J Vet Sci.
2015 Dec;16(4):397-404. 10.4142/jvs.2015.16.4.397.
Effect of serum-derived albumin scaffold and canine adipose tissue-derived mesenchymal stem cells on osteogenesis in canine segmental bone defect model
- Affiliations
-
- 1Department of Veterinary Surgery, College of Veterinary Medicine, Seoul National University, Seoul 08826, Korea. ohkweon@snu.ac.kr
- 2Department of Veterinary Surgery, College of Veterinary Medicine, Kangwon National University, Chuncheon 24341, Korea.
- 3BK21 PLUS Program for Creative Veterinary Science Research, Research Institute for Veterinary Science and College of Veterinary Medicine, Seoul National University, Seoul 08826, Korea.
- KMID: 2133621
- DOI: http://doi.org/10.4142/jvs.2015.16.4.397
Abstract
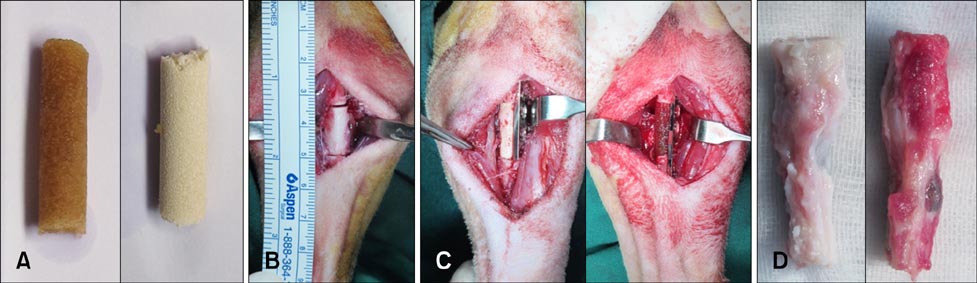
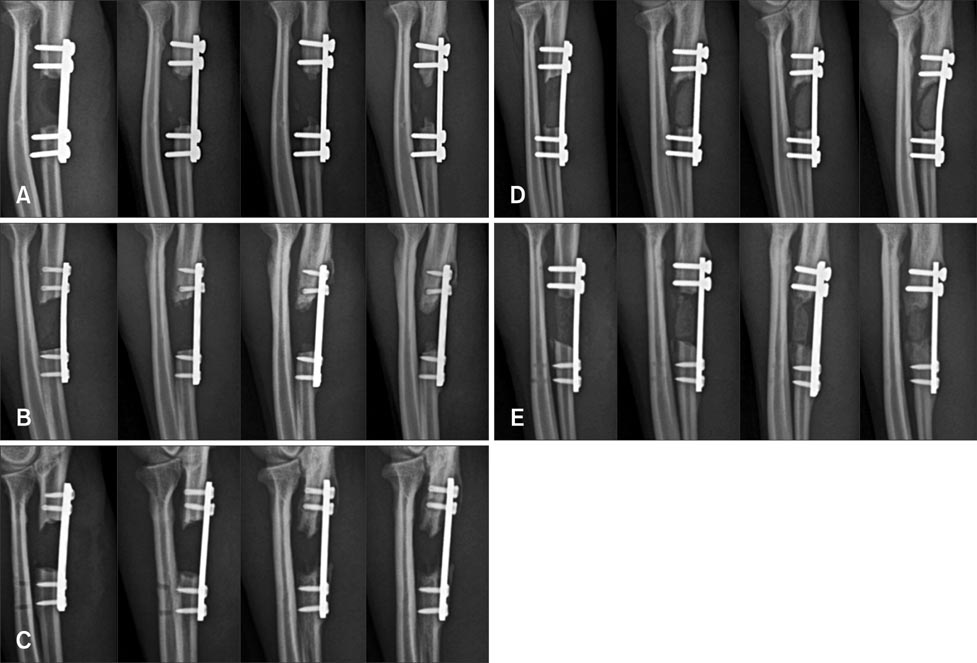
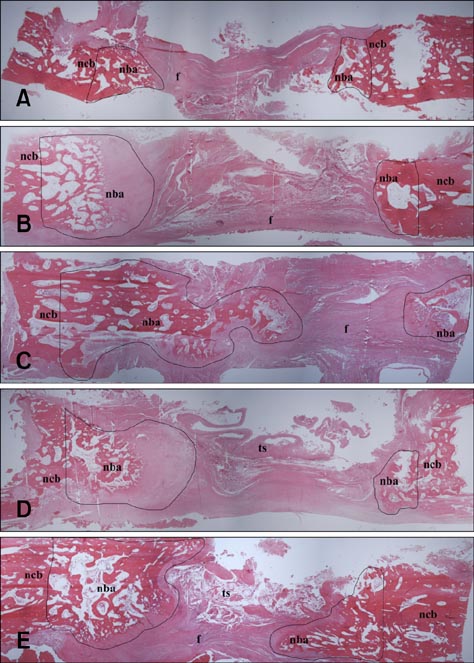
- Composite biological and synthetic grafts with progenitor cells offer an alternative approach to auto- or allografts for fracture repair. This study was conducted to evaluate osteogenesis of autologous serum-derived albumin (ASA) scaffolds seeded with canine adipose tissue-derived mesenchymal stem cells (Ad-MSCs) in a canine segmental bone defect model. ASA scaffold was prepared with canine serum using cross-linking and freeze-drying procedures. Beta-tricalcium phosphate (beta-TCP) was mixed at the cross-linking stage. Ad-MSCs were seeded into the scaffold and incubated for one day before implantation. After 16 weeks, the grafts were harvested for histological analysis. The dogs were divided into five groups: control, ASA scaffolds with and without Ad-MSCs, and ASA scaffolds including beta-TCP with and without Ad-MSCs. ASA scaffolds with Ad-MSCs had a significantly larger area of increased opacity at the proximal and distal host cortex-implant interfaces in radiographs 16 weeks after implantation compared to the groups with beta-TCP (p < 0.05). Histomorphometric analysis showed that ASA scaffolds with Ad-MSCs had significantly greater new bone formation than other groups (p < 0.05). These results suggest that Ad-MSCs seeded into ASA scaffolds enhanced osteogenesis in the bone defect model, but that beta-TCP in the ASA scaffold might prevent penetration of the cells required for bone healing.
Figure
Reference
-
1. Aho AJ, Ekfors T, Dean PB, Aro HT, Ahonen A, Nikkanen V. Incorporation and clinical results of large allografts of the extremities and pelvis. Clin Orthop Relat Res. 1994; 307:200–213.2. Arinzeh TL, Tran T, Mcalary J, Daculsi G. A comparative study of biphasic calcium phosphate ceramics for human mesenchymal stem-cell-induced bone formation. Biomaterials. 2005; 26:3631–3638.
Article3. Bi LX, Simmons DJ, Mainous E. Expression of BMP-2 by rat bone marrow stromal cells in culture. Calcif Tissue Int. 1999; 64:63–68.
Article4. Bucholz RW. Nonallograft osteoconductive bone graft substitutes. Clin Orthop Relat Res. 2002; 395:44–52.
Article5. Byeon YE, Ryu HH, Park SS, Koyama Y, Kikuchi M, Kim WH, Kang KS, Kweon OK. Paracrine effect of canine allogenic umbilical cord blood-derived mesenchymal stromal cells mixed with β-tricalcium phosphate on bone regeneration in ectopic implantations. Cytotherapy. 2010; 12:626–636.
Article6. Clokie CML, Moghadam H, Jackson MT, Sandor GKB. Closure of critical sized defects with allogenic and alloplastic bone substitutes. J Craniofac Surg. 2002; 13:111–121.
Article7. Drosse I, Volkmer E, Capanna R, De Biase P, Mutschler W, Schieker M. Tissue engineering for bone defect healing: an update on a multi-component approach. Injury. 2008; 39:Suppl 2. S9–S20.
Article8. Ehrler DM, Vaccaro AR. The use of allograft bone in lumbar spine surgery. Clin Orthop Relat Res. 2000; 371:38–45.
Article9. Frank O, Heim M, Jakob M, Barbero A, Schäfer D, Bendik I, Dick W, Heberer M, Martin I. Real-time quantitative RT-PCR analysis of human bone marrow stromal cells during osteogenic differentiation in vitro. J Cell Biochem. 2002; 85:737–746.
Article10. Gallego L, Junquera L, García E, García V, Álvarez-Viejo M, Costilla S, Fresno MF, Meana Á. Repair of rat mandibular bone defects by alveolar osteoblasts in a novel plasma-derived albumin scaffold. Tissue Eng Part A. 2010; 16:1179–1187.
Article11. Gallego L, Junquera L, Meana Á, Álvarez-Viejo M, Fresno M. Ectopic bone formation from mandibular osteoblasts cultured in a novel human serum-derived albumin scaffold. J Biomater Appl. 2010; 25:367–381.
Article12. Garbuz DS, Masri BA, Czitrom AA. Biology of allografting. Orthop Clin North Am. 1998; 29:199–204.
Article13. Harris SE, Sabatini M, Harris MA, Feng JQ, Wozney J, Mundy GR. Expression of bone morphogenetic protein messenger RNA in prolonged cultures of fetal rat calvarial cells. J Bone Miner Res. 1994; 9:389–394.
Article14. Head WC, Emerson RH Jr, Malinin TI. Structural bone grafting for femoral reconstruction. Clin Orthop Relat Res. 1999; 369:223–229.
Article15. Hutmacher DW. Scaffolds in tissue engineering bone and cartilage. Biomaterials. 2000; 21:2529–2543.
Article16. Ishaug SL, Crane GM, Miller MJ, Yasko AW, Yaszemski MJ, Mikos AG. Bone formation by three-dimensional stromal osteoblast culture in biodegradable polymer scaffolds. J Biomed Mater Res. 1997; 36:17–28.
Article17. Jang BJ, Byeon YE, Lim JH, Ryu HH, Kim WH, Koyama Y, Kikuchi M, Kang KS, Kweon OK. Implantation of canine umbilical cord blood-derived mesenchymal stem cells mixed with beta-tricalcium phosphate enhances osteogenesis in bone defect model dogs. J Vet Sci. 2008; 9:387–393.
Article18. Kang BJ, Kim Y, Lee SH, Kim WH, Woo HM, Kweon OK. Collagen I gel promotes homogenous osteogenic differentiation of adipose tissue-derived mesenchymal stem cells in serum-derived albumin scaffold. J Biomater Sci Polym Ed. 2013; 24:1233–1243.
Article19. Kang BJ, Ryu HH, Park SS, Kim Y, Woo HM, Kim WH, Kweon OK. Effect of matrigel on the osteogenic potential of canine adipose tissue-derived mesenchymal stem cells. J Vet Med Sci. 2012; 74:827–836.
Article20. Kang BJ, Ryu HH, Park SS, Koyama Y, Kikuchi M, Woo HM, Kim WH, Kweon OK. Comparing the osteogenic potential of canine mesenchymal stem cells derived from adipose tissues, bone marrow, umbilical cord blood, and Wharton's jelly for treating bone defects. J Vet Sci. 2012; 13:299–310.
Article21. Kikuchi M, Koyama Y, Yamada T, Imamura Y, Okada T, Shirahama N, Akita K, Takakuda K, Tanaka J. Development of guided bone regeneration membrane composed of β-tricalcium phosphate and poly (L-lactide-co-glycolide-co-ε-caprolactone) composites. Biomaterials. 2004; 25:5979–5986.
Article22. Nilsson OS, Urist MR, Dawson EG, Schmalzried TP, Finerman GAM. Bone repair induced by bone morphogenetic protein in ulnar defects in dogs. J Bone Joint Surg Br. 1986; 68:635–642.
Article23. Oonishi H. Orthopaedic applications of hydroxyapatite. Biomaterials. 1991; 12:171–178.
Article24. Pittenger MF, Mackay AM, Beck SC, Jaiswal RK, Douglas R, Mosca JD, Moorman MA, Simonetti DW, Craig S, Marshak DR. Multilineage potential of adult human mesenchymal stem cells. Science. 1999; 284:143–147.
Article25. Ryu HH, Lim JH, Byeon YE, Park JR, Seo MS, Lee YW, Kim WH, Kang KS, Kweon OK. Functional recovery and neural differentiation after transplantation of allogenic adipose-derived stem cells in a canine model of acute spinal cord injury. J Vet Sci. 2009; 10:273–284.
Article26. Salgado AJ, Coutinho OP, Reis RL. Bone tissue engineering: state of the art and future trends. Macromol Biosci. 2004; 4:743–765.
Article27. Sanchez-Sotelo J, Munuera L, Madero R. Treatment of fractures of the distal radius with a remodellable bone cement: a prospective, randomised study using Norian SRS. J Bone Joint Surg Br. 2000; 82:856–863.28. Sandhu HS, Grewal HS, Parvataneni H. Bone grafting for spinal fusion. Orthop Clin North Am. 1999; 30:685–698.
Article29. Sartoris DJ, Holmes RE, Resnick D. Coralline hydroxyapatite bone graft substitutes: radiographic evaluation. J Foot Surg. 1992; 31:301–313.30. Schäffler A, Büchler C. Concise review: adipose tissue-derived stromal cells-basic and clinical implications for novel cell-based therapies. Stem cells. 2007; 25:818–827.
Article31. Tseng SS, Lee MA, Reddi AH. Nonunions and the potential of stem cells in fracture-healing. J Bone Joint Surg Am. 2008; 90:Suppl 1. 92–98.
Article32. Ueki K, Takazakura D, Marukawa K, Shimada M, Nakagawa K, Takatsuka S, Yamamoto E. The use of polylactic acid/polyglycolic acid copolymer and gelatin sponge complex containing human recombinant bone morphogenetic protein-2 following condylectomy in rabbits. J Craniomaxillofac Surg. 2003; 31:107–114.
Article33. van Hemert WL, Willems K, Anderson PG, van Heerwaarden RJ, Wymenga AB. Tricalcium phosphate granules or rigid wedge preforms in open wedge high tibial osteotomy: a radiological study with a new evaluation system. Knee. 2004; 11:451–456.
Article34. Zeltinger J, Sherwood JK, Graham DA, Müeller R, Griffith LG. Effect of pore size and void fraction on cellular adhesion, proliferation, and matrix deposition. Tissue Eng. 2001; 7:557–572.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Chondrogenesis of Mesenchymal Stem Cell Derived from Canine Adipose Tissue
- Bone Regeneration from Adipose Stem Cells
- Stem cell properties of cells derived from canine periodontal ligament
- An Implant for Bone Formation using Autologous Mesenchymal Stem Cells
- Osteogenic Differentiation of Human Adipose-derived Stem Cells within PLGA(Poly(D,L-lactic-co-glycolic acid)) Scaffold in the Nude Mouse