Yonsei Med J.
2009 Dec;50(6):744-750. 10.3349/ymj.2009.50.6.744.
Update on Recent Advances in the Management of Aspirin Exacerbated Respiratory Disease
- Affiliations
-
- 1Department of Allergy and Rheumatology, Ajou University School of Medicine, Suwon, Korea. hspark@ajou.ac.kr
- KMID: 1777085
- DOI: http://doi.org/10.3349/ymj.2009.50.6.744
Abstract
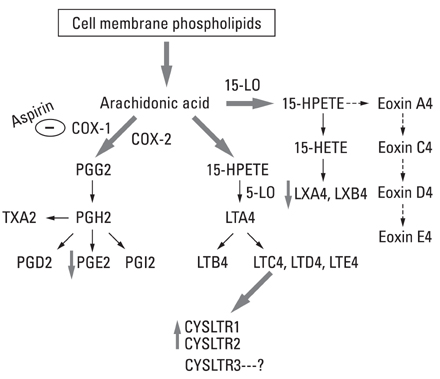
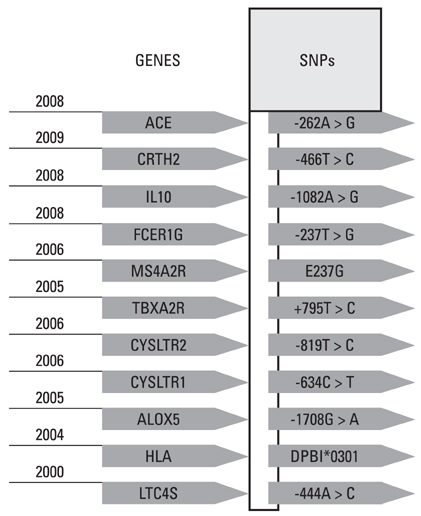
- Aspirin intolerant asthma (AIA) is frequently characterized as an aspirin (ASA)-exacerbated respiratory disease (AERD). It is a clinical syndrome associated with chronic severe inflammation in the upper and lower airways resulting in chronic rhinitis, sinusitis, recurrent polyposis, and asthma. AERD generally develops secondary to abnormalities in inflammatory mediators and arachidonic acid biosynthesis expression. Upper and lower airway eosinophil infiltration is a key feature of AERD; however, the exact mechanisms of such chronic eosinophilic inflammation are not fully understood. Cysteinyl leukotriene over-production may be a key factor in the induction of eosinophilic activation. Genetic studies have suggested a role for variability of genes in disease susceptibility and response to medication. Potential genetic biomarkers contributing to the AERD phenotype include HLA-DPB1* 301, LTC4S, ALOX5, CYSLT, PGE2, TBXA2R, TBX21, MS4A2, IL10 -1082A > G, ACE -262A > T, and CRTH2 -466T > C; the four-locus SNP set was composed of B2ADR 46A > G, CCR3 -520T > G, CysLTR1 -634C > T, and FCER1B -109T > C. Management of AERD is an important issue. Aspirin ingestion may result in significant morbidity and mortality, and patients must be advised regarding aspirin risk. Leukotriene receptor antagonists (LTRA) that inhibit leukotriene pathways have an established role in long-term AERD management and rhinosinusitis. Aspirin desensitization may be required for the relief of upper and lower airway symptoms in AERD patients. Future research should focus on identification of biomarkers for a comprehensive diagnostic approach.
Keyword
MeSH Terms
Figure
Cited by 1 articles
-
Samter’s Triad: State of the Art
Sung-Dong Kim, Kyu-Sup Cho
Clin Exp Otorhinolaryngol. 2018;11(2):71-80. doi: 10.21053/ceo.2017.01606.
Reference
-
1. Yoshida S, Ishizaki Y, Onuma K, Shoji T, Nakagawa H, Amayasu H. Selective cyclo-oxygenase 2 inhibitor in patients with aspirin-induced asthma. J Allergy Clin Immunol. 2000. 106:1201–1202.
Article2. Szczeklik A, Nizankowska E, Duplaga M. Natural history of aspirin-induced asthma. AIANE investigators. European Network on Aspirin-Induced Asthma. Eur Respir J. 2000. 16:432–436.3. Hedman J, Kaprio J, Poussa T, Nieminen MM. Prevalence of asthma, aspirin intolerance, nasal polyposis and chronic obstructive pulmonary disease in a population-based study. Int J Epidemiol. 1999. 28:717–722.
Article4. Vally H, Taylor ML, Thompson PJ. The prevalence of aspirin intolerant asthma (AIA) in Australian asthmatic patients. Thorax. 2002. 57:569–574.
Article5. Mascia K, Haselkorn T, Deniz YM, Miller DP, Bleecker ER, Borish L. TENOR Study Group. Aspirin sensitivity and severity of asthma: evidence for irreversible airway obstruction in patients with severe or difficult-to-treat asthma. J Allergy Clin Immunol. 2005. 116:970–975.6. Palikhe NS, Kim SH, Cho BY, Ye YM, Hur GY, Park HS. Association of three sets of high-affinity IgE receptor (FcepsilonR1) polymorphisms with aspirin-intolerant asthma. Respir Med. 2008. 102:1132–1139.
Article7. Sladek K, Szczeklik A. Cysteinyl leukotrienes overproduction and mast cell activation in aspirin-provoked bronchospasm in asthma. Eur Respir J. 1993. 6:391–399.8. Park HS. Early and late onset asthmatic responses following lysine-aspirin inhalation in aspirin-sensitive asthmatic patients. Clin Exp Allergy. 1995. 25:38–40.
Article9. Kowalski ML, Ptasinska A, Jedrzejczak M, Bienkiewicz B, Cieslak M, Grzegorczyk J, et al. Aspirin-triggered 15-HETE generation in peripheral blood leukocytes is a specific and sensitive Aspirin-Sensitive Patients Identification Test (ASPITest). Allergy. 2005. 60:1139–1145.
Article10. Gaber F, Daham K, Higashi A, Higashi N, Gülich A, Delin I, et al. Increased levels of cysteinyl-leukotrienes in saliva, induced sputum, urine and blood from patients with aspirin-intolerant asthma. Thorax. 2008. 63:1076–1082.
Article11. Hamad AM, Sutcliffe AM, Knox AJ. Aspirin-induced asthma: clinical aspects, pathogenesis and management. Drugs. 2004. 64:2417–2432.12. Lee TH, Woszczek G, Farooque SP. Leukotriene (E4): perspective on the forgotten mediator. J Allergy Clin Immunol. 2009. 124:417–421.
Article13. Kupczyk M, Antczak A, Kuprys-Lipinska I, Kuna P. Lipoxin A4 generation is decreased in aspirin-sensitive patients in lysine-aspirin nasal challenge in vivo model. Allergy. 2009. 64:1746–1752.
Article14. Hui Y, Funk CD. Cysteinyl leukotriene receptors. Biochem Pharmacol. 2002. 64:1549–1557.
Article15. Capra V. Molecular and functional aspects of human cysteinyl leukotriene receptors. Pharmacol Res. 2004. 50:1–11.
Article16. Ciana P, Fumagalli M, Trincavelli ML, Verderio C, Rosa P, Lecca D, et al. The orphan receptor GPR17 identified as a new dual uracil nucleotides/cysteinyl-leukotrienes receptor. EMBO J. 2006. 25:4615–4627.
Article17. Peters-Golden M, Henderson WR Jr. Leukotrienes. N Engl J Med. 2007. 357:1841–1854.
Article18. Sousa AR, Parikh A, Scadding G, Corrigan CJ, Lee TH. Leukotriene-receptor expression on nasal mucosal inflammatory cells in aspirin-sensitive rhinosinusitis. N Engl J Med. 2002. 347:1493–1499.
Article19. Szczeklik A, Nizankowska E, Bochenek G, Nagraba K, Mejza F, Swierczynska M. Safety of a specific COX-2 inhibitor in aspirin-induced asthma. Clin Exp Allergy. 2001. 31:219–225.
Article20. Picado C, Fernandez-Morata JC, Juan M, Roca-Ferrer J, Fuentes M, Xaubet A, et al. Cyclooxygenase-2 mRNA is downexpressed in nasal polyps from aspirin-sensitive asthmatics. Am J Respir Crit Care Med. 1999. 160:291–296.
Article21. Morgan T, Bajrovic N, Silar M, Kosnik M, Korosec P. Aspirin-induced COX-2 overexpression in monocytes of aspirin-intolerant patients. Int Arch Allergy Immunol. 2009. 149:378–384.
Article22. Picado C, Castillo JA, Montserrat JM, Agusti-Vidal A. Aspirin-intolerance as a precipitating factor of life-threatening attacks of asthma requiring mechanical ventilation. Eur Respir J. 1989. 2:127–129.23. Kowalski ML, Pawliczak R, Wozniak J, Siuda K, Poniatowska M, Iwaszkiewicz J, et al. Differential metabolism of arachidonic acid in nasal polyp epithelial cells cultured from aspirin-sensitive and aspirin-tolerant patients. Am J Respir Crit Care Med. 2000. 161:391–398.
Article24. Chambers LS, Black JL, Ge Q, Carlin SM, Au WW, Poniris M, et al. PAR-2 activation, PGE2, and COX-2 in human asthmatic and nonasthmatic airway smooth muscle cells. Am J Physiol Lung Cell Mol Physiol. 2003. 285:L619–L627.25. Taniguchi M, Higashi N, Ono E, Mita H, Akiyama K. Hyperleukotrieneuria in patients with allergic and inflammatory disease. Allergol Int. 2008. 57:313–320.
Article26. Palikhe NS, Kim SH, Cho BY, Ye YM, Choi GS, Park HS. Genetic variability in CRTH2 polymorphism increases eotaxin-2 levels in patients with aspirin exacerbated respiratory disease. Allergy. 2009. 10. 01. [Epub ahead of print].27. Szczeklik A, Sladek K, Dworski R, Nizankowska E, Soja J, Sheller J, et al. J Bronchial aspirin challenge causes specific eicosanoid response in aspirin-sensitive asthmatics. Am J Respir Crit Care Med. 1996. 154:1608–1614.
Article28. Novel mechanisms involving new and old eicosanoids in bronchial inflammation. 2009. In : XXVII Congress of the European Academy of Allergy and Clinical Immunology; June 6-10; Warsaw.29. Feltenmark S, Gautam N, Brunnström A, Griffiths W, Backman L, Edenius C, et al. Eoxins are proinflammatory arachidonic acid metabolites produced via the 15-lipoxygenase-1 pathway in human eosinophils and mast cells. Proc Natl Acad Sci U S A. 2008. 105:680–685.
Article30. Lee JY, Kim HM, Ye YM, Bahn JW, Suh CH, Nahm D, et al. Role of staphylococcal superantigen-specific IGE antibodies in aspirin-intolerant asthma. Allergy Asthma Proc. 2006. 27:341–346.
Article31. Suh YJ, Yoon SH, Sampson AP, Kim HJ, Kim SH, Nahm DH, et al. Specific immunoglobulin E for staphylococcal enterotoxins in nasal polyps from patients with aspirin-intolerant asthma. Clin Exp Allergy. 2004. 34:1270–1275.
Article32. Szczeklik A. Aspirin-induced asthma as a viral disease. Clin Allergy. 1988. 18:15–20.
Article33. Okada H, Ohnishi T, Hirashima M, Fujita J, Yamaji Y, Takahara J, et al. Anti-asthma effect of an antiviral drug, acyclovir: a clinical case and experimental study. Clin Exp Allergy. 1997. 27:431–437.
Article34. Szczeklik A, Schmitz-Schumann M, Nizankowska E, Milewski M, Roehlig F, Virchow C. Altered distribution of IgG subclasses in aspirin-induced asthma: high IgG4, low IgG1. Clin Exp Allergy. 1992. 22:283–287.
Article35. Choi JH, Lee KW, Oh HB, Lee KJ, Suh YJ, Park CS, et al. HLA association in aspirin-intolerant asthma: DPB1*0301 as a strong marker in a Korean population. J Allergy Clin Immunol. 2004. 113:562–564.36. Sanak M, Pierzchalska M, Bazan-Socha S, Szczeklik A. Enhanced expression of the leukotriene C (4) synthase due to overactive transcription of an allelic variant associated with aspirin-intolerant asthma. Am J Respir Cell Mol Biol. 2000. 23:290–296.
Article37. Van Sambeek , Stevenson DD, Baldasaro M, Lam BK, Zhao J, Yoshida S, et al. 5' flanking region polymorphism of the gene encoding leukotriene C4 synthase does not correlate with the aspirin-intolerant asthma phenotype in the United States. J Allergy Clin Immunol. 2000. 106:72–76.
Article38. Choi JH, Kim SH, Bae JS, Yu HL, Suh CH, Nahm DH, et al. Lack of an association between a newly identified promoter polymorphism (-1702G > A) of the leukotriene C4 synthase gene and aspirin-intolerant asthma in a Korean population. Tohoku J Exp Med. 2006. 208:49–56.
Article39. Sayers I, Barton S, Rorke S, Sawyer J, Peng Q, Beghé B, et al. Promoter polymorphism in the 5-lipoxygenase (ALOX5) and 5-lipoxygenase-activating protein (ALOX5AP) genes and asthma susceptibility in a caucasian population. Clin Exp Allergy. 2003. 33:1103–1110.
Article40. Kim SH, Bae JS, Suh CH, Nahm DH, Holloway JW, Park HS. Polymorphism of tandem repeat in promoter of 5-lipoxygenase in ASA-intolerant asthma: a positive association with airway hyperresponsiveness. Allergy. 2005. 60:760–765.
Article41. Kim SH, Oh JM, Kim YS, Palmer LJ, Suh CH, Nahm DH, et al. Cysteinyl leukotriene receptor 1 promoter polymorphism is associated with aspirin-intolerant asthma in males. Clin Exp Allergy. 2006. 36:433–439.
Article42. Kim SH, Ye YM, Hur GY, Lee SK, Sampson AP, Lee HY, et al. CysLTR1 promoter polymorphism and requirement for leukotriene receptor antagonist in aspirin-intolerant asthma patients. Pharmacogenomics. 2007. 8:1143–1150.
Article43. Park JS, Chang HS, Park CS, Lee JH, Lee YM, Choi JH, et al. Association analysis of cysteinyl-leukotriene receptor 2 (CYSLTR2) polymorphisms with aspirin intolerance in asthmatics. Pharmacogenet Genomics. 2005. 15:483–492.
Article44. Kim SH, Bae JS, Holloway JW, Lee JT, Suh CH, Nahm DH, et al. A polymorphism of MS4A2 (-109T > C) encoding the beta-chain of the high-affinity immunoglobulin E receptor (FcepsilonR1beta) is associated with a susceptibility to aspirin-intolerant asthma. Clin Exp Allergy. 2006. 36:877–883.
Article45. Kim SH, Yang EM, Lee HN, Cho BY, Ye YM, Park HS. Combined effect of IL-10 and TGF-beta1 promoter polymorphisms as a risk factor for aspirin-intolerant asthma and rhinosinusitis. Allergy. 2009. 64:1221–1225.
Article46. Kim SH, Choi JH, Park HS, Holloway JW, Lee SK, Park CS, et al. Association of thromboxane A2 receptor gene polymorphism with the phenotype of acetyl salicylic acid-intolerant asthma. Clin Exp Allergy. 2005. 35:585–590.
Article47. Akahoshi M, Obara K, Hirota T, Matsuda A, Hasegawa K, Takahashi N, et al. Functional promoter polymorphism in the TBX21 gene associated with aspirin-induced asthma. Hum Genet. 2005. 117:16–26.
Article48. Kim SH, Kim YK, Park HW, Jee YK, Kim SH, Bahn JW, et al. Association between polymorphisms in prostanoid receptor genes and aspirin-intolerant asthma. Pharmacogenet Genomics. 2007. 17:295–304.
Article49. Kim TH, Chang HS, Park SM, Nam BY, Park JS, Rhim T, et al. Association of angiotensin I-converting enzyme gene polymorphisms with aspirin intolerance in asthmatics. Clin Exp Allergy. 2008. 38:1727–1737.
Article50. Kim SH, Jeong HH, Cho BY, Kim M, Lee HY, Lee J, et al. Association of four-locus gene interaction with aspirin-intolerant asthma in Korean asthmatics. J Clin Immunol. 2008. 28:336–342.
Article51. Soto-Quiros ME. Practical handling of the asthma based on the control of the disease: Update of GINA guidelines. Anales de Paediatr. 2008. 68:317–319. http://www.elsevier.es/revistas/ctl_servlet?_f=7064&articuloid=13117700.52. Obase Y, Shimoda T, Tomari SY, Mitsuta K, Kawano T, Matsuse H, et al. Effects of pranlukast on chemical mediators in induced sputum on provocation tests in atopic and aspirin-intolerant asthmatic patients. Chest. 2002. 121:143–150.
Article53. Israel E. The protective effects of leukotriene modifiers in aspirin-induced asthma. Postgrad Med. 2000. 108:40–44.54. Mygind N, Laursen LC, Dahl M. Systemic corticosteroid treatment for seasonal allergic rhinitis: a common but poorly documented therapy. Allergy. 2000. 55:11–15.
Article55. Berges-Gimeno MP, Simon RA, Stevenson DD. The effect of leukotriene-modifier drugs on aspirin-induced asthma and rhinitis reactions. Clin Exp Allergy. 2002. 32:1491–1496.
Article56. Nayak A. A review of montelukast in the treatment of asthma and allergic rhinitis. Expert Opin Pharmacother. 2004. 5:679–686.
Article57. Lee JY, Simon RA, Stevenson DD. Selection of aspirin dosages for aspirin desensitization treatment in patients with aspirin-exacerbated respiratory disease. J Allergy Clin Immunol. 2007. 119:157–164.
Article58. Juergens UR, Christiansen SC, Stevenson DD, Zuraw BL. Inhibition of monocyte leukotriene B4 production after aspirin desensitization. J Allergy Clin Immunol. 1995. 96:148–156.
Article59. Arm JP, Austen KF. Leukotriene receptors and aspirin sensitivity. N Engl J Med. 2002. 347:1524–1526.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Update on the Management of Aspirin-Exacerbated Respiratory Disease
- Aspirin-Exacerbated Respiratory Disease: Evaluation and Management
- Diagnostic Value of Clinical Parameters in the Prediction of Aspirin-Exacerbated Respiratory Disease in Asthma
- Which Factors Associated With Activated Eosinophils Contribute to the Pathogenesis of Aspirin-Exacerbated Respiratory Disease?
- Aspirin induces IL-4 production: augmented IL-4 production in aspirin-exacerbated respiratory disease