J Korean Med Sci.
2015 Jun;30(6):816-822. 10.3346/jkms.2015.30.6.816.
Size Control of 99mTc-tin Colloid Using PVP and Buffer Solution for Sentinel Lymph Node Detection
- Affiliations
-
- 1Department of Nuclear Medicine, Molecular Imaging & Therapeutic Medicine Research Center, Cyclotron Research Center, Institute for Medical Science, Biomedical Research Institute, Chonbuk National University Medical School and Hospital, Jeonju, Korea. jayj
- KMID: 2160613
- DOI: http://doi.org/10.3346/jkms.2015.30.6.816
Abstract
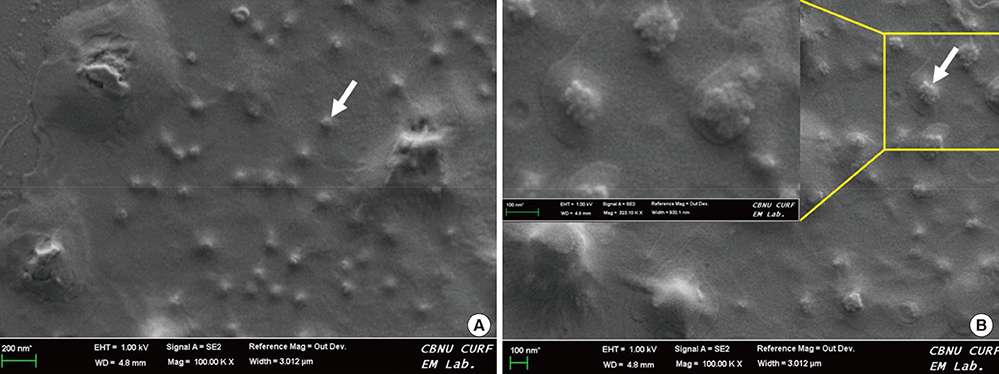
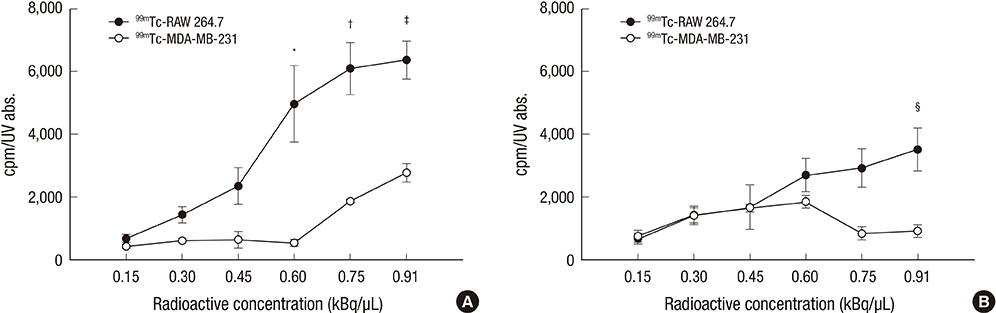
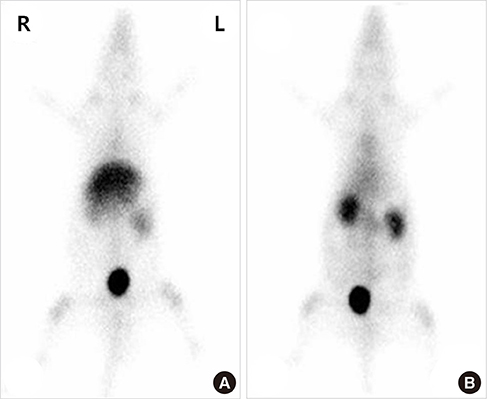
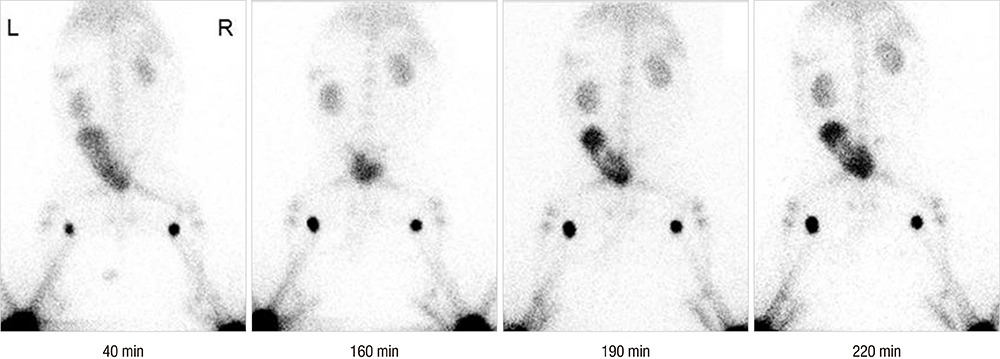
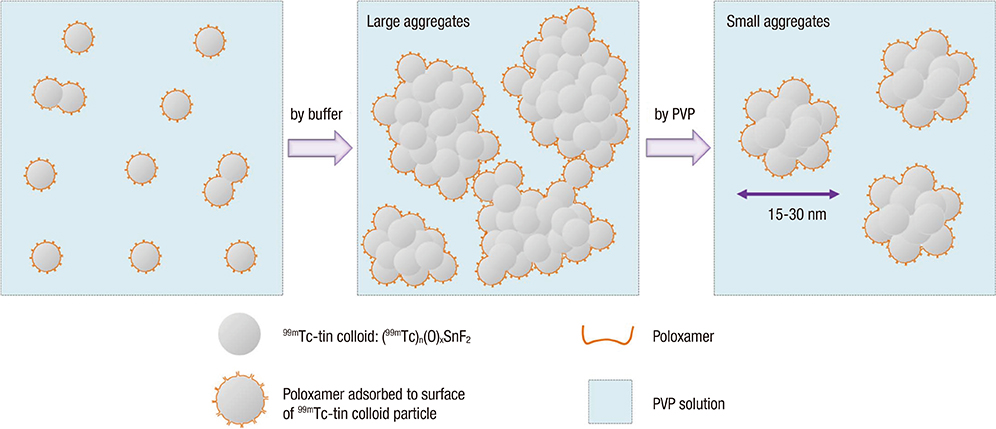
- Colloidal particle size is an important characteristic that allows mapping sentinel nodes in lymphoscintigraphy. This investigation aimed to introduce different ways of making a 99mTc-tin colloid with a size of tens of nanometers. All agents, tin fluoride, sodium fluoride, poloxamer-188, and polyvinylpyrrolidone (PVP), were mixed and labeled with 99mTc. Either phosphate or sodium bicarbonate buffers were used to adjust the pH levels. When the buffers were added, the size of the colloids increased. However, as the PVP continued to increase, the size of the colloids was controlled to within tens of nanometers. In all samples, phosphate buffer added PVP (30 mg) stabilized tin colloid (99mTc-PPTC-30) and sodium bicarbonate solution added PVP (50 mg) stabilized tin colloid (99mTc-BPTC-50) were chosen for in vitro and in vivo studies. 99mTc-BPTC-50 (<20 nm) was primarily located in bone marrow and was then secreted through the kidneys, and 99mTc-PPTC-30 (>100 nm) mainly accumulated in the liver. When a rabbit was given a toe injection, the node uptake of 99mTc-PPTC-30 decreased over time, while 99mTc-BPTC-50 increased. Therefore, 99mTc-BPTC-50 could be a good candidate radiopharmaceutical for sentinel node detection. The significance of this study is that nano-sized tin colloid can be made very easily and quickly by PVP.
MeSH Terms
-
Animals
Buffers
Cell Line, Tumor
Humans
Lymph Nodes/*radionuclide imaging
Lymphatic Metastasis
Metal Nanoparticles/chemistry/ultrastructure
Mice
Neoplasms, Experimental/*radionuclide imaging
Particle Size
Povidone/*chemistry
Rabbits
Radiopharmaceuticals/*chemical synthesis
Reproducibility of Results
Sensitivity and Specificity
Technetium Compounds/*chemistry
Tin/*chemistry
Tin Compounds/*chemistry
Buffers
Radiopharmaceuticals
Povidone
Technetium Compounds
Tin
Tin Compounds
Figure
Reference
-
1. Giuliano AE, Dale PS, Turner RR, Morton DL, Evans SW, Krasne DL. Improved axillary staging of breast cancer with sentinel lymphadenectomy. Ann Surg. 1995; 222:394–399. discussion 9-401.2. Na CJ, Kim J, Choi S, Han YH, Jeong HJ, Sohn MH, Youn HJ, Lim ST. The clinical value of hybrid sentinel lymphoscintigraphy to predict metastatic sentinel lymph nodes in breast cancer. Nucl Med Mol Imaging. 2015; 49:26–32.3. Morton DL, Thompson JF, Essner R, Elashoff R, Stern SL, Nieweg OE, Roses DF, Karakousis CP, Mozzillo N, Reintgen D, et al. Validation of the accuracy of intraoperative lymphatic mapping and sentinel lymphadenectomy for early-stage melanoma: a multicenter trial. Multicenter Selective Lymphadenectomy Trial Group. Ann Surg. 1999; 230:453–463. discussion 63-5.4. Nune SK, Gunda P, Majeti BK, Thallapally PK, Forrest ML. Advances in lymphatic imaging and drug delivery. Adv Drug Deliv Rev. 2011; 63:876–885.5. Boolbol SK, Fey JV, Borgen PI, Heerdt AS, Montgomery LL, Paglia M, Petrek JA, Cody HS 3rd, Van Zee KJ. Intradermal isotope injection: a highly accurate method of lymphatic mapping in breast carcinoma. Ann Surg Oncol. 2001; 8:20–24.6. Kim BT. Sentinel lymph node imaging in breast vancer. Korean J Nucl Med. 1999; 33:243–246.7. Plut EM, Hinkle GH, Guo W, Lee RJ. Kit formulation for the preparation of radioactive blue liposomes for sentinel node lymphoscintigraphy. J Pharm Sci. 2002; 91:1717–1732.8. Eshima D, Eshima LA, Gotti NM, Herda SC, Algozine CA, Burris TG, Vansant JP, Alazraki NP, Taylor AT. Technetium-99m-sulfur colloid for lymphoscintigraphy: effects of preparation parameters. J Nucl Med. 1996; 37:1575–1578.9. Hung JC, Wiseman GA, Wahner HW, Mullan BP, Taggart TR, Dunn WL. Filtered technetium-99m-sulfur colloid evaluated for lymphoscintigraphy. J Nucl Med. 1995; 36:1895–1901.10. Mariani G, Moresco L, Viale G, Villa G, Bagnasco M, Canavese G, Buscombe J, Strauss HW, Paganelli G. Radioguided sentinel lymph node biopsy in breast cancer surgery. J Nucl Med. 2001; 42:1198–1215.11. Hauser W, Atkins HL, Richards P. Lymph node scanning with 99mTc-sulfur colloid. Radiology. 1969; 92:1369–1371.12. Pedersen B, Kristensen K. Evaluation of methods for sizing of colloidal radiopharmaceuticals. Eur J Nucl Med. 1981; 6:521–526.13. Tsopelas C. Particle size analysis of (99m)Tc-labeled and unlabeled antimony trisulfide and rhenium sulfide colloids intended for lymphoscintigraphic application. J Nucl Med. 2001; 42:460–466.14. Tsopelas C. Lymphoscintigraphy is more effective using higher specific activity 99mTc -antimony trisulfide colloid in the rat. Hell J Nucl Med. 2014; 17:19–26.15. Bombardieri E, Bonadonna G, Gianni L. Breast cancer: nuclear medicine in diagnosis and therapeutic options. Berlin: Springer;2007. p. 84.16. Trifirò G, Viale G, Gentilini O, Travaini LL, Paganelli G. Sentinel node detection in pre-operative axillary staging. Eur J Nucl Med Mol Imaging. 2004; 31:S46–S55.17. Cook SE, Park IK, Kim EM, Jeong HJ, Park TG, Choi YJ, Akaike T, Cho CS. Galactosylated polyethylenimine-graft-poly(vinyl pyrrolidone) as a hepatocyte-targeting gene carrier. J Control Release. 2005; 105:151–163.18. Liu X, Xu Y, Wu Z, Chen H. Poly(N-vinylpyrrolidone)-modified surfaces for biomedical applications. Macromol Biosci. 2013; 13:147–154.19. Wang L, Zeng R, Li C, Qiao R. Self-assembled polypeptide-block-poly (vinylpyrrolidone) as prospective drug-delivery systems. Colloids Surf B Biointerfaces. 2009; 74:284–292.20. Saha GB. Fundamentals of nuclear pharmacy. New York: Springer;1997. p. 101.21. Watanabe A. Cancer metastases research. 4th ed. New York: Nova Science Publishers;2008. p. 54–55.22. Illum L, Davis SS. The organ uptake of intravenously administered colloidal particles can be altered using a non-ionic surfactant (poloxamer 338). FEBS Lett. 1984; 167:79–82.23. Higashi H, Natsugoe S, Uenosono Y, Ehi K, Arigami T, Nakabeppu Y, Nakajo M, Aikou T. Particle size of tin and phytate colloid in sentinel node identification. J Surg Res. 2004; 121:1–4.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Comparison of the Results for Sentinel Lymph Node Mapping in the Breast Cancer Patients using 99mTc-Antimony Trisulfide Colloid, 99mTc-Tin Colloid, and 99mTc-Human Serum Albumin
- Camparison of the Efficiency for Tc-99m Tin-colloid and Tc-99m Phytate in Sentinel Node Detection in Breast Cancer Patients
- Sentinel Lymph Node Radiolocalization with 99mTc Filtered Tin Colloid in Clinically Node-Negative Squamous Cell Carcinomas of the Oral Cavity
- Clinical Significance of Sentinel Lymph Node Detection in Vulvar Cancer
- A Case of Anaphylactic Reactions to Isosulfan Blue Dye in A Cervical Cancer Patient