Chonnam Med J.
2016 May;52(2):91-100. 10.4068/cmj.2016.52.2.91.
Vilazodone for the Treatment of Depression: An Update
- Affiliations
-
- 1Department of Psychiatry, The Catholic University of Korea College of Medicine, Seoul, Korea. pae@catholic.ac.kr
- 2International Health Care Center, Seoul St. Mary's Hospital, The Catholic University of Korea College of Medicine, Seoul, Korea.
- 3Department of Psychiatry, College of Medicine, Korea University, Seoul, Korea.
- 4Department of Psychiatry and Behavioural Sciences, Duke University Medical Center, Durham, NC, USA.
- 5Global Medical Education, New York, NY, USA.
- KMID: 2164318
- DOI: http://doi.org/10.4068/cmj.2016.52.2.91
Abstract
- Vilazodone is a novel antidepressant having a selective serotonin (5-HT) reuptake inhibitor and 5-HT(1A) receptor partial agonist profile, so it has been regarded as a serotonin partial agonist-reuptake inhibitor (SPARI). We aimed to provide Vilazodone's clinical implications mainly by reviewing published clinical trials. Vilazodone has been speculated to have three potential benefits including faster onset of action, greater efficacy, and better tolerability owning to its SPARI properties. However, no studies conducted so far have directly proven the above speculations. Five initial phase II trials failed to distinguish vilazodone from placebo in the treatment of MDD, but 4 randomized clinical trials (RCT), 3 post-hoc or pooled analysis, 1 long-term open label study, and a meta-analysis showed vilazodone's superior efficacy over placebo. The studies also showed vilazodone is generally safe and tolerable. However, diarrhea, nausea, headache, dizziness, dry mouth, and insomnia warrant close attention in clinical practice because they have been constantly noted throughout the clinical studies. 2 RCTs recently documented the efficacy and safety of vilazodone in patients with generalized anxiety disorder, which could be a start of broadening vilazodone's usage or FDA approval in diverse anxiety disorders.
Keyword
MeSH Terms
-
Antidepressive Agents
Anxiety Disorders
Depression*
Diarrhea
Dizziness
Headache
Humans
Mouth
Nausea
Receptor, Serotonin, 5-HT1A
Serotonin
Serotonin Uptake Inhibitors
Sleep Initiation and Maintenance Disorders
Vilazodone Hydrochloride*
Antidepressive Agents
Receptor, Serotonin, 5-HT1A
Serotonin
Serotonin Uptake Inhibitors
Vilazodone Hydrochloride
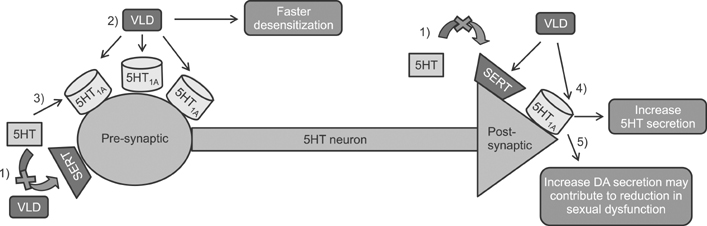
Figure
Reference
-
1. Kessler RC, Petukhova M, Sampson NA, Zaslavsky AM, Wittchen HU. Twelve-month and lifetime prevalence and lifetime morbid risk of anxiety and mood disorders in the United States. Int J Methods Psychiatr Res. 2012; 21:169–184.
Article2. Kulkarni SK, Dhir A. Current investigational drugs for major depression. Expert Opin Investig Drugs. 2009; 18:767–788.
Article3. Rush AJ, Trivedi MH, Wisniewski SR, Nierenberg AA, Stewart JW, Warden D, et al. Acute and longer-term outcomes in depressed outpatients requiring one or several treatment steps: a STAR*D report. Am J Psychiatry. 2006; 163:1905–1917.
Article4. Serretti A, Chiesa A. Treatment-emergent sexual dysfunction related to antidepressants: a meta-analysis. J Clin Psychopharmacol. 2009; 29:259–266.5. Kim JM, Hong JP, Kim SD, Kang HJ, Lee YS. Development of a Korean Version of the Perceived Deficits Questionnaire-Depression for Patients with Major Depressive Disorder. Clin Psychopharmacol Neurosci. 2016; 14:26–32.
Article6. Massart R, Mongeau R, Lanfumey L. Beyond the monoaminergic hypothesis: neuroplasticity and epigenetic changes in a transgenic mouse model of depression. Philos Trans R Soc Lond B Biol Sci. 2012; 367:2485–2494.
Article7. Artigas F. Serotonin receptors involved in antidepressant effects. Pharmacol Ther. 2013; 137:119–131.
Article8. Wang SM, Han C, Lee SJ, Patkar AA, Masand PS, Pae CU. Five potential therapeutic agents as antidepressants: a brief review and future directions. Expert Rev Neurother. 2015; 15:1015–1029.
Article9. Forest Pharmaceuticals I. Viibryd (Vilazodone) Prescribing Information. updated April 201120 July 2012. Available from: http://www.frx.com/pi/viibryd_pi.pdf.10. Hughes ZA, Starr KR, Langmead CJ, Hill M, Bartoszyk GD, Hagan JJ, et al. Neurochemical evaluation of the novel 5-HT1A receptor partial agonist/serotonin reuptake inhibitor, vilazodone. Eur J Pharmacol. 2005; 510:49–57.
Article11. Boinpally R, Henry D, Gupta S, Edwards J, Longstreth J, Periclou A. Pharmacokinetics and Safety of Vilazodone in Hepatic Impairment. Am J Ther. 2015; 22:269–277.
Article12. Boinpally R, Alcorn H, Adams MH, Longstreth J, Edwards J. Pharmacokinetics of vilazodone in patients with mild or moderate renal impairment. Clin Drug Investig. 2013; 33:199–206.
Article13. Administration. UFaD. Vilazodone Drug Approval Package. updated 1 March 2011; cited 2012 20 July. Available from: http://www.accessdata.fda.gov/drugsatfda_docs/nda/2011/022567Orig1s000TOC.cfm.14. Stahl SM. Mechanism of action of the SPARI vilazodone: serotonin 1A partial agonist and reuptake inhibitor. CNS Spectr. 2014; 19:105–109.
Article15. Sprouse JS, Aghajanian GK. Electrophysiological responses of serotoninergic dorsal raphe neurons to 5-HT1A and 5-HT1B agonists. Synapse. 1987; 1:3–9.
Article16. Casanovas JM, Lésourd M, Artigas F. The effect of the selective 5-HT1A agonists alnespirone (S-20499) and 8-OH-DPAT on extracellular 5-hydroxytryptamine in different regions of rat brain. Br J Pharmacol. 1997; 122:733–741.
Article17. Lanfumey L, Hamon M. Central 5-HT(1A) receptors: regional distribution and functional characteristics. Nucl Med Biol. 2000; 27:429–435.
Article18. Blier P, Ward NM. Is there a role for 5-HT1A agonists in the treatment of depression? Biol Psychiatry. 2003; 53:193–203.
Article19. Celada P, Puig M, Amargós-Bosch M, Adell A, Artigas F. The therapeutic role of 5-HT1A and 5-HT2A receptors in depression. J Psychiatry Neurosci. 2004; 29:252–265.20. Starr KR, Price GW, Watson JM, Atkinson PJ, Arban R, Melotto S, et al. SB-649915-B, a novel 5-HT1A/B autoreceptor antagonist and serotonin reuptake inhibitor, is anxiolytic and displays fast onset activity in the rat high light social interaction test. Neuropsychopharmacology. 2007; 32:2163–2172.
Article21. Hughes ZA, Starr KR, Scott CM, Newson MJ, Sharp T, Watson JM, et al. Simultaneous blockade of 5-HT1A/B receptors and 5-HT transporters results in acute increases in extracellular 5-HT in both rats and guinea pigs: in vivo characterization of the novel 5-HT1A/B receptor antagonist/5-HT transport inhibitor SB-649-915-B. Psychopharmacology (Berl). 2007; 192:121–133.
Article22. Duxon MS, Starr KR, Upton N. Latency to paroxetine-induced anxiolysis in the rat is reduced by co-administration of the 5-HT(1A) receptor antagonist WAY100635. Br J Pharmacol. 2000; 130:1713–1719.
Article23. Hogg S, Dalvi A. Acceleration of onset of action in schedule-induced polydipsia: combinations of SSRI and 5-HT1A and 5-HT1B receptor antagonists. Pharmacol Biochem Behav. 2004; 77:69–75.
Article24. Page ME, Cryan JF, Sullivan A, Dalvi A, Saucy B, Manning DR, et al. Behavioral and neurochemical effects of 5-(4-[4-(5-Cyano-3-indolyl)-butyl)-butyl]-1-piperazinyl)-benzofuran-2-carboxamide (EMD 68843): a combined selective inhibitor of serotonin reuptake and 5-hydroxytryptamine(1A) receptor partial agonist. J Pharmacol Exp Ther. 2002; 302:1220–1227.
Article25. Wang SM, Han C, Lee SJ, Patkar AA, Masand PS, Pae CU. Vilazodone for the treatment of major depressive disorder: focusing on its clinical studies and mechanism of action. Psychiatry Investig. 2015; 12:155–163.
Article26. Hellerstein DJ, Flaxer J. Vilazodone for the treatment of major depressive disorder: an evidence-based review of its place in therapy. Core Evid. 2015; 10:49–62.
Article27. Laughren TP, Gobburu J, Temple RJ, Unger EF, Bhattaram A, Dinh PV, et al. Vilazodone: clinical basis for the US Food and Drug Administration's approval of a new antidepressant. J Clin Psychiatry. 2011; 729:1166–1173.28. Rickels K, Athanasiou M, Robinson DS, Gibertini M, Whalen H, Reed CR. Evidence for efficacy and tolerability of vilazodone in the treatment of major depressive disorder: a randomized, double-blind, placebo-controlled trial. J Clin Psychiatry. 2009; 70:326–333.
Article29. Khan A, Cutler AJ, Kajdasz DK, Gallipoli S, Athanasiou M, Robinson DS, et al. A randomized, double-blind, placebo-controlled, 8-week study of vilazodone, a serotonergic agent for the treatment of major depressive disorder. J Clin Psychiatry. 2011; 72:441–447.
Article30. Khan A, Sambunaris A, Edwards J, Ruth A, Robinson DS. Vilazodone in the treatment of major depressive disorder: efficacy across symptoms and severity of depression. Int Clin Psychopharmacol. 2014; 29:86–92.31. Jain R, Chen D, Edwards J, Mathews M. Early and sustained improvement with vilazodone in adult patients with major depressive disorder: post hoc analyses of two phase III trials. Curr Med Res Opin. 2014; 30:263–270.
Article32. Mathews M, Gommoll C, Chen D, Nunez R, Khan A. Efficacy and safety of vilazodone 20 and 40 mg in major depressive disorder: a randomized, double-blind, placebo-controlled trial. Int Clin Psychopharmacol. 2015; 30:67–74.
Article33. Croft HA, Pomara N, Gommoll C, Chen D, Nunez R, Mathews M. Efficacy and safety of vilazodone in major depressive disorder: a randomized, double-blind, placebo-controlled trial. J Clin Psychiatry. 2014; 75:e1291–e1298.34. Citrome L, Gommoll CP, Tang X, Nunez R, Mathews M. Evaluating the efficacy of vilazodone in achieving remission in patients with major depressive disorder: post-hoc analyses of a phase IV trial. Int Clin Psychopharmacol. 2015; 30:75–81.
Article35. Robinson DS, Kajdasz DK, Gallipoli S, Whalen H, Wamil A, Reed CR. A 1-year, open-label study assessing the safety and tolerability of vilazodone in patients with major depressive disorder. J Clin Psychopharmacol. 2011; 31:643–646.
Article36. Zhang XF, Wu L, Wan DJ, Liu RZ, Dong Z, Chen M, et al. Evaluation of the efficacy and safety of vilazodone for treating major depressive disorder. Neuropsychiatr Dis Treat. 2015; 11:1957–1965.37. Gommoll C, Durgam S, Mathews M, Forero G, Nunez R, Tang X, et al. A double-blind, randomized, placebo-controlled, fixed-dose phase III study of vilazodone in patients with generalized anxiety disorder. Depress Anxiety. 2015; 32:451–459.
Article38. Citrome L. Vilazodone for major depressive disorder: a systematic review of the efficacy and safety profile for this newly approved antidepressant - what is the number needed to treat, number needed to harm and likelihood to be helped or harmed? Int J Clin Pract. 2012; 66:356–368.
Article39. Le Poul E, Laaris N, Doucet E, Laporte AM, Hamon M, Lanfumey L. Early desensitization of somato-dendritic 5-HT1A autoreceptors in rats treated with fluoxetine or paroxetine. Naunyn Schmiedebergs Arch Pharmacol. 1995; 352:141–148.
Article40. Li Q, Muma NA, Battaglia G, Van de Kar LD. A desensitization of hypothalamic 5-HT1A receptors by repeated injections of paroxetine: reduction in the levels of G(i) and G(o) proteins and neuroendocrine responses, but not in the density of 5-HT1A receptors. J Pharmacol Exp Ther. 1997; 282:1581–1590.41. Wang SM, Han C, Lee SJ, Patkar AA, Masand PS, Pae CU. A review of current evidence for vilazodone in major depressive disorder. Int J Psychiatry Clin Pract. 2013; 17:160–169.
Article42. Clayton AH, Kennedy SH, Edwards JB, Gallipoli S, Reed CR. The effect of vilazodone on sexual function during the treatment of major depressive disorder. J Sex Med. 2013; 10:2465–2476.
Article43. Murck H, Frieboes RM, Antonijevic IA, Steiger A. Distinct temporal pattern of the effects of the combined serotonin-reuptake inhibitor and 5-HT1A agonist EMD 68843 on the sleep EEG in healthy men. Psychopharmacology (Berl). 2001; 155:187–192.
Article44. Loane C, Politis M. Buspirone: what is it all about? Brain Res. 2012; 1461:111–118.
Article45. Hoge EA, Ivkovic A, Fricchione GL. Generalized anxiety disorder: diagnosis and treatment. BMJ. 2012; 345:e7500.
Article46. Gommoll C, Forero G, Mathews M, Nunez R, Tang X, Durgam S, et al. Vilazodone in patients with generalized anxiety disorder: a double-blind, randomized, placebo-controlled, flexible-dose study. Int Clin Psychopharmacol. 2015; 30:297–306.47. Rush AJ, Warden D, Wisniewski SR, Fava M, Trivedi MH, Gaynes BN, et al. STAR*D: revising conventional wisdom. CNS Drugs. 2009; 23:627–647.48. Bouwer C, Stein DJ. Buspirone is an effective augmenting agent of serotonin selective re-uptake inhibitors in severe treatment-refractory depression. S Afr Med J. 1997; 87:4 Suppl. 534–537. 54049. Segraves RT. Sexual dysfunction associated with antidepressant therapy. Urol Clin North Am. 2007; 34:575–579.
Article50. Segraves RT, Balon R. Antidepressant-induced sexual dysfunction in men. Pharmacol Biochem Behav. 2014; 121:132–137.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Vilazodone for the Treatment of Major Depressive Disorder: Focusing on Its Clinical Studies and Mechanism of Action
- The Association between Dizziness and Anxiety: Update to the Treatment
- Interaction between Personality and Depression Outcome
- Bipolar Diathesis in Patients with Treatment-Resistant Depression
- Diagnosis and Treatment of Postpartum Depression