Obstet Gynecol Sci.
2015 May;58(3):210-216. 10.5468/ogs.2015.58.3.210.
Long (27-nucleotides) small inhibitory RNAs targeting E6 protein eradicate effectively the cervical cancer cells harboring human papilloma virus
- Affiliations
-
- 1Department of Obstetrics and Gynecology, Liz & Marian Hospital, Cheonan, Korea.
- 2Department of Obstetrics and Gynecology, University of Ulsan College of Medicine, Asan Medical Center, Seoul, Korea. ytkim@amc.seoul.kr
- 3Genolution Inc., Seoul, Korea.
- KMID: 2148933
- DOI: http://doi.org/10.5468/ogs.2015.58.3.210
Abstract
OBJECTIVE
This study was to identify small inhibitory RNAs (siRNAs) that are effective in inhibiting growth of cervical cancer cell lines harboring human papilloma virus (HPV) and to examine how siRNAs interact with interferon beta (IFN-beta) and thimerosal.
METHODS
The HPV18-positive HeLa and C-4I cell lines were used. Four types of siRNAs were designed according to their target (both E6 and E7 vs. E6 only) and sizes (21- vs. 27-nucleotides); Ex-18E6/21, Ex-18E6/27, Sp-18E6/21, and Sp-18E6/27. Each siRNA-transfected cells were cultured with or without IFN-b and thimerosal and their viability was measured.
RESULTS
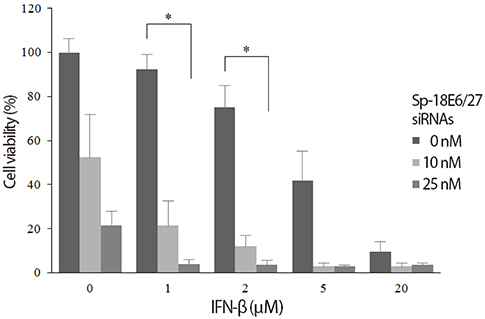
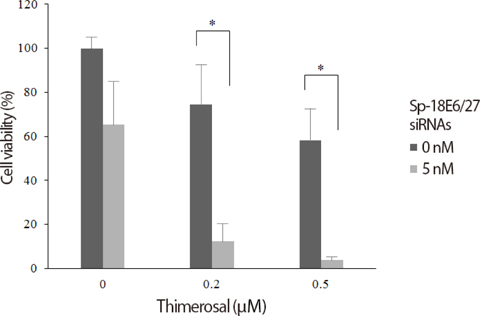
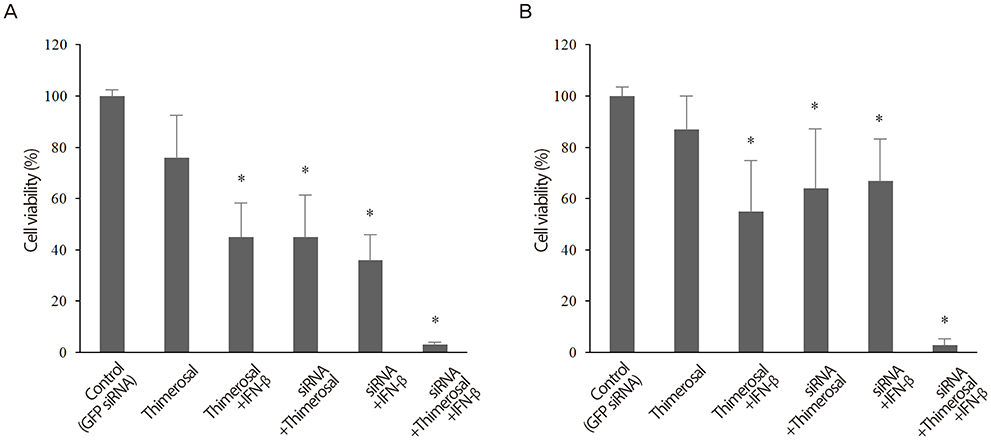
The viabilities of HPV18-positive tumor cells were reduced by 21- and 27-nucleotide siRNAs in proportion to the siRNA concentrations. Of the two types of siRNAs, the 27-nucleotide siRNA constructs showed greater inhibitory efficacy. Sp-18E6 siRNAs, which selectively downregulates E6 protein only, were more effective than the E6- and E7-targeting Ex-18E6 siRNAs. siRNAs and IFN-beta showed the synergistic effect to inhibit HeLa cell survival and the effect was proportional to both siRNA and IFN-beta concentrations. Thimerosal in the presence of siRNA exerted a dose-dependent inhibition of C-4I cell survival. Finally, co-treatment with siRNA, IFN-beta, and thimerosal induced the most profound decrease in the viability of both cell lines.
CONCLUSION
Long (27-nucleotides) siRNAs targeting E6-E7 mRNAs effectively reduce the viability of HPV18-positive cervical cancer cells and show the synergistic effect in combination with IFN-b and thimerosal. It is necessary to find the rational design of siRNAs and effective co-factors to eradicate particular cervical cancer.
Keyword
MeSH Terms
Figure
Reference
-
1. Bosch FX, de Sanjose S. The epidemiology of human papillomavirus infection and cervical cancer. Dis Markers. 2007; 23:213–227.2. Manavi M, Hudelist G, Fink-Retter A, Gschwantler-Kaulich D, Pischinger K, Czerwenka K. Human papillomavirus DNA integration and messenger RNA transcription in cervical low- and high-risk squamous intraepithelial lesions in Austrian women. Int J Gynecol Cancer. 2008; 18:285–294.3. Khan MJ, Castle PE, Lorincz AT, Wacholder S, Sherman M, Scott DR, et al. The elevated 10-year risk of cervical precancer and cancer in women with human papillomavirus (HPV) type 16 or 18 and the possible utility of type-specific HPV testing in clinical practice. J Natl Cancer Inst. 2005; 97:1072–1079.4. Munger K, Baldwin A, Edwards KM, Hayakawa H, Nguyen CL, Owens M, et al. Mechanisms of human papillomavirus-induced oncogenesis. J Virol. 2004; 78:11451–11460.5. Elbashir SM, Harborth J, Lendeckel W, Yalcin A, Weber K, Tuschl T. Duplexes of 21-nucleotide RNAs mediate RNA interference in cultured mammalian cells. Nature. 2001; 411:494–498.6. Amarzguioui M, Holen T, Babaie E, Prydz H. Tolerance for mutations and chemical modifications in a siRNA. Nucleic Acids Res. 2003; 31:589–595.7. Ghittoni R, Accardi R, Hasan U, Gheit T, Sylla B, Tommasino M. The biological properties of E6 and E7 oncoproteins from human papillomaviruses. Virus Genes. 2010; 40:1–13.8. Magaldi TG, Almstead LL, Bellone S, Prevatt EG, Santin AD, DiMaio D. Primary human cervical carcinoma cells require human papillomavirus E6 and E7 expression for ongoing proliferation. Virology. 2012; 422:114–124.9. Grassmann K, Rapp B, Maschek H, Petry KU, Iftner T. Identification of a differentiation-inducible promoter in the E7 open reading frame of human papillomavirus type 16 (HPV-16) in raft cultures of a new cell line containing high copy numbers of episomal HPV-16 DNA. J Virol. 1996; 70:2339–2349.10. Jiang M, Milner J. Selective silencing of viral gene expression in HPV-positive human cervical carcinoma cells treated with siRNA, a primer of RNA interference. Oncogene. 2002; 21:6041–6048.11. Niu XY, Peng ZL, Duan WQ, Wang H, Wang P. Inhibition of HPV 16 E6 oncogene expression by RNA interference in vitro and in vivo. Int J Gynecol Cancer. 2006; 16:743–751.12. DeFilippis RA, Goodwin EC, Wu L, DiMaio D. Endogenous human papillomavirus E6 and E7 proteins differentially regulate proliferation, senescence, and apoptosis in HeLa cervical carcinoma cells. J Virol. 2003; 77:1551–1563.13. Stark GR, Kerr IM, Williams BR, Silverman RH, Schreiber RD. How cells respond to interferons. Annu Rev Biochem. 1998; 67:227–264.14. Minks MA, Benvin S, Maroney PA, Baglioni C. Synthesis of 2'5'-oligo(A) in extracts of interferon-treated HeLa cells. J Biol Chem. 1979; 254:5058–5064.15. Elbashir SM, Martinez J, Patkaniowska A, Lendeckel W, Tuschl T. Functional anatomy of siRNAs for mediating efficient RNAi in Drosophila melanogaster embryo lysate. EMBO J. 2001; 20:6877–6888.16. Kim DH, Behlke MA, Rose SD, Chang MS, Choi S, Rossi JJ. Synthetic dsRNA Dicer substrates enhance RNAi potency and efficacy. Nat Biotechnol. 2005; 23:222–226.17. Chiantore MV, Vannucchi S, Accardi R, Tommasino M, Percario ZA, Vaccari G, et al. Interferon-β induces cellular senescence in cutaneous human papilloma virus-transformed human keratinocytes by affecting p53 transactivating activity. PLoS One. 2012; 7:e36909.18. Gonzalez-Sanchez JL, Martinez-Chequer JC, Hernandez-Celaya ME, Barahona-Bustillos E, Andrade-Manzano AF. Randomized placebo-controlled evaluation of intramuscular interferon beta treatment of recurrent human papillomavirus. Obstet Gynecol. 2001; 97:621–624.19. Yang J, Pu YG, Zeng ZM, Yu ZJ, Huang N, Deng QW. Interferon for the treatment of genital warts: a systematic review. BMC Infect Dis. 2009; 9:156.20. Parran DK, Barker A, Ehrich M. Effects of thimerosal on NGF signal transduction and cell death in neuroblastoma cells. Toxicol Sci. 2005; 86:132–140.21. Woo KJ, Lee TJ, Bae JH, Jang BC, Song DK, Cho JW, et al. Thimerosal induces apoptosis and G2/M phase arrest in human leukemia cells. Mol Carcinog. 2006; 45:657–666.22. Koivusalo R, Krausz E, Helenius H, Hietanen S. Chemotherapy compounds in cervical cancer cells primed by reconstitution of p53 function after short interfering RNAmediated degradation of human papillomavirus 18 E6 mRNA: opposite effect of siRNA in combination with different drugs. Mol Pharmacol. 2005; 68:372–382.23. Putral LN, Bywater MJ, Gu W, Saunders NA, Gabrielli BG, Leggatt GR, et al. RNA interference against human papillomavirus oncogenes in cervical cancer cells results in increased sensitivity to cisplatin. Mol Pharmacol. 2005; 68:1311–1319.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- HPV E6 antisense induces apoptosis in CaSki cells via suppression of E6 splicing
- The Detection of the p53 Protein in Cervical Cancer and CIN by Immunohistochemistry
- Roles of Oncogenic Long Non-coding RNAs in Cancer Development
- Functional analysis of CDC7 protein which is interacted with HPV E6
- Potential Implications of Long Noncoding RNAs in Autoimmune Diseases