Korean J Physiol Pharmacol.
2012 Apr;16(2):145-151. 10.4196/kjpp.2012.16.2.145.
Differential Effects of Tautomycetin and Its Derivatives on Protein Phosphatase Inhibition, Immunosuppressive Function and Antitumor Activity
- Affiliations
-
- 1Research Center for Molecular Medicine, Dalian University of Technology, Dalian 116023, China. rqiu2001@yahoo.com, tangli63b@yahoo.com
- KMID: 2071761
- DOI: http://doi.org/10.4196/kjpp.2012.16.2.145
Abstract
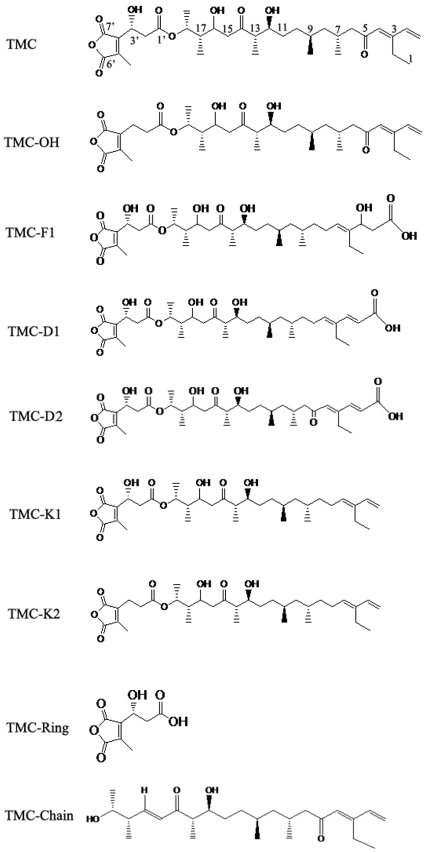
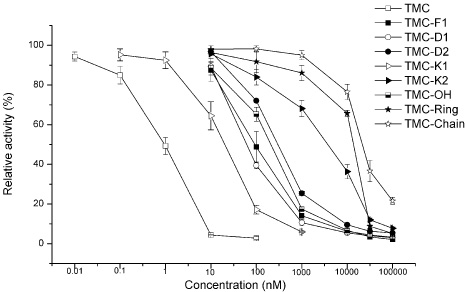
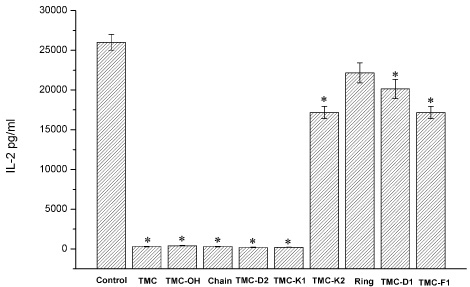
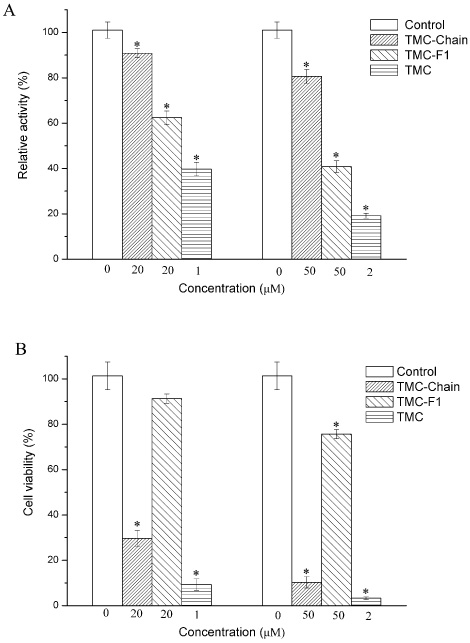
- In the present work, we studied the structure-activity relationship (SAR) of tautomycetin (TMC) and its derivatives. Further, we demonstrated the correlation between the immunosuppressive fuction, anticancer activity and protein phosphatase type 1 (PP1) inhibition of TMC and its derivatives. We have prepared some TMC derivatives via combinatorial biosynthesis, isolation from fermentation broth or chemical degradation of TMC. We found that the immunosuppressive activity was correlated with anticancer activity for TMC and its analog compounds, indicating that TMC may home at the same targets for its immunosuppressive and anticancer activities. Interestingly, TMC-F1, TMC-D1 and TMC-D2 all retained significant, albeit reduced PP1 inhibitory activity compared to TMC. However, only TMC-D2 showed immunosuppressive and anticancer activities in studies carried out in cell lines. Moreover, TMC-Chain did not show any significant inhibitory activity towards PP1 but showed strong growth inhibitory effect. This observation implicates that the maleic anhydride moiety of TMC is critical for its phosphatase inhibitory activity whereas the C1-C18 moiety of TMC is essential for the inhibition of tumor cell proliferation. Furthermore, we measured in vivo phosphatase activities of PP1 in MCF-7 cell extracts treated with TMC and its related compounds, and the results indicate that the cytotoxicity of TMC doesn't correlate with its in vivo PP1 inhibition activity. Taken together, our study suggests that the immunosuppressive and anticancer activities of TMC are not due to the inhibition of PP1. Our results provide a novel insight for the elucidation of the underlying molecular mechanisms of TMC's important biological functions.
MeSH Terms
Figure
Cited by 1 articles
-
Multiple Effects of a Novel Epothilone Analog on Cellular Processes and Signaling Pathways Regulated by Rac1 GTPase in the Human Breast Cancer Cells
Hong Zhang, Fan An, Li Tang, Rongguo Qiu
Korean J Physiol Pharmacol. 2014;18(2):109-120. doi: 10.4196/kjpp.2014.18.2.109.
Reference
-
1. Cheng XC, Kihara T, Ying X, Uramoto M, Osada H, Kusakabe H, Wang BN, Kobayashi Y, Ko K, Yamaguchi I, Shen YC, Isono K. A new antibiotic, tautomycetin. J Antibiot (Tokyo). 1989. 42:141–144.2. Mitsuhashi S, Matsuura N, Ubukata M, Oikawa H, Shima H, Kikuchi K. Tautomycetin is a novel and specific inhibitor of serine/threonine protein phosphatase type 1, PP1. Biochem Biophys Res Commun. 2001. 287:328–331.3. Ishihara H, Martin BL, Brautigan DL, Karaki H, Ozaki H, Kato Y, Fusetani N, Watabe S, Hashimoto K, Uemura D, Hartshorne DJ. Calyculin A and okadaic acid: inhibitors of protein phosphatase activity. Biochem Biophys Res Commun. 1989. 159:871–877.4. Li YM, Casida JE. Cantharidin-binding protein: identification as protein phosphatase 2A. Proc Natl Acad Sci USA. 1992. 89:11867–11870.5. Roberge M, Tudan C, Hung SM, Harder KW, Jirik FR, Anderson H. Antitumor drug fostriecin inhibits the mitotic entry checkpoint and protein phosphatases 1 and 2A. Cancer Res. 1994. 54:6115–6121.6. Nishiwaki-Matsushima R, Nishiwaki S, Ohta T, Yoshizawa S, Suganuma M, Harada K, Watanabe MF, Fujiki H. Structure-function relationships of microcystins, liver tumor promoters, in interaction with protein phosphatase. Jpn J Cancer Res. 1991. 82:993–996.7. Pearson L, Mihali T, Moffitt M, Kellmann R, Neilan B. On the chemistry, toxicology and genetics of the cyanobacterial toxins, microcystin, nodularin, saxitoxin and cylindrospermopsin. Mar Drugs. 2010. 8:1650–1680.8. Bollen M. Combinatorial control of protein phosphatase-1. Trends Biochem Sci. 2001. 26:426–431.9. Cohen PT. Protein phosphatase 1--targeted in many directions. J Cell Sci. 2002. 115:241–256.10. Heneberg P. Use of protein tyrosine phosphatase inhibitors as promising targeted therapeutic drugs. Curr Med Chem. 2009. 16:706–733.11. Lee JH, Lee JS, Kim SE, Moon BS, Kim YC, Lee SK, Lee SK, Choi KY. Tautomycetin inhibits growth of colorectal cancer cells through p21cip/WAF1 induction via the extracellular signal-regulated kinase pathway. Mol Cancer Ther. 2006. 5:3222–3231.12. Shim JH, Lee HK, Chang EJ, Chae WJ, Han JH, Han DJ, Morio T, Yang JJ, Bothwell A, Lee SK. Immunosuppressive effects of tautomycetin in vivo and in vitro via T cell-specific apoptosis induction. Proc Natl Acad Sci USA. 2002. 99:10617–10622.13. Lee SH, Hahn SJ, Min G, Kim J, Jo SH, Choe H, Choi BH. Inhibitory actions of HERG currents by the immunosuppressant drug cyclosporin A. Korean J Physiol Pharmacol. 2011. 15:291–297.14. Chung J, Kuo CJ, Crabtree GR, Blenis J. Rapamycin-FKBP specifically blocks growth-dependent activation of and signaling by the 70 kd S6 protein kinases. Cell. 1992. 69:1227–1236.15. Gust B, Challis GL, Fowler K, Kieser T, Chater KF. PCR-targeted Streptomyces gene replacement identifies a protein domain needed for biosynthesis of the sesquiterpene soil odor geosmin. Proc Natl Acad Sci USA. 2003. 100:1541–1546.16. Van Gompel JJ, Kunnimalaiyaan M, Holen K, Chen H. ZM336372, a Raf-1 activator, suppresses growth and neuroendocrine hormone levels in carcinoid tumor cells. Mol Cancer Ther. 2005. 4:910–917.17. Yan Y, Mumby MC. Distinct roles for PP1 and PP2A in phosphorylation of the retinoblastoma protein. PP2a regulates the activities of G(1) cyclin-dependent kinases. J Biol Chem. 1999. 274:31917–31924.18. Kawamura T, Matsuzawa S, Mizuno Y, Kikuchi K, Oikawa H, Oikawa M, Ubukata M, Ichihara A. Different moieties of tautomycin involved in protein phosphatase inhibition and induction of apoptosis. Biochem Pharmacol. 1998. 55:995–1003.19. Matsuzawa S, Kawamura T, Mitsuhashi S, Suzuki T, Matsuo Y, Suzuki M, Mizuno Y, Kikuchi K. Thyrsiferyl 23-acetate and its derivatives induce apoptosis in various T- and B-leukemia cells. Bioorg Med Chem. 1999. 7:381–387.20. Liu S, Yu Z, Yu X, Huang SX, Luo Y, Wu L, Shen W, Yang Z, Wang L, Gunawan AM, Chan RJ, Shen B, Zhang ZY. SHP2 is a target of the immunosuppressant tautomycetin. Chem Biol. 2011. 18:101–110.21. Egloff MP, Johnson DF, Moorhead G, Cohen PT, Cohen P, Barford D. Structural basis for the recognition of regulatory subunits by the catalytic subunit of protein phosphatase 1. EMBO J. 1997. 16:1876–1887.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Immunosuppressive Effects of Tautomycetin on T Cells
- Effects of Tautomycetin on the Proliferation and Fibronectin Secretion in Vascular Smooth Muscle Cells and Glomerular Mesangial Cells
- Regulation of chicken protein tyrosine phosphatase 1 and human protein tyrosine phosphatase 1B activity by casein kinase II- and p56lck-mediated phosphorylation
- Effects of enamel matrix derivatives on biologic activities of human periodontal fibloblasts to demineralized root surface
- Effect of Inorganic Polyphosphate on Cultured Periodontal Ligament Cells