J Gynecol Oncol.
2015 Jan;26(1):46-53. 10.3802/jgo.2015.26.1.46.
Distinguishing benign from malignant pelvic mass utilizing an algorithm with HE4, menopausal status, and ultrasound findings
- Affiliations
-
- 1Department of Obstetrics and Gynecology, Faculty of Medicine, Ramathibodi Hospital, Mahidol University, Bangkok, Thailand. sarikapan.wil@mahidol.ac.th
- 2Department of Obstetrics and Gynecology, University of Hong Kong, Hong Kong.
- 3Department of Obstetrics and Gynecology, National Taiwan University Hospital, Taipei, Taiwan.
- 4Department of Obstetrics and Gynecology, Asan Medical Center, University of Ulsan College of Medicine, Seoul, Korea.
- 5Department of Obstetrics and Gynecology, The Jikei University, Tokyo, Japan.
- 6Department of Laboratory Medicine, Changi General Hospital, Singapore.
- 7Department of Pathology, Hospital Sultanah Aminah, Johor Bahru, Johor, Malaysia.
- 8Abbott Diagnostics, Abbott Park, IL, USA.
- 9Department of Obstetrics and Gynecology, MCU-FDT Medical Foundation, Caloocan, Philippines.
- KMID: 2158802
- DOI: http://doi.org/10.3802/jgo.2015.26.1.46
Abstract
OBJECTIVE
The purpose of this study was to develop a risk prediction score for distinguishing benign ovarian mass from malignant tumors using CA-125, human epididymis protein 4 (HE4), ultrasound findings, and menopausal status. The risk prediction score was compared to the risk of malignancy index and risk of ovarian malignancy algorithm (ROMA).
METHODS
This was a prospective, multicenter (n=6) study with patients from six Asian countries. Patients had a pelvic mass upon imaging and were scheduled to undergo surgery. Serum CA-125 and HE4 were measured on preoperative samples, and ultrasound findings were recorded. Regression analysis was performed and a risk prediction model was developed based on the significant factors. A bootstrap technique was applied to assess the validity of the HE4 model.
RESULTS
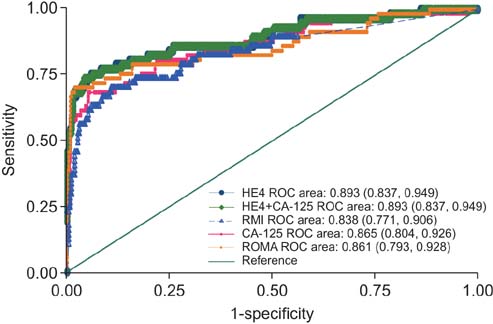
A total of 414 women with a pelvic mass were enrolled in the study, of which 328 had documented ultrasound findings. The risk prediction model that contained HE4, menopausal status, and ultrasound findings exhibited the best performance compared to models with CA-125 alone, or a combination of CA-125 and HE4. This model classified 77.2% of women with ovarian cancer as medium or high risk, and 86% of women with benign disease as very-low, low, or medium-low risk. This model exhibited better sensitivity than ROMA, but ROMA exhibited better specificity. Both models performed better than CA-125 alone.
CONCLUSION
Combining ultrasound with HE4 can improve the sensitivity for detecting ovarian cancer compared to other algorithms.
Keyword
MeSH Terms
-
Adult
*Algorithms
Biomarkers, Tumor/*blood
CA-125 Antigen/blood
Decision Support Techniques
Diagnosis, Differential
Female
Humans
Menopause
Middle Aged
Ovarian Neoplasms/*diagnosis/ultrasonography
Predictive Value of Tests
Prospective Studies
Proteins/*analysis
ROC Curve
Risk Assessment/methods
Sensitivity and Specificity
Biomarkers, Tumor
CA-125 Antigen
Proteins
Figure
Cited by 1 articles
-
The power of the Risk of Ovarian Malignancy Algorithm considering menopausal status: a comparison with CA 125 and HE4
Kyung Hee Han, Noh Hyun Park, Jin Ju Kim, Sunmie Kim, Hee Seung Kim, Maria Lee, Yong Sang Song
J Gynecol Oncol. 2019;30(6):. doi: 10.3802/jgo.2019.30.e83.
Reference
-
1. Jemal A, Siegel R, Ward E, Hao Y, Xu J, Thun MJ. Cancer statistics, 2009. CA Cancer J Clin. 2009; 59:225–249.2. du Bois A, Rochon J, Pfisterer J, Hoskins WJ. Variations in institutional infrastructure, physician specialization and experience, and outcome in ovarian cancer: a systematic review. Gynecol Oncol. 2009; 112:422–436.3. Bristow RE, Chang J, Ziogas A, Anton-Culver H. Adherence to treatment guidelines for ovarian cancer as a measure of quality care. Obstet Gynecol. 2013; 121:1226–1234.4. Jacobs I, Oram D, Fairbanks J, Turner J, Frost C, Grudzinskas JG. A risk of malignancy index incorporating CA 125, ultrasound and menopausal status for the accurate preoperative diagnosis of ovarian cancer. Br J Obstet Gynaecol. 1990; 97:922–929.5. Moore RG, McMeekin DS, Brown AK, DiSilvestro P, Miller MC, Allard WJ, et al. A novel multiple marker bioassay utilizing HE4 and CA125 for the prediction of ovarian cancer in patients with a pelvic mass. Gynecol Oncol. 2009; 112:40–46.6. Abraham J. OVA1 test for preoperative assessment of ovarian cancer. Community Oncol. 2010; 7:249–251.7. Nunes N, Yazbek J, Ambler G, Hoo W, Naftalin J, Jurkovic D. Prospective evaluation of the IOTA logistic regression model LR2 for the diagnosis of ovarian cancer. Ultrasound Obstet Gynecol. 2012; 40:355–359.8. Jacobs I, Bast RC Jr. The CA 125 tumour-associated antigen: a review of the literature. Hum Reprod. 1989; 4:1–12.9. Escudero JM, Auge JM, Filella X, Torne A, Pahisa J, Molina R. Comparison of serum human epididymis protein 4 with cancer antigen 125 as a tumor marker in patients with malignant and nonmalignant diseases. Clin Chem. 2011; 57:1534–1544.10. Gao L, Cheng HY, Dong L, Ye X, Liu YN, Chang XH, et al. The role of HE4 in ovarian cancer: inhibiting tumour cell proliferation and metastasis. J Int Med Res. 2011; 39:1645–1660.11. Moore RG, Brown AK, Miller MC, Skates S, Allard WJ, Verch T, et al. The use of multiple novel tumor biomarkers for the detection of ovarian carcinoma in patients with a pelvic mass. Gynecol Oncol. 2008; 108:402–408.12. Moore RG, Miller MC, Disilvestro P, Landrum LM, Gajewski W, Ball JJ, et al. Evaluation of the diagnostic accuracy of the risk of ovarian malignancy algorithm in women with a pelvic mass. Obstet Gynecol. 2011; 118:280–288.13. Kim YM, Whang DH, Park J, Kim SH, Lee SW, Park HA, et al. Evaluation of the accuracy of serum human epididymis protein 4 in combination with CA125 for detecting ovarian cancer: a prospective case-control study in a Korean population. Clin Chem Lab Med. 2011; 49:527–534.14. Ruggeri G, Bandiera E, Zanotti L, Belloli S, Ravaggi A, Romani C, et al. HE4 and epithelial ovarian cancer: comparison and clinical evaluation of two immunoassays and a combination algorithm. Clin Chim Acta. 2011; 412:1447–1453.15. Bandiera E, Romani C, Specchia C, Zanotti L, Galli C, Ruggeri G, et al. Serum human epididymis protein 4 and risk for ovarian malignancy algorithm as new diagnostic and prognostic tools for epithelial ovarian cancer management. Cancer Epidemiol Biomarkers Prev. 2011; 20:2496–2506.16. Moore RG, Jabre-Raughley M, Brown AK, Robison KM, Miller MC, Allard WJ, et al. Comparison of a novel multiple marker assay vs the Risk of Malignancy Index for the prediction of epithelial ovarian cancer in patients with a pelvic mass. Am J Obstet Gynecol. 2010; 203:228.17. Karlsen MA, Sandhu N, Hogdall C, Christensen IJ, Nedergaard L, Lundvall L, et al. Evaluation of HE4, CA125, risk of ovarian malignancy algorithm (ROMA) and risk of malignancy index (RMI) as diagnostic tools of epithelial ovarian cancer in patients with a pelvic mass. Gynecol Oncol. 2012; 127:379–383.18. Kaijser J, Van Gorp T, Van Hoorde K, Van Holsbeke C, Sayasneh A, Vergote I, et al. A comparison between an ultrasound based prediction model (LR2) and the risk of ovarian malignancy algorithm (ROMA) to assess the risk of malignancy in women with an adnexal mass. Gynecol Oncol. 2013; 129:377–383.19. Chan KK, Chen CA, Nam JH, Ochiai K, Wilailak S, Choon AT, et al. The use of HE4 in the prediction of ovarian cancer in Asian women with a pelvic mass. Gynecol Oncol. 2013; 128:239–244.20. Pencina MJ, D'Agostino RB Sr, D'Agostino RB Jr, Vasan RS. Evaluating the added predictive ability of a new marker: from area under the ROC curve to reclassification and beyond. Stat Med. 2008; 27:157–172.21. Harrell FE Jr, Lee KL, Mark DB. Multivariable prognostic models: issues in developing models, evaluating assumptions and adequacy, and measuring and reducing errors. Stat Med. 1996; 15:361–387.22. Schumacher M, Hollander N, Sauerbrei W. Resampling and crossvalidation techniques: a tool to reduce bias caused by model building? Stat Med. 1997; 16:2813–2827.23. Kim KH, Zsebik GN, Straughn JM Jr, Landen CN Jr. Management of complex pelvic masses using a multivariate index assay: a decision analysis. Gynecol Oncol. 2012; 126:364–368.24. Stiekema A, Lok CA, Kenter GG, van Driel WJ, Vincent AD, Korse CM. A predictive model combining human epididymal protein 4 and radiologic features for the diagnosis of ovarian cancer. Gynecol Oncol. 2014; 132:573–577.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Human Epididymis Protein 4 as a Diagnostic Marker of Ovarian Cancer and Its Reference Interval in Korean Population
- Comparison of HE4, CA125, and Risk of Ovarian Malignancy Algorithm in the Prediction of Ovarian Cancer in Korean Women
- Discrimination between Benign and Malignant Pelvic Masses Using the Risk of Malignancy Index 1
- Performance Evaluation of the ARCHITECT HE4 Assay
- Four risk of malignancy indices in evaluation of pelvic masses