Aspirin-Exacerbated Respiratory Disease: Evaluation and Management
- Affiliations
-
- 1Division of Allergy, Asthma & Immunology, Naval Medical Center Portsmouth, Portsmouth, VA, USA.
- 2Division of Allergy, Asthma & Immunology, Scripps Clinic, San Diego, CA, USA. DStevemd@aol.com
- KMID: 2260371
- DOI: http://doi.org/10.4168/aair.2011.3.1.3
Abstract
- The clinical syndrome of aspirin-exacerbated respiratory disease (AERD) is a condition where inhibition of cyclooxygenase-1 (COX-1) induces attacks of upper and lower airway reactions, including rhinorrhea and varying degrees of bronchospasm and laryngospasm. Although the reaction is not IgE-mediated, patients can also present with anaphylactic hypersensitivity reactions, including hypotension, after exposure to COX-1 inhibiting drugs. All patients with AERD have underlying nasal polyps and intractable sinus disease which may be difficult to treat with standard medical and surgical interventions. This review article focuses on the management of AERD patients with a particular emphasis on aspirin desensitization and continuous treatment with aspirin.
Keyword
MeSH Terms
Figure
Cited by 4 articles
-
Association of TLR3 gene polymorphism with IgG subclass deficiency and the severity in patients with aspirin-intolerant asthma
Seung-Hyun Kim, Eun-Mi Yang, Hye-Min Jung, Duy Le Pham, Hyun-Na Choi, Ga-Young Ban, Hae-Sim Park
Allergy Asthma Respir Dis. 2016;4(4):264-270. doi: 10.4168/aard.2016.4.4.264.Successful sequential desensitization in a patient with drug hypersensitivity to three kinds of antiplatelet agents
Gun-Woo Kim, Sung-Yoon Kang, Kyoung-Hee Sohn, Sae-Hoon Kim, Sang-Heon Cho, Kyung-Up Min, Yoon-Seok Chang
Allergy Asthma Respir Dis. 2016;4(5):374-377. doi: 10.4168/aard.2016.4.5.374.WDR46 is a Genetic Risk Factor for Aspirin-Exacerbated Respiratory Disease in a Korean Population
Charisse Flerida A. Pasaje, Joon Seol Bae, Byung-Lae Park, Hyun Sub Cheong, Jeong-Hyun Kim, Soo-Taek Uh, Choon-Sik Park, Hyoung Doo Shin
Allergy Asthma Immunol Res. 2012;4(4):199-205. doi: 10.4168/aair.2012.4.4.199.Samter’s Triad: State of the Art
Sung-Dong Kim, Kyu-Sup Cho
Clin Exp Otorhinolaryngol. 2018;11(2):71-80. doi: 10.21053/ceo.2017.01606.
Reference
-
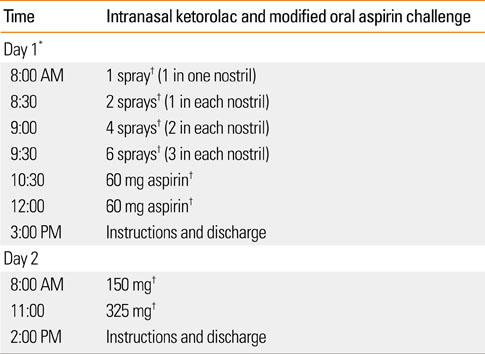
1. Szczeklik A, Nizankowska-Mogilnicka E, Sanak M. Adkinson NF, Bochner BS, Busse WW, Holgate ST, Lemanske RF, Simons FER, editors. Hypersensitivity to aspirin and non-steroidal anti-inflammatory drugs. Middleton's allergy: principles and practice. 2009. 7th ed. New York: Mosby;1227–1240.2. Stevenson DD. Aspirin sensitivity and desensitization for asthma and sinusitis. Curr Allergy Asthma Rep. 2009. 9:155–163.3. Jenkins C, Costello J, Hodge L. Systematic review of prevalence of aspirin induced asthma and its implications for clinical practice. BMJ. 2004. 328:434.4. McDonald JR, Mathison DA, Stevenson DD. Aspirin intolerance in asthma. Detection by oral challenge. J Allergy Clin Immunol. 1972. 50:198–207.5. Weber RW, Hoffman M, Raine DA Jr, Nelson HS. Incidence of bronchoconstriction due to aspirin, azo dyes, non-azo dyes, and preservatives in a population of perennial asthmatics. J Allergy Clin Immunol. 1979. 64:32–37.6. Delaney JC. The diagnosis of aspirin idiosyncrasy by analgesic challenge. Clin Allergy. 1976. 6:177–181.7. Berges-Gimeno MP, Simon RA, Stevenson DD. The natural history and clinical characteristics of aspirin-exacerbated respiratory disease. Ann Allergy Asthma Immunol. 2002. 89:474–478.8. Szczeklik A, Nizankowska E, Duplaga M. AIANE Investigators. European Network on Aspirin-Induced Asthma. Natural history of aspirin-induced asthma. Eur Respir J. 2000. 16:432–436.9. Samter M, Beers RF Jr. Intolerance to aspirin. Clinical studies and consideration of its pathogenesis. Ann Intern Med. 1968. 68:975–983.10. Widal MF, Abrami P, Lermeyez J. Idiosyncratic anaphylaxis. Presse Med. 1922. 30:189–192.11. Stevenson DD, Szczeklik A. Clinical and pathologic perspectives on aspirin sensitivity and asthma. J Allergy Clin Immunol. 2006. 118:773–786.12. Mascia K, Borish L, Patrie J, Hunt J, Phillips CD, Steinke JW. Chronic hyperplastic eosinophilic sinusitis as a predictor of aspirin-exacerbated respiratory disease. Ann Allergy Asthma Immunol. 2005. 94:652–657.13. Stevenson DD. Aspirin desensitization in patients with AERD. Clin Rev Allergy Immunol. 2003. 24:159–168.14. Pfaar O, Klimek L. Eicosanoids, aspirin-intolerance and the upper airways--current standards and recent improvements of the desensitization therapy. J Physiol Pharmacol. 2006. 57:Suppl 12. 5–13.15. Milewski M, Mastalerz L, Nizankowska E, Szczeklik A. Nasal provocation test with lysine-aspirin for diagnosis of aspirin-sensitive asthma. J Allergy Clin Immunol. 1998. 101:581–586.16. Nizankowska E, Bestynska-Krypel A, Cmiel A, Szczeklik A. Oral and bronchial provocation tests with aspirin for diagnosis of aspirin-induced asthma. Eur Respir J. 2000. 15:863–869.17. Parikh AA, Scadding GK. Intranasal lysine-aspirin in aspirin-sensitive nasal polyposis: a controlled trial. Laryngoscope. 2005. 115:1385–1390.18. Dursun AB, Woessner KA, Simon RA, Karasoy D, Stevenson DD. Predicting outcomes of oral aspirin challenges in patients with asthma, nasal polyps, and chronic sinusitis. Ann Allergy Asthma Immunol. 2008. 100:420–425.19. Gyllfors P, Bochenek G, Overholt J, Drupka D, Kumlin M, Sheller J, Nizankowska E, Isakson PC, Mejza F, Lefkowith JB, Dahlén SE, Szczeklik A, Murray JJ, Dahlen B. Biochemical and clinical evidence that aspirin-intolerant asthmatic subjects tolerate the cyclooxygenase 2-selective analgetic drug celecoxib. J Allergy Clin Immunol. 2003. 111:1116–1121.20. Woessner KM, Simon RA, Stevenson DD. The safety of celecoxib in patients with aspirin-sensitive asthma. Arthritis Rheum. 2002. 46:2201–2206.21. Stevenson DD, Simon RA. Lack of cross-reactivity between rofecoxib and aspirin in aspirin-sensitive patients with asthma. J Allergy Clin Immunol. 2001. 108:47–51.22. Berges-Gimeno MP, Simon RA, Stevenson DD. Long-term treatment with aspirin desensitization in asthmatic patients with aspirin-exacerbated respiratory disease. J Allergy Clin Immunol. 2003. 111:180–186.23. Stevenson DD, Hankammer MA, Mathison DA, Christiansen SC, Simon RA. Aspirin desensitization treatment of aspirin-sensitive patients with rhinosinusitis-asthma: long-term outcomes. J Allergy Clin Immunol. 1996. 98:751–758.24. Kowalski ML, Grzelewska-Rzymowska I, Szmidt M, Rozniecki J. Clinical efficacy of aspirin in "desensitised" aspirin-sensitive asthmatics. Eur J Respir Dis. 1986. 69:219–225.25. Klimek L, Pfaar O. Aspirin intolerance: does desensitization alter the course of the disease? Immunol Allergy Clin North Am. 2009. 29:669–675.26. McMains KC, Kountakis SE. Medical and surgical considerations in patients with Samter's triad. Am J Rhinol. 2006. 20:573–576.27. Berges-Gimeno MP, Simon RA, Stevenson DD. Early effects of aspirin desensitization treatment in asthmatic patients with aspirin-exacerbated respiratory disease. Ann Allergy Asthma Immunol. 2003. 90:338–341.28. Shaker M, Lobb A, Jenkins P, O'Rourke D, Takemoto SK, Sheth S, Burroughs T, Dykewicz MS. An economic analysis of aspirin desensitization in aspirin-exacerbated respiratory disease. J Allergy Clin Immunol. 2008. 121:81–87.29. Stevenson DD, Simon RA. Selection of patients for aspirin desensitization treatment. J Allergy Clin Immunol. 2006. 118:801–804.30. Macy E, Bernstein JA, Castells MC, Gawchik SM, Lee TH, Settipane RA, Simon RA, Wald J, Woessner KM. Aspirin challenge and desensitization for aspirin-exacerbated respiratory disease: a practice paper. Ann Allergy Asthma Immunol. 2007. 98:172–174.31. Daffern PJ, Muilenburg D, Hugli TE, Stevenson DD. Association of urinary leukotriene E4 excretion during aspirin challenges with severity of respiratory responses. J Allergy Clin Immunol. 1999. 104:559–564.32. Israel E, Fischer AR, Rosenberg MA, Lilly CM, Callery JC, Shapiro J, Cohn J, Rubin P, Drazen JM. The pivotal role of 5-lipoxygenase products in the reaction of aspirin-sensitive asthmatics to aspirin. Am Rev Respir Dis. 1993. 148:1447–1451.33. Sousa AR, Parikh A, Scadding G, Corrigan CJ, Lee TH. Leukotriene-receptor expression on nasal mucosal inflammatory cells in aspirin-sensitive rhinosinusitis. N Engl J Med. 2002. 347:1493–1499.34. Christie PE, Tagari P, Ford-Hutchinson AW, Charlesson S, Chee P, Arm JP, Lee TH. Urinary leukotriene E4 concentrations increase after aspirin challenge in aspirin-sensitive asthmatic subjects. Am Rev Respir Dis. 1991. 143:1025–1029.35. Berges-Gimeno MP, Simon RA, Stevenson DD. The effect of leukotriene-modifier drugs on aspirin-induced asthma and rhinitis reactions. Clin Exp Allergy. 2002. 32:1491–1496.36. Dahlén SE, Malmström K, Nizankowska E, Dahlén B, Kuna P, Kowalski M, Lumry WR, Picado C, Stevenson DD, Bousquet J, Pauwels R, Holgate ST, Shahane A, Zhang J, Reiss TF, Szczeklik A. Improvement of aspirin-intolerant asthma by montelukast, a leukotriene antagonist: a randomized, double-blind, placebo-controlled trial. Am J Respir Crit Care Med. 2002. 165:9–14.37. White A, Ludington E, Mehra P, Stevenson DD, Simon RA. Effect of leukotriene modifier drugs on the safety of oral aspirin challenges. Ann Allergy Asthma Immunol. 2006. 97:688–693.38. White AA, Stevenson DD, Simon RA. The blocking effect of essential controller medications during aspirin challenges in patients with aspirin-exacerbated respiratory disease. Ann Allergy Asthma Immunol. 2005. 95:330–335.39. Williams AN, Simon RA, Woessner KM, Stevenson DD. The relationship between historical aspirin-induced asthma and severity of asthma induced during oral aspirin challenges. J Allergy Clin Immunol. 2007. 120:273–277.40. Hope AP, Woessner KA, Simon RA, Stevenson DD. Rational approach to aspirin dosing during oral challenges and desensitization of patients with aspirin-exacerbated respiratory disease. J Allergy Clin Immunol. 2009. 123:406–410.41. Lee JY, Simon RA, Stevenson DD. Selection of aspirin dosages for aspirin desensitization treatment in patients with aspirin-exacerbated respiratory disease. J Allergy Clin Immunol. 2007. 119:157–164.42. Rozsasi A, Polzehl D, Deutschle T, Smith E, Wiesmiller K, Riechelmann H, Keck T. Long-term treatment with aspirin desensitization: a prospective clinical trial comparing 100 and 300 mg aspirin daily. Allergy. 2008. 63:1228–1234.43. Lee RU, White D, Ding D, Durson AB, Woessner KM, Simon RA, Stevenson DD. Intranasal ketorolac and modified oral aspirin challenge enhances desensitization in aspirin exacerbated respiratory disease. Ann Allergy Asthma Immunol. 2010. 105:130–135.44. White A, Bigby T, Stevenson D. Intranasal ketorolac challenge for the diagnosis of aspirin-exacerbated respiratory disease. Ann Allergy Asthma Immunol. 2006. 97:190–195.45. Casadevall J, Ventura PJ, Mullol J, Picado C. Intranasal challenge with aspirin in the diagnosis of aspirin intolerant asthma: evaluation of nasal response by acoustic rhinometry. Thorax. 2000. 55:921–924.46. Patriarca G, Bellioni P, Nucera E, Schiavino D, Papa G, Schinco G, Fais G, Pirotta LR. Intranasal treatment with lysine acetylsalicylate in patients with nasal polyposis. Ann Allergy. 1991. 67:588–592.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Update on the Management of Aspirin-Exacerbated Respiratory Disease
- Diagnostic Value of Clinical Parameters in the Prediction of Aspirin-Exacerbated Respiratory Disease in Asthma
- Update on Recent Advances in the Management of Aspirin Exacerbated Respiratory Disease
- Aspirin induces IL-4 production: augmented IL-4 production in aspirin-exacerbated respiratory disease
- WDR46 is a Genetic Risk Factor for Aspirin-Exacerbated Respiratory Disease in a Korean Population