Immune Netw.
2012 Apr;12(2):58-65. 10.4110/in.2012.12.2.58.
Reactivation of Silenced WT1 Transgene by Hypomethylating Agents: Implications for in vitro Modeling of Chemoimmunotherapy
- Affiliations
-
- 1Laboratory of Hematological Disease and Transplant Immunology, Seoul St. Mary's Hospital, College of Medicine, The Catholic University of Korea, Seoul 137-701, Korea. yoojink@catholic.ac.kr
- 2Department of Hematology, Catholic Blood and Marrow Transplantation Center, Seoul St. Mary's Hospital, College of Medicine, The Catholic University of Korea, Seoul 137-701, Korea.
- KMID: 2150735
- DOI: http://doi.org/10.4110/in.2012.12.2.58
Abstract
- BACKGROUND
A cell line with transfected Wilms' tumor protein 1 (WT1) is has been used for the preclinical evaluation of novel treatment strategies of WT1 immunotherapy for leukemia due to the lack of appropriate murine leukemia cell line with endogenous WT1. However, silencing of the transgene occurs. Regarding the effects of hypomethylating agents (HMAs) on reactivation of silenced genes, HMAs are considered to be immune enhancers.
METHODS
We treated murine WT1- transfected C1498 (mWT1-C1498) with increasing doses of decitabine (DAC) and azacitidine (AZA) to analyze their effects on transgene reactivation.
RESULTS
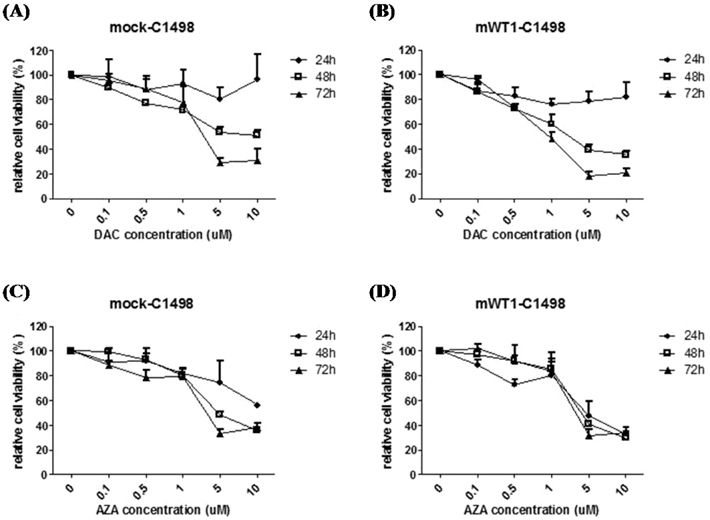
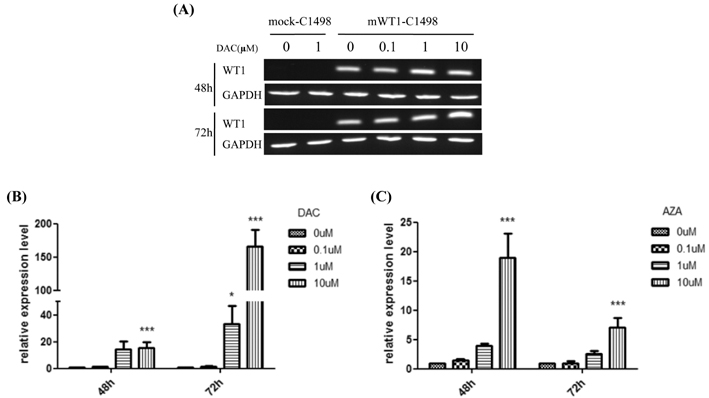
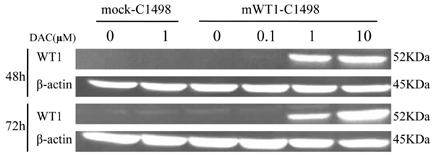
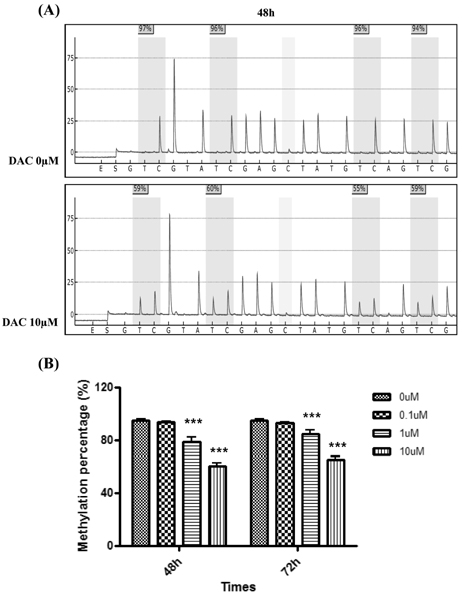
DAC and AZA decreased the number of viable cells in a dose- or time-dependent manner. Quantification of WT1 mRNA level was analyzed by real-time polymerase chain reaction after mWT1-C1498 treated with increasing dose of HMA. DAC treatment for 48 h induced 1.4-, 14.6-, and 15.5-fold increment of WT1 mRNA level, compared to untreated sample, at 0.1, 1, and 10microM, respectively. Further increment of WT1 expression in the presence of 1 and 10microM DAC was evident at 72 h. AZA treatment also induced up-regulation of mRNA, but not to the same degree as with DAC treatment. The correlation between the incremental increases in WT1 mRNA by DAC was confirmed by Western blot and concomitant down-regulation of WT1 promoter methylation was revealed.
CONCLUSION
The in vitro data show that HMA can induce reactivation of WT1 transgene and that DAC is more effective, at least in mWT1-C1498 cells, which suggests that the combination of DAC and mWT1-C1498 can be used for the development of the experimental model of HMA-combined WT1 immunotherapy targeting leukemia.
Keyword
MeSH Terms
Figure
Reference
-
1. Roddie C, Peggs KS. Donor lymphocyte infusion following allogeneic hematopoietic stem cell transplantation. Expert Opin Biol Ther. 2011. 11:473–487.
Article2. Cheever MA, Allison JP, Ferris AS, Finn OJ, Hastings BM, Hecht TT, Mellman I, Prindiville SA, Viner JL, Weiner LM, Matrisian LM. The prioritization of cancer antigens: a national cancer institute pilot project for the acceleration of translational research. Clin Cancer Res. 2009. 15:5323–5337.
Article3. Sugiyama H. WT1 (Wilms' tumor gene 1): biology and cancer immunotherapy. Jpn J Clin Oncol. 2010. 40:377–387.
Article4. Menssen HD, Renkl HJ, Rodeck U, Kari C, Schwartz S, Thiel E. Detection by monoclonal antibodies of the Wilms' tumor (WT1) nuclear protein in patients with acute leukemia. Int J Cancer. 1997. 70:518–523.
Article5. Baird PN, Simmons PJ. Expression of the Wilms' tumor gene (WT1) in normal hemopoiesis. Exp Hematol. 1997. 25:312–320.6. Yamagami T, Sugiyama H, Inoue K, Ogawa H, Tatekawa T, Hirata M, Kudoh T, Akiyama T, Murakami A, Maekawa T. Growth inhibition of human leukemic cells by WT1 (Wilms tumor gene) antisense oligodeoxynucleotides: implications for the involvement of WT1 in leukemogenesis. Blood. 1996. 87:2878–2884.
Article7. Scheibenbogen C, Letsch A, Thiel E, Schmittel A, Mailaender V, Baerwolf S, Nagorsen D, Keilholz U. CD8 T-cell responses to Wilms tumor gene product WT1 and proteinase 3 in patients with acute myeloid leukemia. Blood. 2002. 100:2132–2137.
Article8. Rezvani K, Brenchley JM, Price DA, Kilical Y, Gostick E, Sewell AK, Li J, Mielke S, Douek DC, Barrett AJ. T-cell responses directed against multiple HLA-A*0201-restricted epitopes derived from Wilms' tumor 1 protein in patients with leukemia and healthy donors: identification, quantification, and characterization. Clin Cancer Res. 2005. 11:8799–8807.
Article9. Ohminami H, Yasukawa M, Fujita S. HLA class I-restricted lysis of leukemia cells by a CD8(+) cytotoxic T-lymphocyte clone specific for WT1 peptide. Blood. 2000. 95:286–293.
Article10. Mailänder V, Scheibenbogen C, Thiel E, Letsch A, Blau IW, Keilholz U. Complete remission in a patient with recurrent acute myeloid leukemia induced by vaccination with WT1 peptide in the absence of hematological or renal toxicity. Leukemia. 2004. 18:165–166.
Article11. Rezvani K, Yong AS, Mielke S, Savani BN, Musse L, Superata J, Jafarpour B, Boss C, Barrett AJ. Leukemia-associated antigen-specific T-cell responses following combined PR1 and WT1 peptide vaccination in patients with myeloid malignancies. Blood. 2007. 111:236–242.
Article12. Van Tendeloo VF, Van de Velde A, Van Driessche A, Cools N, Anguille S, Ladell K, Gostick E, Vermeulen K, Pieters K, Nijs G, Stein B, Smits EL, Schroyens WA, Gadisseur AP, Vrelust I, Jorens PG, Goossens H, de Vries IJ, Price DA, Oji Y, Oka Y, Sugiyama H, Berneman ZN. Induction of complete and molecular remissions in acute myeloid leukemia by Wilms' tumor 1 antigen-targeted dendritic cell vaccination. Proc Natl Acad Sci U S A. 2010. 107:13824–13829.
Article13. Keilholz U, Letsch A, Busse A, Asemissen AM, Bauer S, Blau IW, Hofmann WK, Uharek L, Thiel E, Scheibenbogen C. A clinical and immunologic phase 2 trial of Wilms tumor gene product 1 (WT1) peptide vaccination in patients with AML and MDS. Blood. 2009. 113:6541–6548.
Article14. Kim YJ, Cho SG, Lee S, Kim MS, Kim EK, Cho BS, Sohn HJ, Choi HB, Eom KS, Min CK, Kim HJ, Kim YG, Kim DW, Lee JW, Min WS, Kim CC, Kim TG. Potential role of adoptively transferred allogeneic WT1-specific CD4+ and CD8+ T lymphocytes for the sustained remission of refractory AML. Bone Marrow Transplant. 2010. 45:597–599.
Article15. Van Driessche A, Berneman ZN, Van Tendeloo VF. Active specific immunotherapy targeting the Wilms' tumor protein 1 (WT1) for patients with hematological malignancies and solid tumors: lessons from early clinical trials. Oncologist. 2012. 17:250–259.
Article16. Buckler AJ, Pelletier J, Haber DA, Glaser T, Housman DE. Isolation, characterization, and expression of the murine Wilms' tumor gene (WT1) during kidney development. Mol Cell Biol. 1991. 11:1707–1712.
Article17. Fraizer GC, Patmasiriwat P, Zhang X, Saunders GF. Expression of the tumor suppressor gene WT1 in both human and mouse bone marrow. Blood. 1995. 86:4704–4706.
Article18. Oka Y, Udaka K, Tsuboi A, Elisseeva OA, Ogawa H, Aozasa K, Kishimoto T, Sugiyama H. Cancer immunotherapy targeting Wilms' tumor gene WT1 product. J Immunol. 2000. 164:1873–1880.
Article19. Houdebine LM. Transgenic animal bioreactors. Transgenic Res. 2000. 9:305–320.20. Migliaccio AR, Bengra C, Ling J, Pi W, Li C, Zeng S, Keskintepe M, Whitney B, Sanchez M, Migliaccio G, Tuan D. Stable and unstable transgene integration sites in the human genome: extinction of the Green Fluorescent Protein transgene in K562 cells. Gene. 2000. 256:197–214.
Article21. Cedar H. DNA methylation and gene activity. Cell. 1988. 53:3–4.
Article22. Challita PM, Kohn DB. Lack of expression from a retroviral vector after transduction of murine hematopoietic stem cells is associated with methylation in vivo. Proc Natl Acad Sci U S A. 1994. 91:2567–2571.
Article23. Hoeben RC, Migchielsen AA, van der Jagt RC, van Ormondt H, van der Eb AJ. Inactivation of the Moloney murine leukemia virus long terminal repeat in murine fibroblast cell lines is associated with methylation and dependent on its chromosomal position. J Virol. 1991. 65:904–912.
Article24. Jaenisch R, Schnieke A, Harbers K. Treatment of mice with 5-azacytidine efficiently activates silent retroviral genomes in different tissues. Proc Natl Acad Sci U S A. 1985. 82:1451–1455.
Article25. Rosenfeld C, Cheever MA, Gaiger A. WT1 in acute leukemia, chronic myelogenous leukemia and myelodysplastic syndrome: therapeutic potential of WT1 targeted therapies. Leukemia. 2003. 17:1301–1312.
Article26. Lake RA, Robinson BW. Immunotherapy and chemotherapy--a practical partnership. Nat Rev Cancer. 2005. 5:397–405.
Article27. Kim HS, Park HM, Park JS, Sohn HJ, Kim SG, Kim HJ, Oh ST, Kim TG. Dendritic cell vaccine in addition to FOLFIRI regimen improve antitumor effects through the inhibition of immunosuppressive cells in murine colorectal cancer model. Vaccine. 2010. 28:7787–7796.
Article28. Parikh SA, Keating MJ, O'Brien S, Wang X, Ferrajoli A, Faderl S, Burger J, Koller C, Estrov Z, Badoux X, Lerner S, Wierda WG. Frontline chemoimmunotherapy with fludarabine, cyclophosphamide, alemtuzumab, and rituximab for high-risk chronic lymphocytic leukemia. Blood. 2011. 118:2062–2068.
Article29. Guo ZS, Hong JA, Irvine KR, Chen GA, Spiess PJ, Liu Y, Zeng G, Wunderlich JR, Nguyen DM, Restifo NP, Schrump DS. De novo induction of a cancer/testis antigen by 5-aza-2'-deoxycytidine augments adoptive immunotherapy in a murine tumor model. Cancer Res. 2006. 66:1105–1113.
Article30. Fonsatti E, Nicolay HJ, Sigalotti L, Calabrò L, Pezzani L, Colizzi F, Altomonte M, Guidoboni M, Marincola FM, Maio M. Functional up-regulation of human leukocyte antigen class I antigens expression by 5-aza-2'-deoxycytidine in cutaneous melanoma: immunotherapeutic implications. Clin Cancer Res. 2007. 13:3333–3338.
Article31. Sigalotti L, Altomonte M, Colizzi F, Degan M, Rupolo M, Zagonel V, Pinto A, Gattei V, Maio M. 5-Aza-2'-deoxycytidine (decitabine) treatment of hematopoietic malignancies: a multimechanism therapeutic approach? Blood. 2003. 101:4644–4646.
Article32. Choi J, Ritchey J, Prior JL, Holt M, Shannon WD, Deych E, Piwnica-Worms DR, DiPersio JF. In vivo administration of hypomethylating agents mitigate graft-versus-host disease without sacrificing graft-versus-leukemia. Blood. 2010. 116:129–139.
Article33. Goodyear O, Agathanggelou A, Novitzky-Basso I, Siddique S, McSkeane T, Ryan G, Vyas P, Cavenagh J, Stankovic T, Moss P, Craddock C. Induction of a CD8+ T-cell response to the MAGE cancer testis antigen by combined treatment with azacitidine and sodium valproate in patients with acute myeloid leukemia and myelodysplasia. Blood. 2010. 116:1908–1918.
Article34. Gaiger A, Reese V, Disis ML, Cheever MA. Immunity to WT1 in the animal model and in patients with acute myeloid leukemia. Blood. 2000. 96:1480–1489.
Article35. Hollenbach PW, Nguyen AN, Brady H, Williams M, Ning Y, Richard N, Krushel L, Aukerman SL, Heise C, MacBeth KJ. A comparison of azacitidine and decitabine activities in acute myeloid leukemia cell lines. PLoS One. 2010. 5:e9001.
Article36. Grassi G, Maccaroni P, Meyer R, Kaiser H, D'Ambrosio E, Pascale E, Grassi M, Kuhn A, Di Nardo P, Kandolf R, Küpper JH. Inhibitors of DNA methylation and histone deacetylation activate cytomegalovirus promoter-controlled reporter gene expression in human glioblastoma cell line U87. Carcinogenesis. 2003. 24:1625–1635.
Article37. Swindle CS, Klug CA. Mechanisms that regulate silencing of gene expression from retroviral vectors. J Hematother Stem Cell Res. 2002. 11:449–456.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Enhancement of Radiosensitivity by DNA Hypomethylating Drugs through Apoptosis and Autophagy in Human Sarcoma Cells
- Reactivation of Hepatitis B Virus and Its Prevention in Patients with Rheumatic Diseases Receiving Immunosuppressive Therapy
- Prevention of Hepatitis B reactivation in the setting of immunosuppression
- The Diagnostic Utility of Mesothelial Markers in Distinguishing between Reactive Mesothelial Cell and Adenocarcinoma Cells in Serous Effusions with Cytospin Preparation
- Prognostic Significance of WT1 Gene Expression in Patients with Acute Leukemia