Cancer Res Treat.
2009 Jun;41(2):93-99.
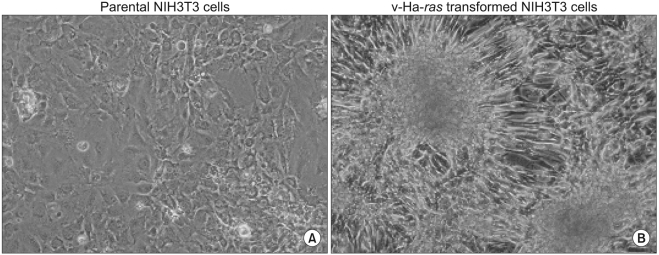
The Difference in Biological Properties between Parental and v-Ha-ras Transformed NIH3T3 Cells
- Affiliations
-
- 1Department of Biology, College of Natural Sciences, University of Incheon, Incheon, Korea. mikelee@incheon.ac.kr
Abstract
- PURPOSE
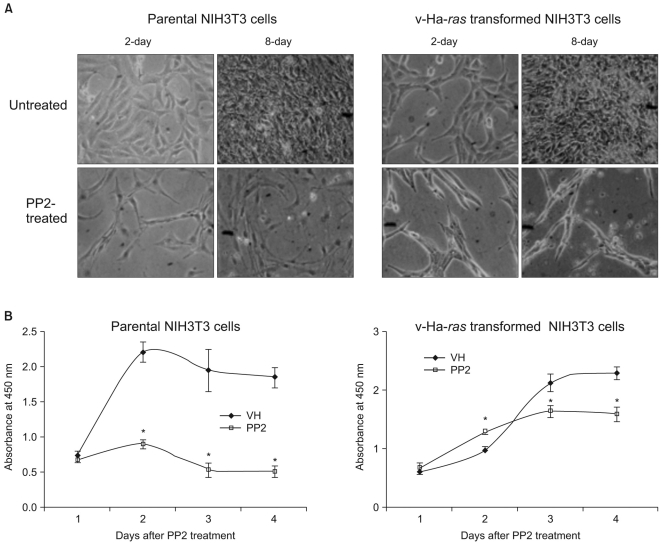
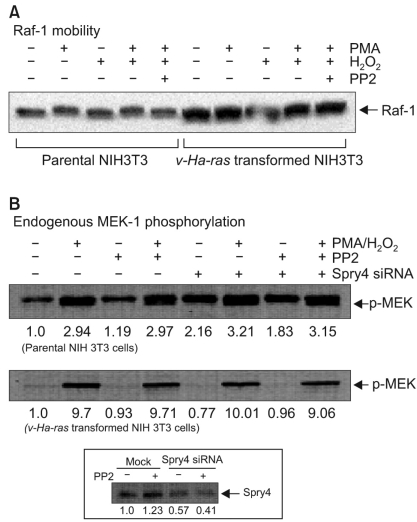
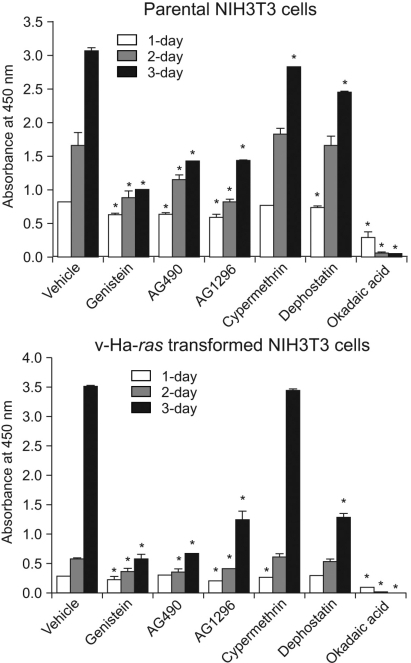
We performed experiments to investigate the change in cellular signaling that occurs during the transformation of a normal cell to a cell capable of cancerous growth, and we did so by using the NIH 3T3 cells that were transformed by transfection with the v-Ha-ras oncogene. MATERIALS AND METHODS: Parental and v-Ha-ras transfected NIH 3T3 cells were chosen as test systems. The siRNA transfections were performed using Lipofectamine 2000. The cell proliferation reagent WST-1 was used for the quantitative determination of cellular proliferation. Immunoblot analysis was performed using the ECL-Plus chemiluminescent system and a KODAK Image Station 4000R. RESULTS: The v-Ha-ras-transformed cells were found to be significantly more resistant to PP2 treatment, which is a potent inhibitor of the Src family tyrosine kinases, than were the parental cells at earlier times after treatment. However, PP2 induced growth arrest and the senescence-like phenotypes in both cell lines after longer treatment. Furthermore, the Raf-1 kinase of the v-Ha-ras-transformed cells was not affected by the expressed level of Sprouty proteins, which are negative regulators of the MAPK pathway, as evidenced by the failure of siRNA-mediated knockdown of Spry4 to activate Raf-1 kinase. Dephostatin (a tyrosine phosphatase inhibitor) effectively inhibited the proliferation of the v-Ha-ras transformed cells, whereas dephostatin had only a small effect on the parental cells' proliferation. This implied an inhibitory role for tyrosine phosphatase that is specific to the signaling pathway in the v-Ha-ras transformed cells. CONCLUSION: Taken together, our results show that the sustained activation of the oncogenic pathways through their resistance to negative feedback regulation might contribute to the transformation of NIH 3T3 cells.
Keyword
MeSH Terms
Figure
Reference
-
2. Spandidos DA, Glarakis IS, Kotsinas A, Ergazaki M, Kiaris H. Ras oncogene activation in benign and malignant colorectal tumours. Tumori. 1995; 81:7–11. PMID: 7571058.3. Kiaris H, Spandidos DA, Jones AS, Vaughan ED, Field JK. Mutations, expression and genomic instability of the H-ras proto-oncogene in squamous cell carcinomas of the head and neck. Br J Cancer. 1995; 72:123–128. PMID: 7599040.
Article4. Marshall MS. Ras target proteins in eucaryotic cells. FASEB J. 1995; 9:1311–1318. PMID: 7557021.5. Terai K, Matsuda M. Ras binding opens c-Raf to expose the docking site for mitogen-activated protein kinase kinase. EMBO Rep. 2005; 6:251–255. PMID: 15711535.
Article6. Sasaki A, Taketomi T, Kato R, Saeki K, Nonami A, Sasaki M, et al. Mammalian Sprouty4 suppresses Ras-independent ERK activation by binding to Raf1. Nat Cell Biol. 2003; 5:427–432. PMID: 12717443.
Article7. Yusoff P, Lao DH, Ong SH, Wong ES, Lim J, Lo TL, et al. Sprouty2 inhibits the Ras/MAP Kinase pathway by inhibiting the activation of Raf. J Biol Chem. 2002; 277:3195–3201. PMID: 11698404.
Article8. Kim HJ, Bar-Sagi D. Modulation of signalling by Sprouty: a developing story. Nat Rev Mol Cell Biol. 2004; 5:441–450. PMID: 15173823.
Article9. Lee M. Raf-1 kinase activation is uncoupled from downstream MEK/ERK pathway in cells treated with Src tyrosine kinase inhibitor PP2. Biochem Biophys Res Commun. 2006; 350:450–456. PMID: 17010316.
Article10. Marais R, Light Y, Paterson HF, Marshall CJ. Ras recruits Raf-1 to the plasma membrane for activation by tyrosine phosphorylation. EMBO J. 1995; 14:3136–3145. PMID: 7542586.
Article11. Hanke JH, Gardner JP, Dow RL, Changelian PS, Brissette WH, Weringer EJ, et al. Discovery of a novel, potent, and Src family-selcetive tyrosine kinase inhibitor: Study of lck and fynT-dependent T cell activation. J Biol Chem. 1996; 271:695–701. PMID: 8557675.12. Rapp UR, Goldsborough MD, Mark GE, Bonner TI, Groffen J, Reynolds FH Jr, et al. Structure and biological activity of v-raf, a unique oncogene transduced by a retrovirus. Proc Natl Acad Sci USA. 1983; 80:4218–4222. PMID: 6308607.
Article13. Heidecker G, Kolch W, Morrison DK, Rapp UR. The role of Raf-1 phosphorylation in signal transduction. Adv Cancer Res. 1992; 58:53–73. PMID: 1312290.
Article14. Lee M, Kim JY, Anderson WB. Src tyrosine kinase inhibitor PP2 markedly enhances Ras-independent activation of Raf-1 protein kinase by phorbol myristate acetate and H2O2. J Biol Chem. 2004; 279:48692–48701. PMID: 15356004.15. Williams NG, Paradis H, Agarwal S, Charest DL, Pelech SL, Roberts TM. Raf-1 and p21v-ras cooperate in the activation of mitogen-activated protein kinase. Proc Natl Acad Sci USA. 1993; 90:5772–5776. PMID: 8390681.
Article16. Williams NG, Roberts TM, Li P. Both p21ras and pp60v-src are required, but neither alone is sufficient, to activate the Raf-1 kinase. Proc Natl Acad Sci USA. 1992; 89:2922–2926. PMID: 1372995.
Article17. Giannoni E, Buricchi F, Raugei G, Ramponi G, Chiarugi P. Intracellular reactive oxygen species activate Src tyrosine kinase during cell adhesion and anchorage-dependent cell growth. Mol Cell Biol. 2005; 25:6391–6403. PMID: 16024778.
Article18. Lee AC, Fenster BE, Ito H, Takeda K, Bae NS, Hirai T, et al. Ras proteins induce senescence by altering the intracellular levels of reactive oxygen species. J Biol Chem. 1999; 274:7936–7940. PMID: 10075689.
Article19. Santillo M, Mondola P, Seru R, Annella T, Cassano S, Ciullo I, et al. Opposing functions of Ki- and Ha-Ras genes in the regulation of redox signals. Curr Biol. 2001; 11:614–619. PMID: 11369207.
Article20. Salmeen A, Andersen JN, Myers MP, Meng TC, Hinks JA, Tonks NK, et al. Redox regulation of protein tyrosine phosphatase 1B involves a sulphenyl-amide intermediate. Nature. 2003; 423:769–773. PMID: 12802338.
Article21. Lee M, Petrovics G, Anderson WB. The synergistic activation of Raf-1 kinase by phorbol myristate acetate and hydrogen peroxide in NIH 3T3 cells. Biochem Biophys Res Commun. 2003; 311:1026–1033. PMID: 14623285.22. Lou YW, Chen YY, Hsu SF, Chen RK, Lee CL, Khoo KH, et al. Redox regulation of the protein tyrosine phosphatase PTP1B in cancer cells. FEBS J. 2008; 275:69–88. PMID: 18067579.
Article23. Ravi RK, Weber E, McMahon M, Williams JR, Baylin S, Mal A, et al. Activated Raf-1 causes growth arrest in human small cell lung cancer cells. J Clin Invest. 1998; 101:153–159. PMID: 9421477.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Molecular assembly of mitogen-activated protein kinase module in ras-transformed NIH3T3 cell line
- Antisense GLUT1 RNA suppresses the transforming phenotypes of NIH 3T3 cells transformed by N-Ras
- Differential Physiological Effects of Raf-1 Kinase Pathways Linked to Protein Kinase C Activation Depending on the Stimulus in v-H-ras-transformed Cells
- Oncogenic Ras downregulates mdr1b expression through generation of reactive oxygen species
- ATG5 knockout promotes paclitaxel sensitivity in drug-resistant cells via induction of necrotic cell death