly longer time to relapse (not reached in the sensitive-group vs. 24.8 months in the resistant-group, p=0.043) and longer overall survival compared to the resistant-group (not reached in the sensitive-group vs. 35.7 months in the resistant-group, p=0.16, statistically insignificant).
CONCLUSION
Patients who receive curative surgical resection significantly benefit from sensitive adjuvant chemotherapy according to ATP-CRA results for time to relapse.
Keyword
MeSH Terms
Figure
Reference
-
1. Parkin DM, Pisani P, Ferlay J. Global cancer statistics. CA Cancer J Clin. 1999; 49:33–64. 1PMID: 10200776.
Article2. Roukos DH. Current status and future perspectives in gastric cancer management. Cancer Treat Rev. 2000; 26:243–255. PMID: 10913380.
Article3. Gianni L, Panzini I, Tassinari D, Mianulli AM, Desiderio F, Ravaioli A. Meta-analyses of randomized trials of adjuvant chemotherapy in gastric cancer. Ann Oncol. 2001; 12:1178–1180. PMID: 11583204.
Article4. Mari E, Floriani I, Tinazzi A, Buda A, Belfiglio M, Valentini M, et al. Efficacy of adjuvant chemotherapy after curative resection for gastric cancer: a meta-analysis of published randomised trials. A study of the GISCAD (Gruppo Italiano per lo Studio dei Carcinomi dell'Apparato Digerente). Ann Oncol. 2000; 11:837–843. PMID: 10997811.5. Macdonald JS, Fleming TR, Peterson RF, Berenberg JL, McClure S, Chapman RA, et al. Adjuvant chemotherapy with 5-FU, adriamycin, and mitomycin-C (FAM) versus surgery alone for patients with locally advanced gastric adenocarcinoma: A Southwest Oncology Group study. Ann Surg Oncol. 1995; 2:488–494. PMID: 8591078.
Article6. Nakajima T, Nashimoto A, Kitamura M, Kito T, Iwanaga T, Okabayashi K, et al. Gastric Cancer Surgical Study Group. Adjuvant mitomycin and fluorouracil followed by oral uracil plus tegafur in serosa-negative gastric cancer: a randomised trial. Lancet. 1999; 354:273–277. PMID: 10440302.
Article7. Kubota T. Cancer chemosensitivity test: from laboratory to clinic. Hum Cell. 1995; 8:189–194. PMID: 8721090.8. Kim R, Emi M, Tanabe K, Uchida Y, Toge T. Chemosensitivity testing for gastrointestinal cancer: survival benefit potential and limitations. Anticancer Drugs. 2003; 14:715–723. PMID: 14551504.
Article9. Nakamura R, Saikawa Y, Kubota T, Kumagai A, Kiyota T, Ohashi M, et al. Role of the MTT chemosensitivity test in the prognosis of gastric cancer patients after postoperative adjuvant chemotherapy. Anticancer Res. 2006; 26:1433–1437. PMID: 16619555.10. Kubota T, Egawa T, Otani Y, Furukawa T, Saikawa Y, Yoshida M, et al. Cancer chemotherapy chemosensitivity testing is useful in evaluating the appropriate adjuvant cancer chemotherapy for stages III/IV gastric cancers without peritoneal dissemination. Anticancer Res. 2003; 23:583–587. PMID: 12680150.11. Moon YW, Choi SH, Kim YT, Sohn JH, Chang J, Kim SK, et al. Adenosine triphosphate-based chemotherapy response assay (ATP-CRA)-guided platinum-based 2-drug chemotherapy for unresectable nonsmall-cell lung cancer. Cancer. 2007; 109:1829–1835. PMID: 17366594.
Article12. Han SS, Choi SH, Lee YK, Kim JW, Park NH, Song YS, et al. Predictive value of individualized tumor response testing by ATP-based chemotherapy response assay in ovarian cancer. Cancer Invest. 2008; 26:426–430. PMID: 18443964.
Article13. Woo SU, Bae JW, Kim HG, Choi SH, Kang DH, Lee JB, et al. Correlation between the in vitro ATP-based chemosensitivity assay and HER2/neu expression in women with breast cancer. J Int Med Res. 2007; 35:753–761. PMID: 18034988.
Article14. Shirasaka T, Shimamato Y, Ohshimo H, Yamaguchi M, Kato T, Yonekura K, et al. Development of a novel form of an oral 5-fluorouracil derivative (S-1) directed to the potentiation of the tumor selective cytotoxicity of 5-fluorouracil by two biochemical modulators. Anticancer Drugs. 1996; 7:548–557. PMID: 8862723.
Article15. Rich TA, Shepard RC, Mosley ST. Four decades of continuing innovation with fluorouracil: current and future approaches to fluorouracil chemoradiation therapy. J Clin Oncol. 2004; 22:2214–2232. PMID: 15169811.
Article16. Greene FL, Page DL, Fleming ID, Fritz A, Balch CM, Haller DG, et al. American Joint Committee on Cancer Staging manual. 2002. 6th ed. Philadelphia: Springer.17. Okines AF, Norman AR, McCloud P, Kang YK, Cunningham D. Meta-analysis of the REAL-2 and ML17032 trials: evaluating capecitabine-based combination chemotherapy and infused 5-fluorouracil-based combination chemotherapy for the treatment of advanced oesophago-gastric cancer. Ann Oncol. 2009; 20:1529–1534. PMID: 19474114.
Article18. Ron IG, Lotan A, Inbar MJ, Chaitchik S. Advanced colorectal carcinoma: redefining the role of oral ftorafur. Anticancer Drugs. 1996; 7:649–654. PMID: 8913433.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- In Vitro Adenosine Triphosphate-Based Chemotherapy Response Assay as a Predictor of Clinical Response to Fluorouracil-Based Adjuvant Chemotherapy in Stage II Colorectal Cancer
- The Results of the ATP Based Chemotherapy Response Assay in Gastric Cancer Tissues
- In Vitro Adenosine Triphosphate Based Chemotherapy Response Assay in Gastric Cancer
- A Study on the Chemosensitivity of Advanced Gastric Cancer
- Heterogeneous Chemosensitivity of Breast Cancer Determined by Adeonsine Triphosphate Based Chemotherapy Response Assay