Diagnostic Value of Clinical Parameters in the Prediction of Aspirin-Exacerbated Respiratory Disease in Asthma
- Affiliations
-
- 1Pharmacogenetic Research Center for Psychotropic Drugs, Korea University, Seoul, Korea.
- 2Genome Research Center for Allergy and Respiratory Disease, Soonchunhyang University Bucheon Hospital, Bucheon, Korea. mdcspark@unitel.co.kr
- 3Division of Allergy and Respirtorty Medicine, Soonchunhyang University Seoul Hospital, Seoul, Korea.
- 4Division of Allergy and Respiratory Disease, Soonchunhyang University Cheonan Hospital, Cheonan, Korea.
- KMID: 2167077
- DOI: http://doi.org/10.4168/aair.2011.3.4.256
Abstract
- PURPOSE
Aspirin-exacerbated respiratory disease (AERD) has attracted a great deal of attention because of its association with increased asthma severity. However, oral aspirin challenge (OAC) to diagnose AERD is a time-consuming procedure, and some patients experience serious complications. Thus, we evaluated diagnostic values of non-invasive clinical parameters to predict AERD in asthmatic patients.
METHODS
A total of 836 Korean subjects were recruited from an asthma cohort. They underwent OAC, and clinical parameters including the history of aspirin hypersensitivity, nasal polyposis, and chronic sinusitis of aspirin-tolerant asthma (ATA) and AERD asthmatic patients were compared.
RESULTS
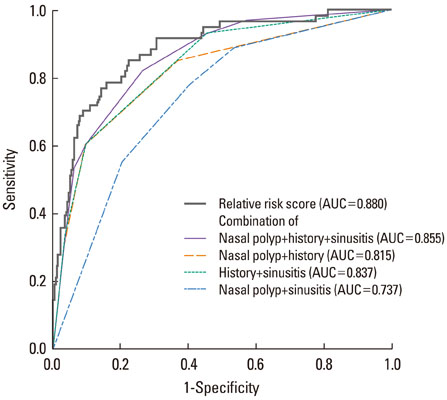
Significant differences (P<0.01) were found in eight parameters: age at diagnosis, body mass index, FEV1%, PC20, history of urticaria, nasal polyps, chronic sinusitis, and history of aspirin hypersensitivity. After logistic regression analysis based on the eight clinical parameters, nasal polyps, history of aspirin intolerance, sinusitis, and log [PC20 methacholine] remained significantly associated with AERD (P<0.05). The sensitivity and specificity of the history of aspirin hypersensitivity to predict AERD were 64.7% and 92.0%, respectively, and the positive and negative predictive values were 56.9% and 94.1%, respectively. Overall, the accuracy of the test was 88.2%. The accuracy of the tests for nasal polyps and chronic sinusitis were 67.3% and 60.4%, respectively.
CONCLUSIONS
Among clinical parameters associated with AERD, the history of aspirin hypersensitivity has the best positive and negative predictive values for the oral aspirin challenge test. Because the false-positive and -negative rates were still high, additional non-invasive methods are needed to reduce the rate of false outcomes.
Keyword
MeSH Terms
Figure
Cited by 3 articles
-
Complementary Participation of Genetics and Epigenetics in Development of NSAID-exacerbated Respiratory Disease
Jong-Uk Lee, Jong Sook Park, Hun Soo Chang, Choon-Sik Park
Allergy Asthma Immunol Res. 2019;11(6):779-794. doi: 10.4168/aair.2019.11.6.779.Clinical Implications of Single Nucleotide Polymorphisms in Diagnosis of Asthma and its Subtypes
Jong-Sook Park, Ji-Hye Son, Choon-Sik Park, Hun Soo Chang
Yonsei Med J. 2019;60(1):1-9. doi: 10.3349/ymj.2019.60.1.1.KAAACI Standardization Committee Report on the procedures and applications of the diagnostic tests for drug allergy
Min-Suk Yang, Ga-Young Ban, Min-Hye Kim, Kyung-Hwan Lim, Hyouk-Soo Kwon, Woo-Jung Song, Jae-Woo Jung, Jeongmin Lee, Dong In Suh, Jae-Woo Kwon, Sae-Hoon Kim, Yoo Seob Shin, Hye-Ryun Kang, Tae-Bum Kim, Byung-Jae Lee, Ai-Young Lee, Hae-Sim Park, Sang-Heon Cho
Allergy Asthma Respir Dis. 2017;5(5):239-247. doi: 10.4168/aard.2017.5.5.239.
Reference
-
1. Stevenson DD, Sanchez-Borges M, Szczeklik A. Classification of allergic and pseudoallergic reactions to drugs that inhibit cyclooxygenase enzymes. Ann Allergy Asthma Immunol. 2001. 87:177–180.2. Mascia K, Haselkorn T, Deniz YM, Miller DP, Bleecker ER, Borish L. Aspirin sensitivity and severity of asthma: evidence for irreversible airway obstruction in patients with severe or difficult-to-treat asthma. J Allergy Clin Immunol. 2005. 116:970–975.3. Marquette CH, Saulnier F, Leroy O, Wallaert B, Chopin C, Demarcq JM, Durocher A, Tonnel AB. Long-term prognosis of near-fatal asthma. A 6-year follow-up study of 145 asthmatic patients who underwent mechanical ventilation for a near-fatal attack of asthma. Am Rev Respir Dis. 1992. 146:76–81.4. Picado C, Castillo JA, Montserrat JM, Agusti-Vidal A. Aspirin-intolerance as a precipitating factor of life-threatening attacks of asthma requiring mechanical ventilation. Eur Respir J. 1989. 2:127–129.5. Gollapudi RR, Teirstein PS, Stevenson DD, Simon RA. Aspirin sensitivity: implications for patients with coronary artery disease. JAMA. 2004. 292:3017–3023.6. Szczeklik A, Nizankowska E, Duplaga M. Natural history of aspirin-induced asthma. AIANE Investigators. European Network on Aspirin-Induced Asthma. Eur Respir J. 2000. 16:432–436.7. Babu KS, Salvi SS. Aspirin and asthma. Chest. 2000. 118:1470–1476.8. Szczeklik A, Stevenson DD. Aspirin-induced asthma: advances in pathogenesis, diagnosis, and management. J Allergy Clin Immunol. 2003. 111:913–921.9. Fahrenholz JM. Natural history and clinical features of aspirin-exacerbated respiratory disease. Clin Rev Allergy Immunol. 2003. 24:113–124.10. Samter M, Beers RF Jr. Concerning the nature of intolerance to aspirin. J Allergy. 1967. 40:281–293.11. Szczeklik A, Nizankowska E. Clinical features and diagnosis of aspirin induced asthma. Thorax. 2000. 55:Suppl 2. S42–S44.12. Global Initiative for Asthma. Global strategy for asthma management and prevention. NHLBI/WHO workshop report. 1995. Bethesda: National Institutes of Health.13. Nizankowska-Mogilnicka E, Bochenek G, Mastalerz L, Swierczyńska M, Picado C, Scadding G, Kowalski ML, Setkowicz M, Ring J, Brockow K, Bachert C, Wohrl S, Dahlén B, Szczeklik A. EAACI/GA2LEN guideline: aspirin provocation tests for diagnosis of aspirin hypersensitivity. Allergy. 2007. 62:1111–1118.14. Kim BS, Park SM, Uhm TG, Kang JH, Park JS, Jang AS, Uh ST, Kim MK, Choi IS, Cho SH, Hong CS, Lee YW, Lee JY, Choi BW, Park HS, Park BL, Shin HD, Chung IY, Park CS. Effect of single nucleotide polymorphisms within the interleukin-4 promoter on aspirin intolerance in asthmatics and interleukin-4 promoter activity. Pharmacogenet Genomics. 2010. 20:748–758.15. Hedman J, Kaprio J, Poussa T, Nieminen MM. Prevalence of asthma, aspirin intolerance, nasal polyposis and chronic obstructive pulmonary disease in a population-based study. Int J Epidemiol. 1999. 28:717–722.16. Kalyoncu AF, Karakaya G, Sahin AA, Bariş YI. Occurrence of allergic conditions in asthmatics with analgesic intolerance. Allergy. 1999. 54:428–435.17. Settipane GA, Chafee FH, Klein DE. Aspirin intolerance. II. A prospective study in an atopic and normal population. J Allergy Clin Immunol. 1974. 53:200–204.18. Stevenson DD. Diagnosis, prevention, and treatment of adverse reactions to aspirin and nonsteroidal anti-inflammatory drugs. J Allergy Clin Immunol. 1984. 74:617–622.19. Vally H, Taylor ML, Thompson PJ. The prevalence of aspirin intolerant asthma (AIA) in Australian asthmatic patients. Thorax. 2002. 57:569–574.20. Jenkins C, Costello J, Hodge L. Systematic review of prevalence of aspirin induced asthma and its implications for clinical practice. BMJ. 2004. 328:434.21. Settipane RA, Stevenson DD. Cross sensitivity with acetaminophen in aspirin-sensitive subjects with asthma. J Allergy Clin Immunol. 1989. 84:26–33.22. De Weck AL, Sanz ML, Gamboa PM, Jermann JM, Kowalski M, Medrala W, Sainte-Laudy J, Schneider MS, Weber JM, Wolanczyk-Medrala A. Nonsteroidal anti-inflammatory drug hypersensitivity syndrome: a multicenter study. II. Basophil activation by nonsteroidal anti-inflammatory drugs and its impact on pathogenesis. J Investig Allergol Clin Immunol. 2010. 20:39–57.23. Devouassoux G, Pachot A, Laforest L, Diasparra J, Freymond N, Van Ganse E, Mougin B, Pacheco Y. Galectin-10 mRNA is overexpressed in peripheral blood of aspirin-induced asthma. Allergy. 2008. 63:125–131.24. Min JW, Jang AS, Park SM, Lee SH, Lee JH, Park SW, Park CS. Comparison of plasma eotaxin family level in aspirin-induced and aspirin-tolerant asthma patients. Chest. 2005. 128:3127–3132.25. Lee SH, Rhim T, Choi YS, Min JW, Kim SH, Cho SY, Paik YK, Park CS. Complement C3a and C4a increased in plasma of patients with aspirin-induced asthma. Am J Respir Crit Care Med. 2006. 173:370–378.26. Sanak M, Kielbasa B, Bochenek G, Szczeklik A. Exhaled eicosanoids following oral aspirin challenge in asthmatic patients. Clin Exp Allergy. 2004. 34:1899–1904.27. Choi JH, Park HS, Oh HB, Lee JH, Suh YJ, Park CS, Shin HD. Leukotriene-related gene polymorphisms in ASA-intolerant asthma: an association with a haplotype of 5-lipoxygenase. Hum Genet. 2004. 114:337–344.28. Kim SH, Choi JH, Park HS, Holloway JW, Lee SK, Park CS, Shin HD. Association of thromboxane A2 receptor gene polymorphism with the phenotype of acetyl salicylic acid-intolerant asthma. Clin Exp Allergy. 2005. 35:585–590.29. Park BL, Park SM, Park JS, Uh ST, Choi JS, Kim YH, Kim MK, Choi IS, Choi BW, Cho SH, Hong CS, Lee YW, Lee JY, Park CS, Shin HD. Association of PTGER gene family polymorphisms with aspirin intolerant asthma in Korean asthmatics. BMB Rep. 2010. 43:445–449.30. Park JS, Chang HS, Park CS, Lee JH, Lee YM, Choi JH, Park HS, Kim LH, Park BL, Choi YH, Shin HD. Association analysis of cysteinyl-leukotriene receptor 2 (CYSLTR2) polymorphisms with aspirin intolerance in asthmatics. Pharmacogenet Genomics. 2005. 15:483–492.31. Kim TH, Chang HS, Park SM, Nam BY, Park JS, Rhim T, Park HS, Kim MK, Choi IS, Cho SH, Chung IY, Park BL, Park CS, Shin HD. Association of angiotensin I-converting enzyme gene polymorphisms with aspirin intolerance in asthmatics. Clin Exp Allergy. 2008. 38:1727–1737.32. Oh SH, Park SM, Park JS, Jang AS, Lee YM, Uh ST, Kim YH, Choi IS, Kim MK, Park BL, Shin HD, Park CS. Association analysis of peroxisome proliferator-activated receptors gamma gene polymorphisms with asprin hypersensitivity in asthmatics. Allergy Asthma Immunol Res. 2009. 1:30–35.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Update on the Management of Aspirin-Exacerbated Respiratory Disease
- Aspirin-Exacerbated Respiratory Disease: Evaluation and Management
- Update on Recent Advances in the Management of Aspirin Exacerbated Respiratory Disease
- Unraveling the Genetic Basis of Aspirin Hypersensitivity in Asthma Beyond Arachidonate Pathways
- Aspirin induces IL-4 production: augmented IL-4 production in aspirin-exacerbated respiratory disease