Korean J Urol.
2009 Jan;50(1):81-88. 10.4111/kju.2009.50.1.81.
Effect of Decursin on the Expression of beta-Catenin and Matrix Metalloproteinase-7 in Prostate Cancer Cell Lines
- Affiliations
-
- 1Department of Urology, College of Medicine, Inje University, Busan, Korea. uroOkang@hanmail.net
- 2Paik Institute of Clinical Research, College of Medicine, Inje University, Busan, Korea.
- 3Pharmaco Genomics Research Center, College of Medicine, Inje University, Busan, Korea.
- 4College of Pharmacy, Chungnam National University, Daejeon, Korea.
- KMID: 2140119
- DOI: http://doi.org/10.4111/kju.2009.50.1.81
Abstract
-
PURPOSE: Alterations in the Wnt/beta-catenin pathway are associated with the development and progression of human prostate cancer. Decursin can attenuate the Wnt/beta-catenin pathway. We investigated the relationship between the Wnt/beta-catenin pathway and decursin in prostate cancer cells.
MATERIALS AND METHODS
PC-3 and LNCaP cell lines were used. Cell viability was measured with methyl-thiazole tetrazolium bromide (MTT) assays, and cell apoptosis analysis was performed by FACScan. The amount of beta-catenin protein after treatment with decursin was measured by Western blot analysis. Expression of MMP-7 mRNA was detected by real-time polymerase chain reaction (RT-PCR).
RESULTS
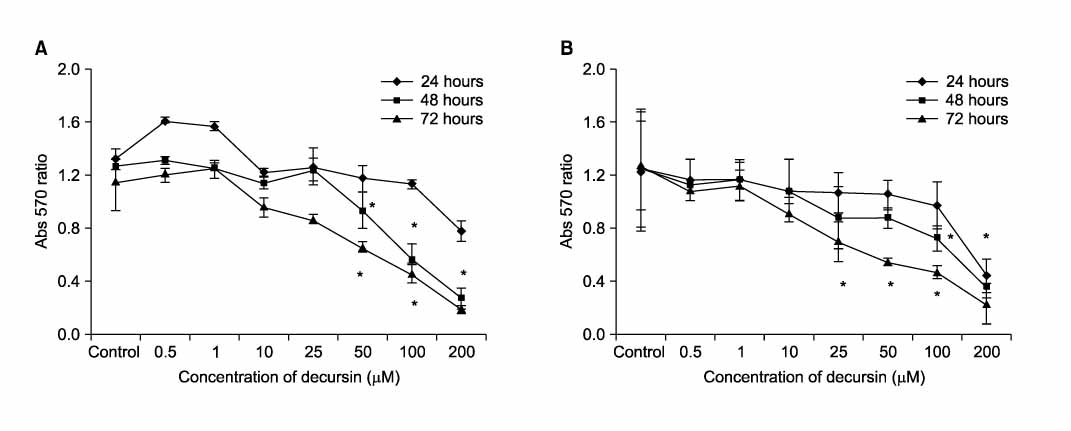
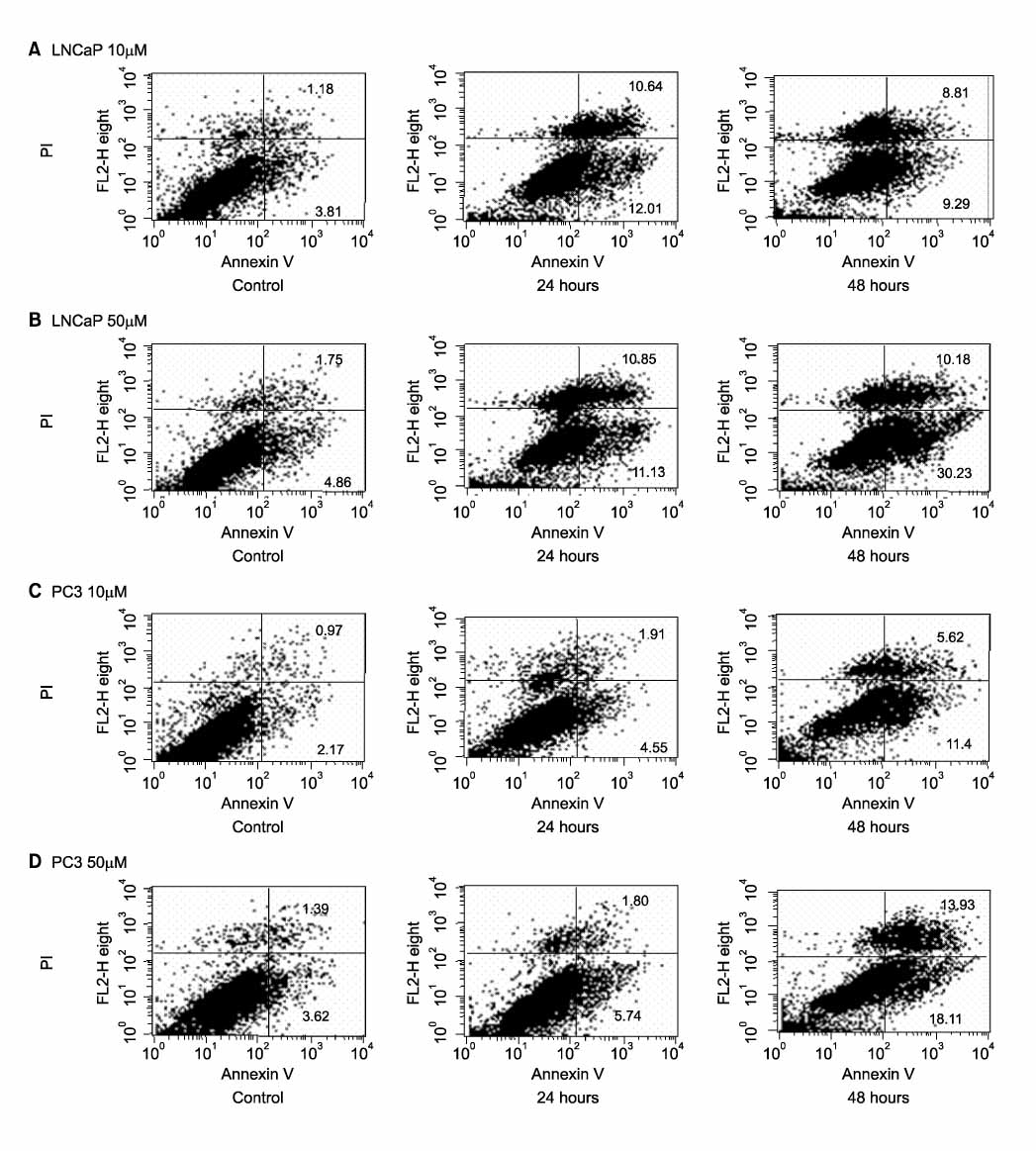
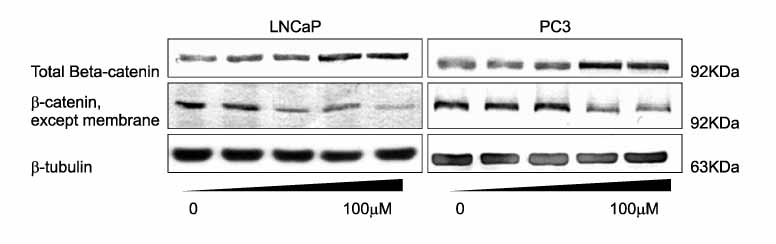
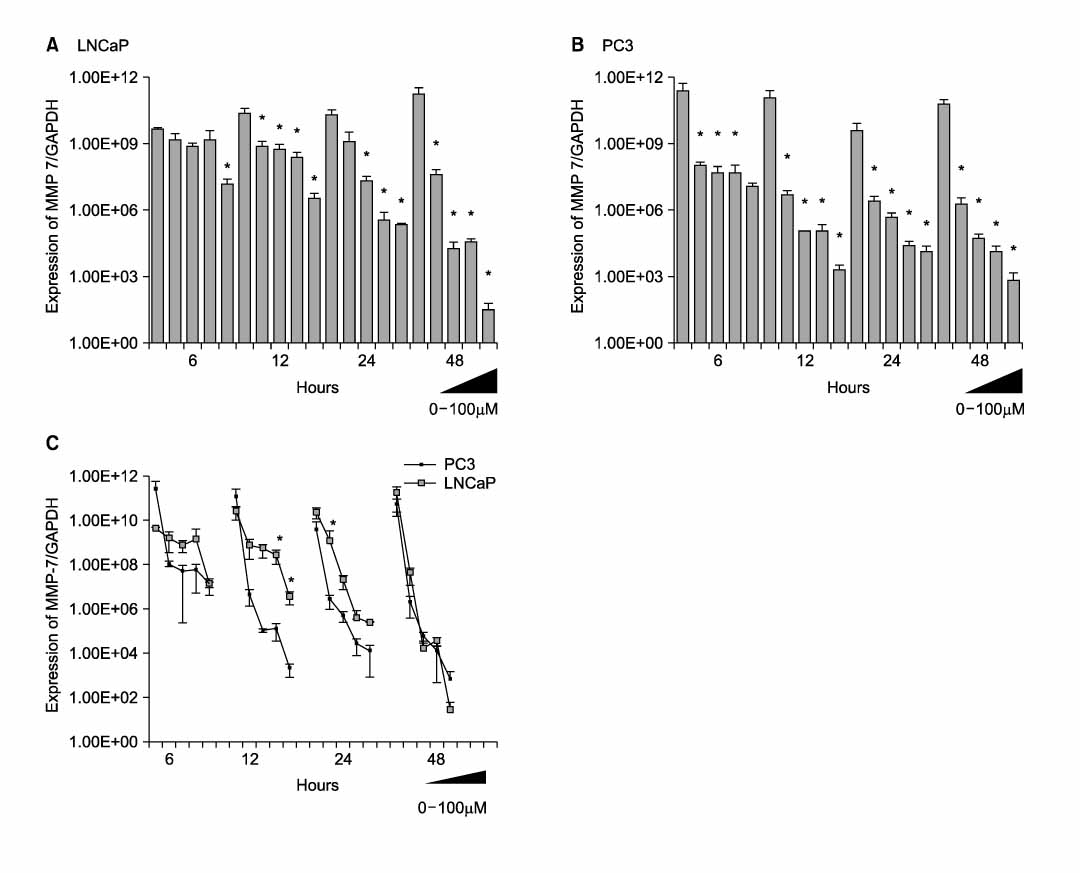
Death and apoptosis were increased after treatment with decursin 0.5-100 micrometer in PC-3 and LNCaP cells. This was revealed dose and time-dependent increase of cancer cell death on 24, 48 and 72 hours. FACScan showed an increment of apoptosis on 24, 48 hours. Expression of intracellular beta-catenin protein was decreased dose-dependently in both of prostate cancer cell lines. Decursin reduced MMP-7 mRNA expression on 6, 12, 24, 48 hours dose-dependently.
CONCLUSIONS
Decursin affects the viability of prostate cancer cells. Increased cancer cell death was associated with increased apoptosis. This study suggests that decursin may play a role in the treatment of prostate cancer.
Keyword
MeSH Terms
Figure
Reference
-
1. Harris KA, Reese DM. Treatment options in hormone-refractory prostate cancer: current and future approaches. Drugs. 2001. 61:2177–2192.2. Logothetis CJ. Docetaxel in the intergrated management of prostate cancer. Current applications and future promise. Oncology. 2002. 16:6 Suppl 6. 63–72.3. Han KS, Cho KS, Lee SH, Hong SJ. Estramustine phosphate based chemotherapy for hormone refractory prostate cancer. Korean J Urol. 2007. 48:684–690.4. Clevers H. Wnt/beta-catenin signaling in development and disease. Cell. 2006. 127:469–480.5. Umbas R, Schalken JA, Aalders TW, Carter BS, Karthaus HF, Schaafsma HE, et al. Expression of the cellular adhesion molecule E-cadherin is reduced or absent in high-grade prostate cancer. Cancer Res. 1992. 52:5104–5109.6. Richmond PJ, Karayiannakis AJ, Nagafuchi A, Kaisary AV, Pignatelli M. Aberrant E-cadherin and alpha-catenin expression in prostate cancer: correlation with patient survival. Cancer Res. 1997. 57:3189–3193.7. Luo J, Lubaroff DM, Hendrix MJ. Suppression of prostate cancer invasive potential and matrix metalloproteinase activity by E-cadherin transfection. Cancer Res. 1999. 59:3552–3556.8. Nelson WJ, Nusse R. Convergence of Wnt, beta-catenin, and cadherin pathways. Science. 2004. 303:1483–1487.9. Verras M, Sun Z. Roles and regulation of Wnt signaling and beta-catenin in prostate cancer. Cancer Lett. 2006. 237:22–32.10. Kim TH, Kim YS, Myoung SC, Lee JH, Won EH. Prostagladin E receptor II and IV increase the expression of martrix metalloproteinase-7 in PC (prostate cancer)-3 cells. Korean J Urol. 2004. 45:478–484.11. Ahn KS, Sim WS, Kim IH. Decursin: a cytotoxic agent and protein kinase C activator from the root of Angelica gigas. Planta Med. 1996. 62:7–9.12. Lee S, Lee YS, Jung SH, Shin KH, Kim BK, Kang SS. Anti-tumor activities of decursinol angelate and decursin from Angelica gigas. Arch Pharm Res. 2003. 26:727–730.13. Yim D, Singh RP, Agarwal C, Lee S, Chi H, Agarwal R. A novel anticancer agent, decursin, induces G1 arrest and apoptosis in human prostate carcinoma cells. Cancer Res. 2005. 65:1035–1044.14. Song GY, Lee JH, Cho M, Park BS, Kim DE, Oh S. Decursin suppresses human androgen-independent PC3 prostate cancer cell proliferation by promoting the degradation of beta-catenin. Mol Pharmacol. 2007. 72:1599–1606.15. Jiang C, Lee HJ, Li GX, Guo J, Malewicz B, Zhao Y, et al. Potent antiandrogen and androgen receptor activities of an Angelica gigas-containing herbal formulation: identification of decursin as a novel and active compound with implications for prevention and treatment of prostate cancer. Cancer Res. 2006. 66:453–463.16. Mosmann T. Rapid colorimetric assay for cellular growth and survival: application to proliferation and cytotoxicity assays. J Immunol Methods. 1983. 65:55–63.17. Dignam JD, Lebovitz RM, Roeder RG. Accurate transcription initiation by RNA polymerase II in a soluble extract from isolated mammalian nuclei. Nucleic Acids Res. 1983. 11:1475–1489.18. Heinlein CA, Chang C. Androgen receptor in prostate cancer. Endocr Rev. 2004. 25:276–308.19. Feldman BJ, Feldman D. The development of androgen-independent prostate cancer. Nat Rev Cancer. 2001. 1:34–45.20. Hyytinen ER, Thalmann GN, Zhau HE, Karhu R, Kallioniemi OP, Chung LW, et al. Genetic changes associated with the acquisition of androgen-independent growth, tumorigenicity and metastatic potential in a prostate cancer model. Br J Cancer. 1997. 75:190–195.21. Chen G, Shukeir N, Potti A, Sircar K, Aprikian A, Goltzman D, et al. Up-regulation of Wnt-1 and beta-catenin production in patients with advanced metastatic prostate carcinoma: potential pathogenetic and prognostic implications. Cancer. 2004. 101:1345–1356.22. Saha B, Arase A, Imam SS, Tsao-Wei D, Naritoku WY, Groshen S, et al. Overexpression of E-cadherin and beta-catenin proteins in metastatic prostate cancer cells in bone. Prostate. 2008. 68:78–84.23. Whitaker HC, Girling J, Warren AY, Leung H, Mills IG, Neal DE. Alterations in beta-catenin expression and localization in prostate cancer. Prostate. 2008. 68:1196–1205.24. Bismar TA, Humphrey PA, Grignon DJ, Wang HL. Expression of beta-catenin in prostatic adenocarcinomas: a comparison with colorectal adenocarcinomas. Am J Clin Pathol. 2004. 121:557–563.25. Jaggi M, Johansson SL, Baker JJ, Smith LM, Galich A, Balaji KC. Aberrant expression of E-cadherin and beta-catenin in human prostate cancer. Urol Oncol. 2005. 23:402–406.26. Van Veldhuizen PJ, Ray G, Banerjee S, Dhar G, Kambhampati S, Dhar A, et al. 2-methoxyestradiol modulates beta-catenin in prostate cancer cells: a possible mediator of 2-methoxyestradiol-induced inhibition of cell growth. Int J Cancer. 2008. 122:567–571.27. Kader AK, Liu J, Shao L, Dinney CP, Lin J, Wang Y, et al. Matrix metalloproteinase polymorphisms are associated with bladder cancer invasiveness. Clin Cancer Res. 2007. 13:2614–2620.28. Leeman MF, Curran S, Murray GI. New insights into the roles of matrix metalloproteinases in colorectal cancer development and progression. J Pathol. 2003. 201:528–534.29. Brabletz T, Jung A, Dag S, Hlubek F, Kirchner T. Beta-catenin regulates the expression of the matrix metalloproteinase-7 in human colorectal cancer. Am J Pathol. 1999. 155:1033–1038.30. Zeng ZS, Shu WP, Cohen AM, Guillem JG. Matrix metalloproteinase-7 expression in colorectal cancer liver metastases: evidence for involvement of MMP-7 activation in human cancer metastases. Clin Cancer Res. 2002. 8:144–148.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Clinical Significance of Wnt/beta-Catenin Signalling and Androgen Receptor Expression in Prostate Cancer
- Suppression of β-catenin Signaling Pathway in Human Prostate Cancer PC3 Cells by Delphinidin
- Expression of beta-catenin and Adenomatous Polyposis Coli(APC) Protein in Squamous Cell Carcinoma of the Laryngeal Cancers
- β-Catenin expression is associated with cell invasiveness in pancreatic cancer
- Prostagladin E Receptor II and IV Increase the Expression of Martrix Metalloproteinase-7 in PC (Prostate Cancer)-3 Cells