Immune Netw.
2011 Feb;11(1):42-49. 10.4110/in.2011.11.1.42.
Anti-inflammatory and Anti-oxidative Effects of Korean Red Ginseng Extract in Human Keratinocytes
- Affiliations
-
- 1Department of Pharmacy, Yeungnam University, Gyeongsan 712-749, Korea.
- 2Department of Herbal Medicinal Pharmacology, Daegu Haany University, Gyeongsan 712-715, Korea. lyu@dhu.ac.kr
- KMID: 2150691
- DOI: http://doi.org/10.4110/in.2011.11.1.42
Abstract
- BACKGROUND
In this study, we have investigated the effect of Korean red ginseng (KRG) extracts on the production of TNF-alpha and IL-8 in human keratinocytes. Also, to examine the antioxidative effect of red ginseng extracts, free radical scavenging activity and superoxide dismutase (SOD) activity in human dermal fibroblasts was measured.
METHODS
To investigate the effect of KRG in atopic dermatitis, we measured the level of TNF-alpha and IL-8 secretion in LPS-stimulated human keratinocytes after the treatment of KRG extracts using enzyme-linked immunosorbent assay. Anti-oxidative activity was investigated by measuring 1,1-diphenyl-2-picrylhydrazyl (DPPH) radical scavenging and SOD activity.
RESULTS
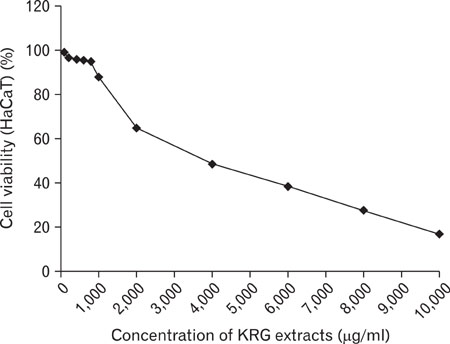
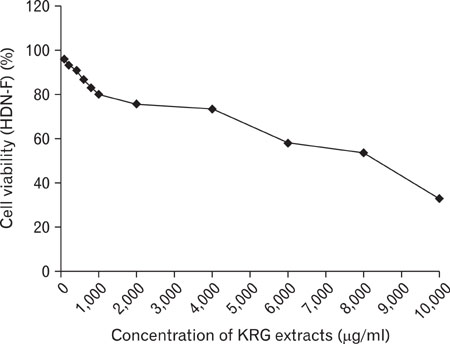
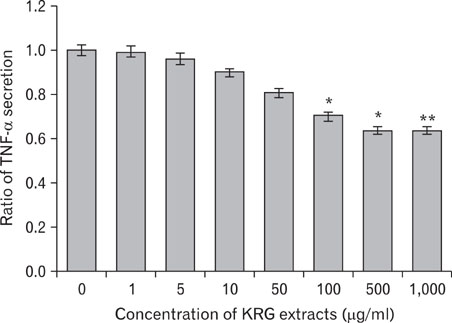
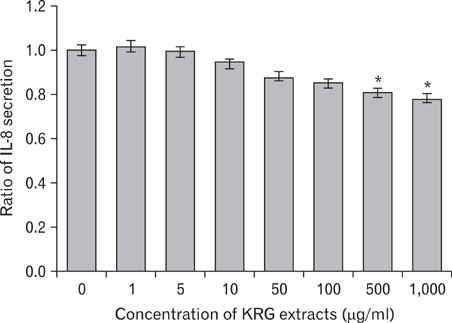
The stimulation of human keratinocytes with KRG extracts shifted the LPS-induced cytokine secretion toward a more immunosuppressive response. KRG dose-dependently decreased TNF-alpha and IL-8 production in HaCaT cells and a significant inhibition of TNF-alpha was shown when cells were treated with 500 and 1,000 microg/ml of KRG extracts. Additionally, KRG extracts showed DPPH radical scavenging and SOD activity in a dose-dependent manner. Particularly, SOD activities of concentrations higher than 60 microg/ml of KRG extracts were significantly different in human dermal fibroblast cells.
CONCLUSION
Based on this study, KRG extracts may be a useful immunosuppressive agent in the treatment of atopic dermatitis.
Keyword
MeSH Terms
Figure
Reference
-
1. Williams H, Stewart A, von Mutius E, Cookson W, Anderson HR. International Study of Asthma and Allergies in Childhood (ISAAC) Phase One and Three Study Groups. Is eczema really on the increase worldwide? J Allergy Clin Immunol. 2008. 121:947–954.
Article2. Spergel JM. From atopic dermatitis to asthma: the atopic march. Ann Allergy Asthma Immunol. 2010. 105:99–106.
Article3. Spergel JM, Paller AS. Atopic dermatitis and the atopic march. J Allergy Clin Immunol. 2003. 112:6 Suppl. S118–S127.
Article4. Homey B, Steinhoff M, Ruzicka T, Leung DY. Cytokines and chemokines orchestrate atopic skin inflammation. J Allergy Clin Immunol. 2006. 118:178–189.
Article5. Pivarcsi A, Nagy I, Kemeny L. Innate immunity in the skin: how keratinocytes fight against pathogens. Curr Immunol Rev. 2005. 1:29–42.
Article6. Feliciani C, Gupta AK, Sauder DN. Keratinocytes and cytokine/growth factors. Crit Rev Oral Biol Med. 1996. 7:300–318.
Article7. Beutler B, Cerami A. Oppenheim II, Shevach EM, editors. Cachectin (tumor necrosis factor): an endogenous mediator of shock and inflammatory response. Immunophysiology; the role of cells and cytokines in immunity and inflammation. 1990. New York: Oxford University Press;226–237.8. Tracey KJ, Vlassara H, Cerami A. Cachectin/tumour necrosis factor. Lancet. 1989. 1(8647):1122–1126.9. Yoshimura T, Matsushima K, Tanaka S, Robinson EA, Appella E, Oppenheim JJ, Leonard EJ. Purification of a human monocyte-derived neutrophil chemotactic factor that has peptide sequence similarity to other host defense cytokines. Proc Natl Acad Sci U S A. 1987. 84:9233–9237.
Article10. Leonard EJ, Yoshimura T, Tanaka S, Raffeld M. Neutrophil recruitment by intradermally injected neutrophil attractant/activation protein-1. J Invest Dermatol. 1991. 96:690–694.
Article11. Wismer JM, McKenzie RC, Sauder DN. Interleukin-8 immunoreactivity in epidermis of cutaneous T-cell lymphoma patients. Lymphokine Cytokine Res. 1994. 13:21–27.12. Schröder JM, Christophers E. Identification of C5ades arg and an anionic neutrophil-activating peptide (ANAP) in psoriatic scales. J Invest Dermatol. 1986. 87:53–58.
Article13. Sies H. Sies H, editor. Oxidative stress: introductory remarks. Oxidative Stress. 1985. London: Academic Press;1–7.
Article14. Nakashima T, Sato E, Niwano Y, Kohno M, Muraoka W, Oda T. Inhibitory or scavenging action of ketoconazole and ciclopiroxolamine against reactive oxygen species released by primed inflammatory cells. Br J Dermatol. 2007. 156:720–727.
Article15. Cochrane CG. Cellular injury by oxidants. Am J Med. 1991. 91(3C):23S–30S.
Article16. Weiss SJ. Tissue destruction by neutrophils. N Engl J Med. 1989. 320:365–376.
Article17. Inoue Y, Isobe M, Shiohara T, Hayashi H. Inhibitory activity of CX-659S, a novel diaminouracil derivative, against the rebound phenomenon following withdrawal of corticosteroid therapy for chronic contact hypersensitivity responses. Int Arch Allergy Immunol. 2003. 131:143–152.
Article18. Artik S, Ruzicka T. Complementary therapy for atopic eczema and other allergic skin diseases. Dermatol Ther. 2003. 16:150–163.
Article19. Koo J, Desai R. Traditional Chinese medicine in dermatology. Dermatol Ther. 2003. 16:98–105.
Article20. Gao XK, Fuseda K, Shibata T, Tanaka H, Inagaki N, Nagai H. Kampo Medicines for Mite Antigen-Induced Allergic Dermatitis in NC/Nga Mice. Evid Based Complement Alternat Med. 2005. 2:191–199.
Article21. Bae EA, Trinh HT, Yoon HK, Kim DH. Compound K, a metabolite of ginsenoside Rb1, inhibits passive cutaneous anaphylaxis reaction in mice. J Ginseng Res. 2009. 33:93–98.
Article22. Ro JY, Ahn YS, Kim KH. Inhibitory effect of ginsenoside on the mediator release in the guinea pig lung mast cells activated by specific antigen-antibody reactions. Int J Immunopharmacol. 1998. 20:625–641.
Article23. Blois MS. Antioxidant determination by the use of a stable free radical. Nature. 1958. 181:1199–2004.
Article24. Beauchamp C, Fridovich I. Superoxide dismutase: improved assays and an assay applicable to acrylamide gels. Anal Biochem. 1971. 44:276–287.
Article25. Fivenson D, Arnold RJ, Kaniecki DJ, Cohen JL, Frech F, Finlay AY. The effect of atopic dermatitis on total burden of illness and quality of life on adults and children in a large managed care organization. J Manag Care Pharm. 2002. 8:333–342.
Article26. Lee JH, Cho SH. Korean red ginseng extract ameliorates skin lesions in NC/Nga mice: an atopic dermatitis model. J Ethnopharmacol. 2011. 133:810–817.
Article27. Leung DY, Boguniewicz M, Howell MD, Nomura I, Hamid QA. New insights into atopic dermatitis. J Clin Invest. 2004. 113:651–657.
Article28. Kang JS, Yoon WK, Han MH, Lee H, Lee CW, Lee KH, Han SB, Lee K, Yang KH, Park SK, Kim HM. Inhibition of atopic dermatitis by topical application of silymarin in NC/Nga mice. Int Immunopharmacol. 2008. 8:1475–1480.
Article29. Shibata S, Fujita M, Itokawa H, Tanaka O, Ishii T. Studies on the constituents of japanese and chinese crude drugs. Xi. Panaxadiol, a Sapogenin of Ginseng Roots. Chem Pharm Bull (Tokyo). 1963. 11:759–761.
Article30. Park EK, Choo MK, Han MJ, Kim DH. Ginsenoside Rh1 possesses antiallergic and anti-inflammatory activities. Int Arch Allergy Immunol. 2004. 133:113–120.
Article31. Köck A, Schwarz T, Kirnbauer R, Urbanski A, Perry P, Ansel JC, Luger TA. Human keratinocytes are a source for tumor necrosis factor alpha: evidence for synthesis and release upon stimulation with endotoxin or ultraviolet light. J Exp Med. 1990. 172:1609–1614.
Article32. Collins T, Lapierre LA, Fiers W, Strominger JL, Pober JS. Recombinant human tumor necrosis factor increases mRNA levels and surface expression of HLA-A,B antigens in vascular endothelial cells and dermal fibroblasts in vitro. Proc Natl Acad Sci U S A. 1986. 83:446–450.
Article33. Giustizieri ML, Mascia F, Frezzolini A, De Pità O, Chinni LM, Giannetti A, Girolomoni G, Pastore S. Keratinocytes from patients with atopic dermatitis and psoriasis show a distinct chemokine production profile in response to T cell-derived cytokines. J Allergy Clin Immunol. 2001. 107:871–877.
Article34. Albanesi C, Scarponi C, Sebastiani S, Cavani A, Federici M, De Pità O, Puddu P, Girolomoni G. IL-4 enhances keratinocyte expression of CXCR3 agonistic chemokines. J Immunol. 2000. 165:1395–1402.
Article35. Albanesi C, Cavani A, Girolomoni G. IL-17 is produced by nickel-specific T lymphocytes and regulates ICAM-1 expression and chemokine production in human keratinocytes: synergistic or antagonist effects with IFN-gamma and TNF-alpha. J Immunol. 1999. 162:494–502.36. Gillitzer R, Berger R, Mielke V, Müller C, Wolff K, Stingl G. Upper keratinocytes of psoriatic skin lesions express high levels of NAP-1/IL-8 mRNA in situ. J Invest Dermatol. 1991. 97:73–79.
Article37. Gottlieb AB, Luster AD, Posnett DN, Carter DM. Detection of a gamma interferon-induced protein IP-10 in psoriatic plaques. J Exp Med. 1988. 168:941–948.
Article38. Grewe M, Bruijnzeel-Koomen CA, Schöpf E, Thepen T, Langeveld-Wildschut AG, Ruzicka T, Krutmann J. A role for Th1 and Th2 cells in the immunopathogenesis of atopic dermatitis. Immunol Today. 1998. 19:359–361.
Article39. Miller DM, Buettner GR, Aust SD. Transition metals as catalysts of "autoxidation" reactions. Free Radic Biol Med. 1990. 8:95–108.
Article40. Slater TF. Free-radical mechanisms in tissue injury. Biochem J. 1984. 222:1–15.
Article41. Fridovich I. Biological effects of the superoxide radical. Arch Biochem Biophys. 1986. 247:1–11.
Article42. Niwa Y, Iizawa O. Abnormalities in serum lipids and leukocyte superoxide dismutase and associated cataract formation in patients with atopic dermatitis. Arch Dermatol. 1994. 130:1387–1392.
Article43. Hodgson EK, Fridovich I. The interaction of bovine erythrocyte superoxide dismutase with hydrogen peroxide: inactivation of the enzyme. Biochemistry. 1975. 14:5294–5299.
Article44. Kono Y, Fridovich I. Superoxide radical inhibits catalase. J Biol Chem. 1982. 257:5751–5754.
Article45. Niwa Y, Sumi H, Kawahira K, Terashima T, Nakamura T, Akamatsu H. Protein oxidative damage in the stratum corneum: Evidence for a link between environmental oxidants and the changing prevalence and nature of atopic dermatitis in Japan. Br J Dermatol. 2003. 149:248–254.
Article46. Cornelius LA, Li LJ, Sepp N, Lawley TJ, Caughman SW. The effect of ultraviolet B (UVB) and reactive oxygen intermediates (ROI) upon dermal microvascular endothelial cell (EC) adhesion molecule expression. J Invest Dermatol. 1992. 98:596.47. Gougerot-Pocidalo M, Revillard J. Fuchs J, Packer L, editors. Oxidative stress, cytokines, and lymphocyte activation. Oxidative Stress in Dermatology. 1993. New York: Marcel Dekker;187–211.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Korean Red Ginseng Tonic Extends Lifespan in D. melanogaster
- Anti-inflammatory effects of ethanolic extract of Annona muricata
- Effects of Ginseng on the Metabolism of Enflurane and Methoxyflurane
- Aster glehni Ethanol Extract Inhibits Inflammatory Responses Regulating Skin Barrier Molecules in Human Keratinocytes
- Effects of Red or Black Ginseng Extract in a Rat Model of Inflammatory Temporomandibular Joint Pain