Korean J Gastroenterol.
2016 May;67(5):272-276. 10.4166/kjg.2016.67.5.272.
Repeated Panniculitis Induced by Pegylated Interferon Alpha 2a in a Patient with Chronic Hepatitis C
- Affiliations
-
- 1Department of Internal Medicine, Hanyang University Guri Hospital, Hanyang University College of Medicine, Guri, Korea. sonjh@hanyang.ac.kr
- 2Department of Pathology, Hanyang University Guri Hospital, Hanyang University College of Medicine, Guri, Korea.
- KMID: 2164336
- DOI: http://doi.org/10.4166/kjg.2016.67.5.272
Abstract
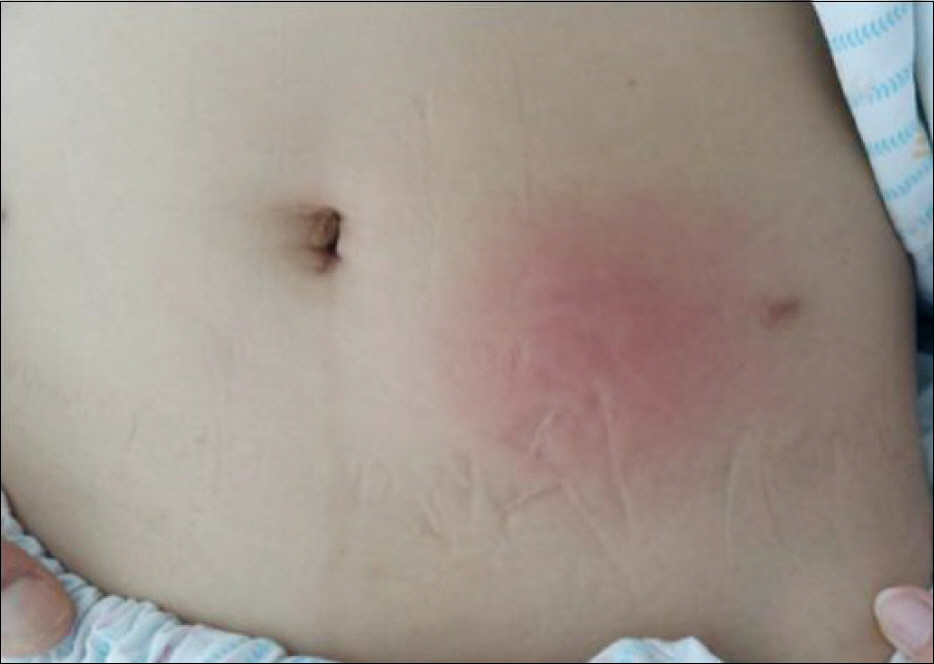
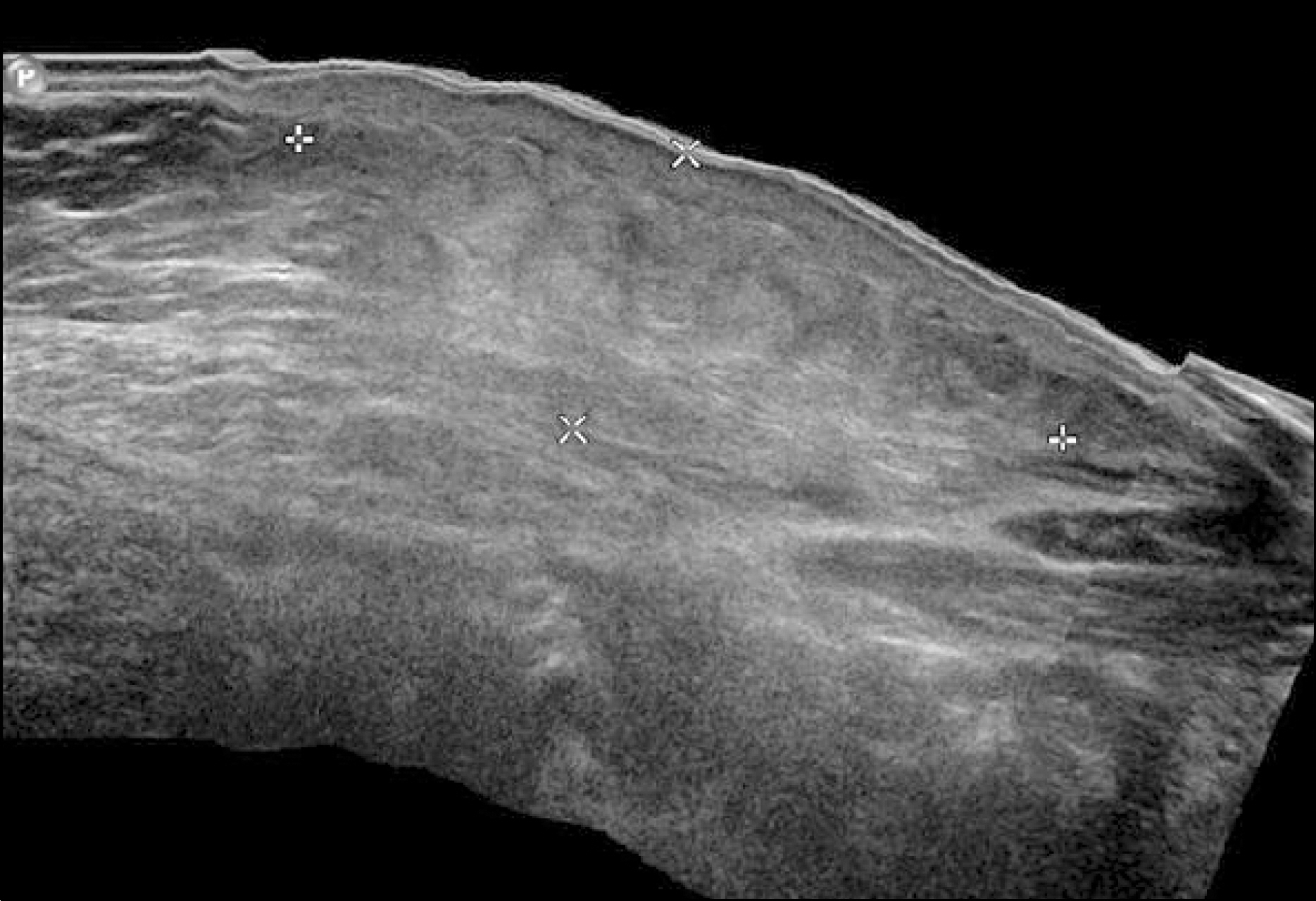
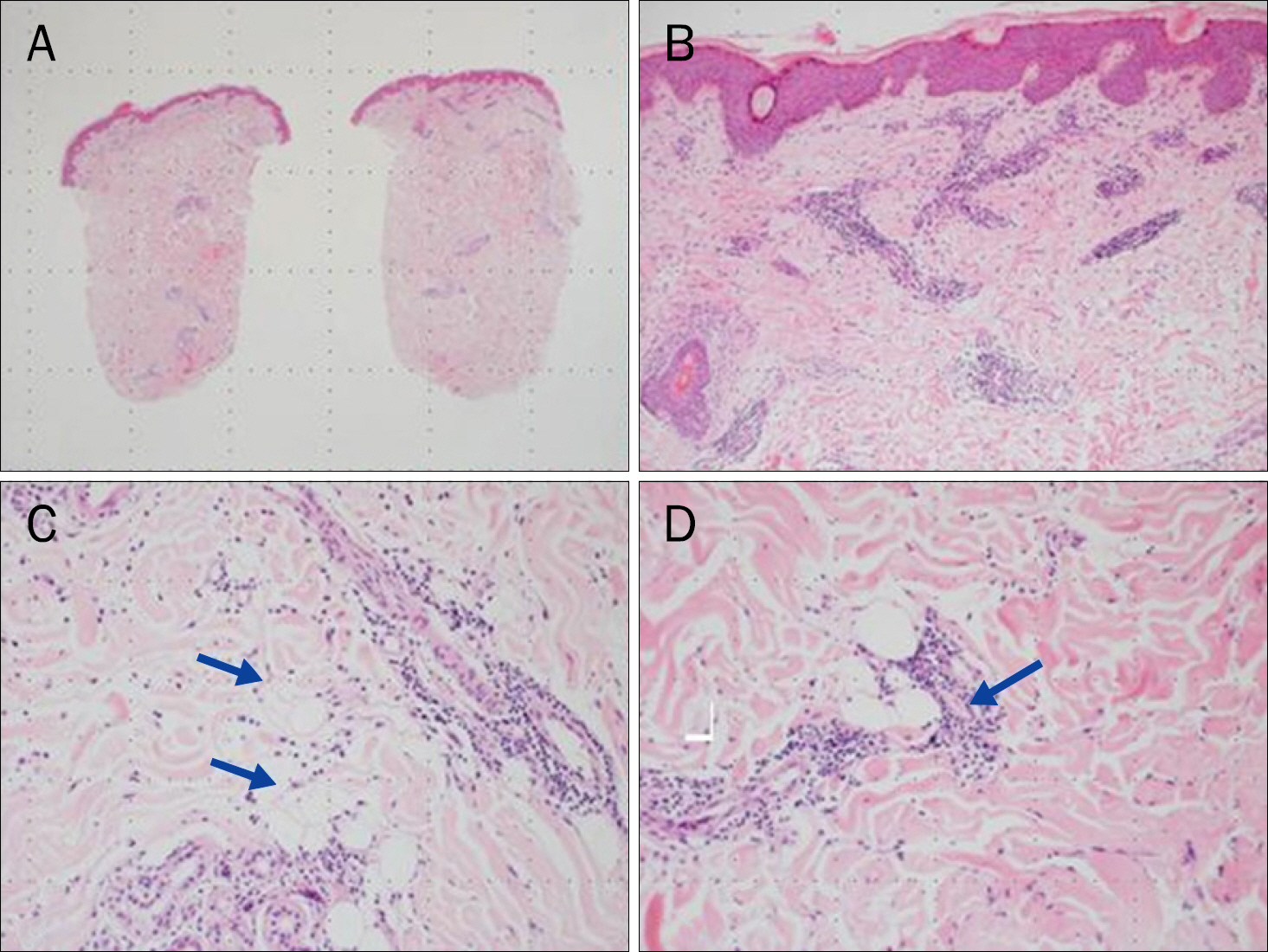
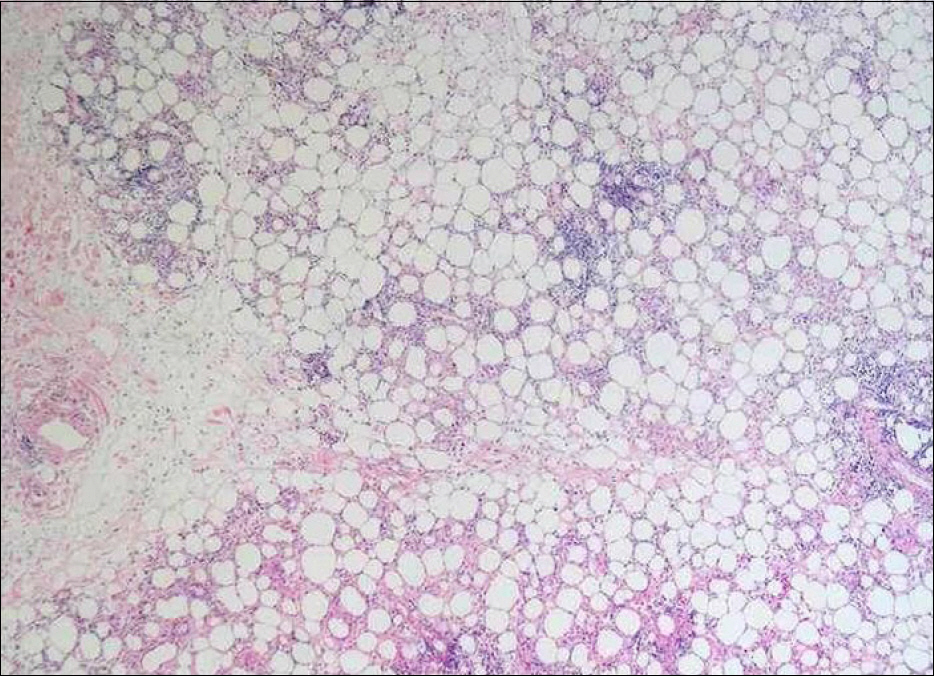
- Pegylated interferon alpha (PEG-IFN-α) is widely used to treat chronic hepatitis C in combination with ribavirin. Many adverse effects of PEG-IFN-α, such as hematologic, psychologic, dermatologic, immunologic, and other abnormalities, have been reported, and some serious adverse events lead to PEG-IFN-α treatment discontinuation. For very rare adverse events such as panniculitis, there are no established guidelines on whether to continue PEG-IFN-α treatment. Published reports on panniculitis induced by PEG-IFN-α 2a are sparse. Herein we report a case of repeated occurrences of panniculitis in a patient with chronic hepatitis C, leading to treatment cessation.
MeSH Terms
Figure
Reference
-
References
1. Hoofnagle JH, Mullen KD, Jones DB, et al. Treatment of chronic non-A, non-B hepatitis with recombinant human alpha interferon. A preliminary report. N Engl J Med. 1986; 315:1575–1578.2. Sulkowski MS, Cooper C, Hunyady B, et al. Management of adverse effects of Peg-IFN and ribavirin therapy for hepatitis C. Nat Rev Gastroenterol Hepatol. 2011; 8:212–223.
Article3. O'Sullivan SS, Cronin EM, Sweeney BJ, Bourke JF, Fitzgibbon J. Panniculitis and lipoatrophy after subcutaneous injection of interferon beta-1b in a patient with multiple sclerosis. J Neurol Neurosurg Psychiatry. 2006; 77:1382–1383.4. Heinzerling L, Dummer R, Burg G, Schmid-Grendelmeier P. Panniculitis after subcutaneous injection of interferon beta in a multiple sclerosis patient. Eur J Dermatol. 2002; 12:194–197.5. Poulin F, Rico P, Côté J, Bégin LR. Interferon beta-induced panniculitis mimicking acute appendicitis. Arch Dermatol. 2009; 145:916–917.
Article6. Baek YS, Kim JY, Choi JE, Ahn HH, Kye YC, Seo SH. Pegylated interferon Alpha2a induced panniculitis in patient with chronic hepatitis C. Korean J Dermatol. 2012; 50:640–643.7. Poynard T, Marcellin P, Lee SS, et al. Randomised trial of interferon alpha2b plus ribavirin for 48 weeks or for 24 weeks versus interferon alpha2b plus placebo for 48 weeks for treatment of chronic infection with hepatitis C virus. International Hepatitis Interventional Therapy Group (IHIT). Lancet. 1998; 352:1426–1432.8. McHutchison JG, Gordon SC, Schiff ER, et al. Interferon alfa-2b alone or in combination with ribavirin as initial treatment for chronic hepatitis C. Hepatitis Interventional Therapy Group. N Engl J Med. 1998; 339:1485–1492.9. Glue P, Fang JW, Rouzier-Panis R, et al. Pegylated interferon-alpha2b: pharmacokinetics, pharmacodynamics, safety, and preliminary efficacy data. Hepatitis C Intervention Therapy Group. Clin Pharmacol Ther. 2000; 68:556–567.10. Bailon P, Palleroni A, Schaffer CA, et al. Rational design of a potent, long-lasting form of interferon: a 40 kDa branched polyethylene glycol-conjugated interferon alpha-2a for the treatment of hepatitis C. Bioconjug Chem. 2001; 12:195–202.11. Dusheiko G. Side effects of alpha interferon in chronic hepatitis C. Hepatology. 1997; 26(3 Suppl 1):112S–121S.12. Eberhard BA, Ilowite NT. Panniculitis and lipodystrophy. Curr Opin Rheumatol. 2002; 14:566–570.
Article13. Ter Poorten MC, Thiers BH. Panniculitis. Dermatol Clin. 2002; 20:421–433.
Article14. Buttmann M, Goebeler M, Toksoy A, et al. Subcutaneous interferon-beta injections in patients with multiple sclerosis initiate inflammatory skin reactions by local chemokine induction. J Neuroimmunol. 2005; 168:175–182.15. Inafuku H, Kasem Khan MA, Nagata T, Nonaka S. Cutaneous ulcerations following subcutaneous interferon beta injection to a patient with multiple sclerosis. J Dermatol. 2004; 31:671–677.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Pegylated Interferon Alpha2a Induced Panniculitis in Patient with Chronic Hepatitis C
- A Case of Cutaneous Sarcoidosis Associated with Pegylated Interferon Alpha-2a and Ribavirin Therapy
- Alopecia Areata in a Patient Treated with Interferon Alpha-2a for Chronic Hepatitis B
- Pulmonary Toxicity by Pegylated Interferon alpha-2a in a Patient with Chronic Hepatitis C
- Thymosin Alpha-1 in Combination with Pegylated Interferon and Ribavirin in Chronic Hepatitis C Patients Who have Failed to Prior Pegylated Interferon and Ribavirin Treatment