Korean J Physiol Pharmacol.
2008 Feb;12(1):1-6. 10.4196/kjpp.2008.12.1.1.
Effects of TGF-beta1 Ribbon Antisense on CCl4-induced Liver Fibrosis
- Affiliations
-
- 1Department of Physiology, College of Medicine, Dongguk University, Gyeongju 780-714, Korea. kodoh@dongguk.ac.kr
- KMID: 2071639
- DOI: http://doi.org/10.4196/kjpp.2008.12.1.1
Abstract
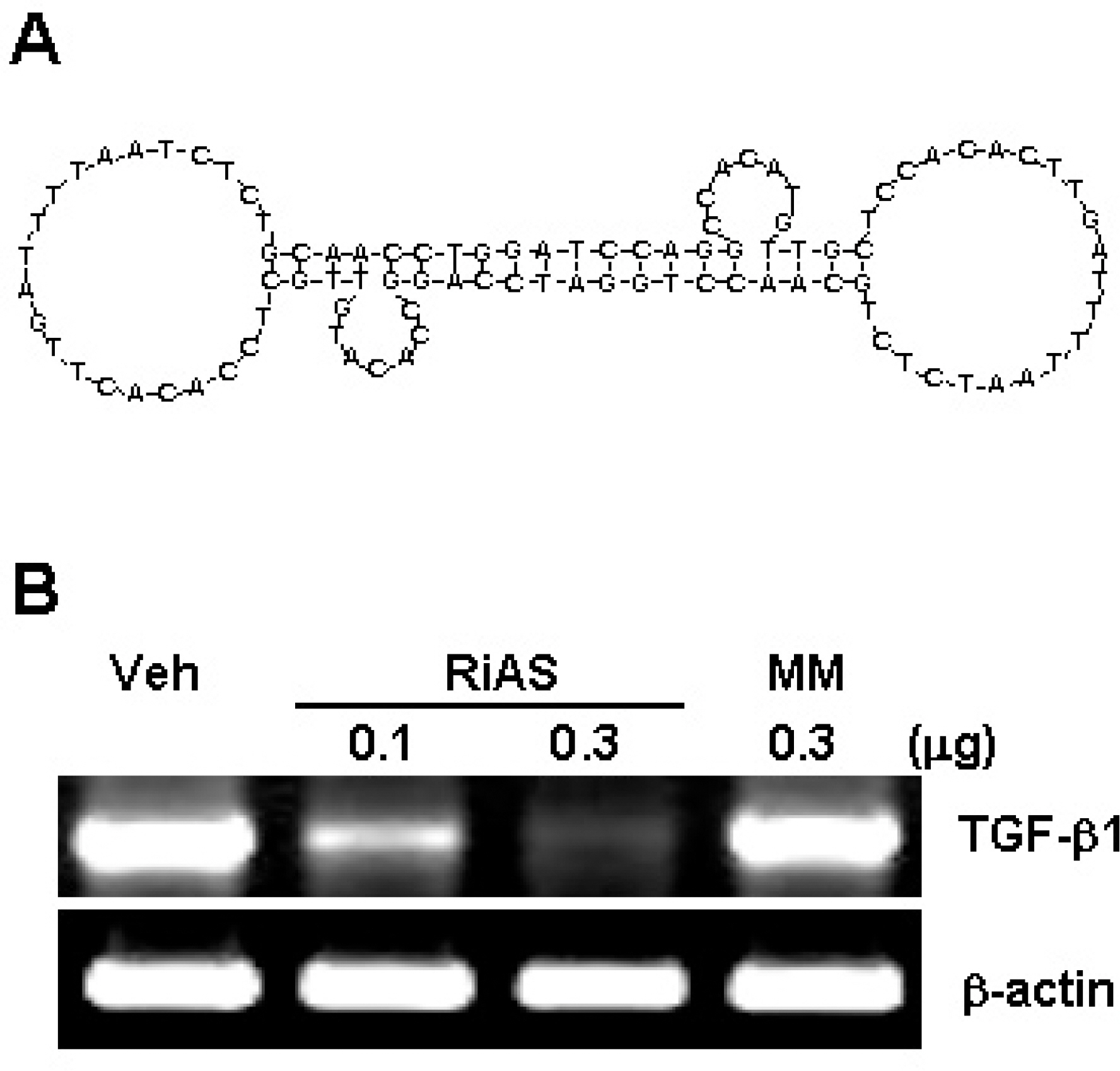
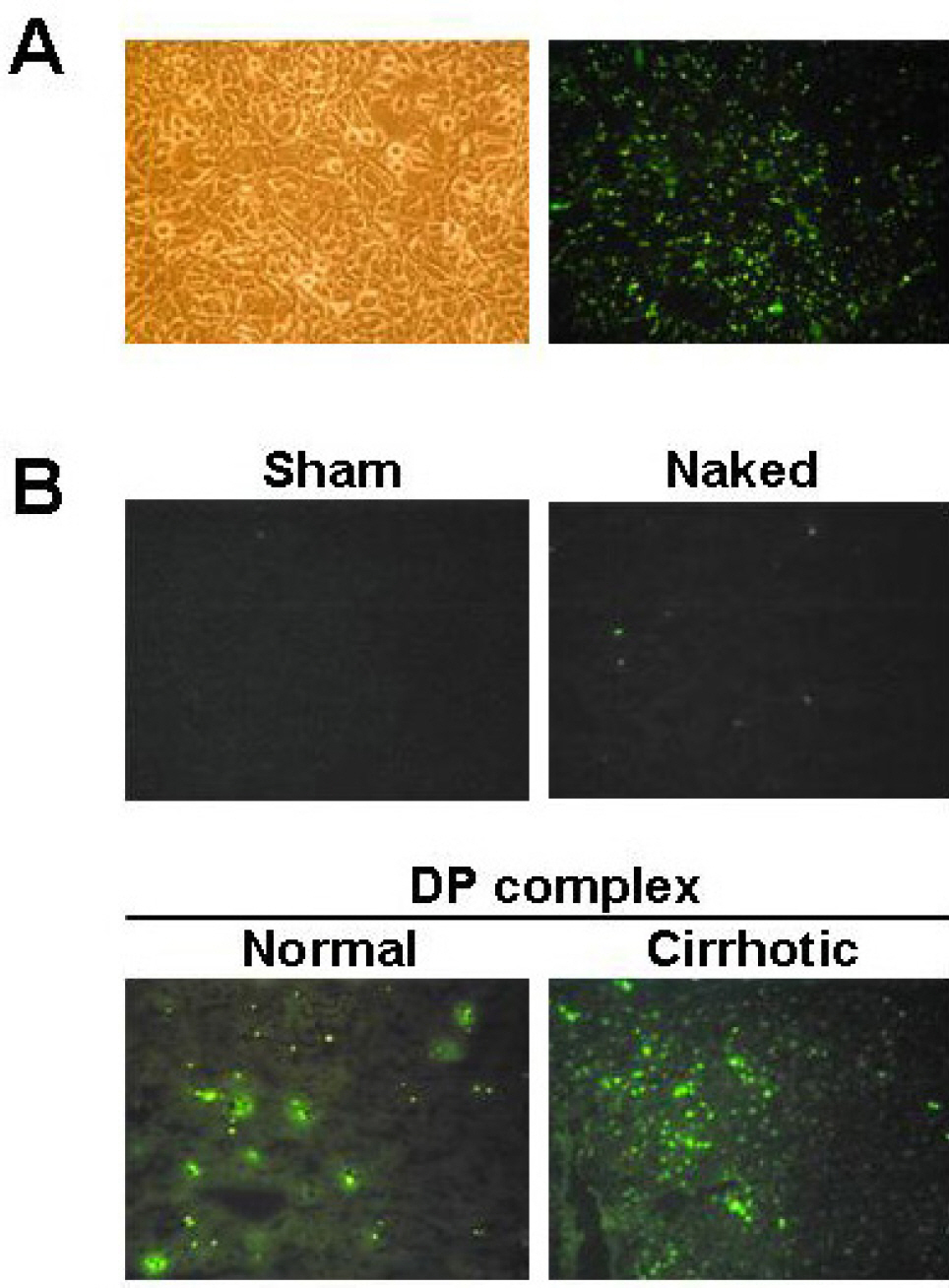
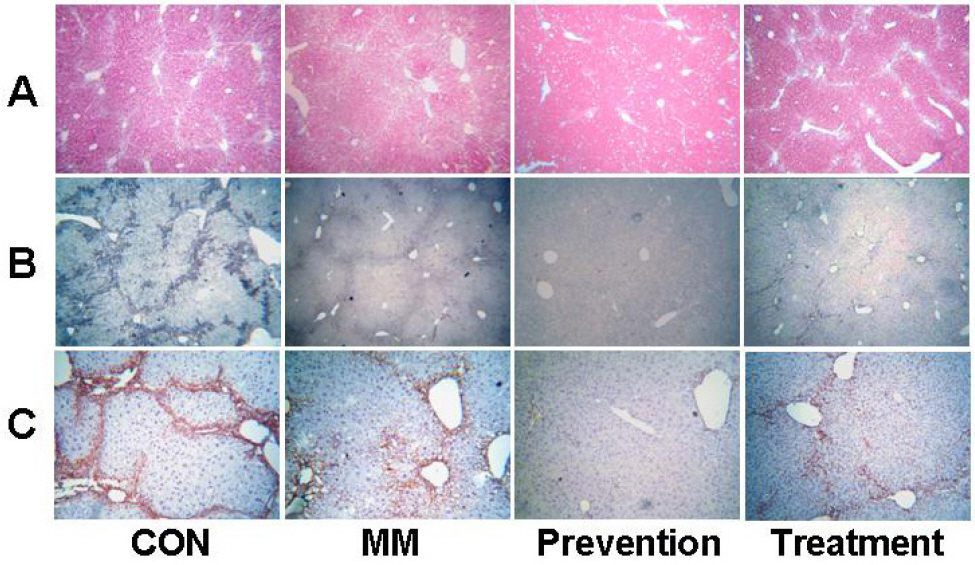
- Ribbon-type antisense oligonucleotide to TGF-beta1(TGF-beta1 RiAS) was designed and tested to prevent or resolve the fibrotic changes induced by CCl4 injection. When Hepa1c1c7 cells were transfected with TGF-beta1 RiAS, the level of TGF-beta1 mRNA was effectively reduced. TGF-beta1 RiAS, mismatched RiAS, and normal saline were each injected to mice via tail veins. When examined for the biochemical effects on the liver, TGF-beta1 mRNA levels were significantly reduced only in the TGF-beta1 RiAS-treated group. The results of immunohistochemical studies showed that TGF-beta1 RiAS prevented the accumulation of collagen and alpha-smooth muscle actin, but could not resolve established fibrosis. These results indicate that ribbon antisense to TGF-beta1 with efficient uptake can effectively prevent fibrosis of the liver.
MeSH Terms
Figure
Reference
-
Alcolado R., Arthur MJ., Iredale JP. Pathogenesis of liver fibrosis. Clin Sci (Lond). 92:103–112. 1997.
ArticleArias M., Lahme B., Van de Leur E., Gressner AM., Weiskirchen R. Adenoviral delivery of an antisense RNA complementary to the 3’ coding sequence of transforming growth factor-beta1 inhibits fibrogenic activities of hepatic stellate cells. Cell Growth Differ. 13:265–273. 2002.Arias M., Sauer-Lehnen S., Treptau J., Janoschek N., Theuerkauf I., Buettner R., Gressner AM., Weiskirchen R. Adenoviral expression of a transforming growth factor-beta1 antisense mRNA is effective in preventing liver fibrosis in bile-duct ligated rats. BMC Gastroenterol. 3:29. 2003.
ArticleArthur MJ., Fibrogenesis II. Metalloproteinases and their inhibitors in liver fibrosis. Am J Physiol Gastrointest Liver Physiol. 279:G245–249. 2000.Bajpai AK., Park JH., Moon IJ., Kang H., Lee YH., Doh KO., Suh SI., Chang BC., Park JG. Rapid blockade of telomerase activity and tumor cell growth by the DPL lipofection of ribbon antisense to hTR. Oncogene. 24:6492–6501. 2005.
ArticleBharath MM., Chandra NR., Rao MRS. Prediction of an HMG-box fold in the C-terminal domain of histone H1: insights into its role in DNA condensation. Proteins. 49:71–81. 2002.
ArticleBissell DM., Roulot D., George J. Transforming growth factor beta and the liver. Hepatology. 34:859–867. 2001.Choi YK., Moon IJ., Jung HK., Jang BC., Seo SI., Park JG. Prevention of tissue injury by ribbon antisense to TGF-beta1 in the kidney. Int J Mol Med. 15:391–399. 2005.Derossi D., Calvet S., Trembleau A., Brunissen A., Chassaing G., Prochiantz A. Cell internalization of the third helix of the Antennapedia homeodomain is receptor-independent. J Biol Chem. 271:18188–18193. 1996.
ArticleDokka S., Toledo D., Shi X., Ye J., Rojanasakul Y. High-efficiency gene transfection of macrophages by lipoplexes. Int J Pharm. 206:97–104. 2000.
ArticleEguchi A., Akuta T., Okuyama H., Senda T., Yokoi H., Inokuchi H., Fujita S., Hayakawa T., Takeda K., Hasegawa M., Nakanishi M. Protein transduction domain of HIV-1 Tat protein promotes efficient delivery of DNA into mammalian cells. J Biol Chem. 276:26204–26210. 2001.
ArticleForbes SJ., Russo FP., Rey V., Burra P., Rugge M., Wright NA., Alison MR. A significant proportion of myofibroblasts are of bone marrow origin in human liver fibrosis. Gastroenterology. 126:955–963. 2004.
ArticleFriedman SL. Molecular regulation of hepatic fibrosis, an integrated cellular response to tissue injury. J Biol Chem. 275:2247–2250. 2000.
ArticleFriedman SL., Roll FJ., Boyles J., Bissell DM. Hepatic lipocytes: the principal collagen-producing cells of normal rat liver. Proc Natl Acad Sci U S A. 82:8681–8685. 1985.
ArticleFulda S., Wick W., Weller M., Debatin KM. Smac agonists sensitize for Apo2L/TRAIL- or anticancer drug-induced apoptosis and induce regression of malignant glioma in vivo. Nat Med. 8:808–815. 2002.
ArticleFutaki S., Suzuki T., Ohashi W., Yagami T., Tanaka S., Ueda K., Sugiura Y. Arginine-rich peptides. An abundant source of membrane-permeable peptides having potential as carriers for intracellular protein delivery. J Biol Chem. 276:5836–5840. 2001.George J., Roulot D., Koteliansky VE., Bissell DM. In vivo inhibition of rat stellate cell activation by soluble transforming growth factor beta type II receptor: a potential new therapy for hepatic fibrosis. Proc Natl Acad Sci U S A. 96:12719–12724. 1999.Gressner AM., Weiskirchen R., Breitkopf K., Dooley S. Roles of TGF-beta in hepatic fibrosis. Front Biosci. 7:d793–807. 2002.
ArticleGryaznov S., Skorski T., Cucco C., Nieborowska-Skorska M., Chiu CY., Lloyd D., Chen JK., Koziolkiewicz M., Calabretta B. Oligonucleotide N3'→P5’ phosphoramidates as antisense agents. Nucleic Acids Res. 24:1508–1514. 1996.
ArticleHammel P., Couvelard A., O'Toole D., Ratouis A., Sauvanet A., Flejou JF., Degott C., Belghiti J., Bernades P., Valla D., Ruszniewski P., Levy P. Regression of liver fibrosis after biliary drainage in patients with chronic pancreatitis and stenosis of the common bile duct. N Engl J Med. 344:418–423. 2001.
ArticleHenry SP., Novotny W., Leeds J., Auletta C., Kornbrust DJ. Inhibition of coagulation by a phosphorothioate oligonucleotide. Antisense Nucleic Acid Drug Dev. 7:503–510. 1997.
ArticleIredale JP., Benyon RC., Pickering J., McCullen M., Northrop M., Pawley S., Hovell C., Arthur MJ. Mechanisms of spontaneous resolution of rat liver fibrosis. Hepatic stellate cell apoptosis and reduced hepatic expression of metalloproteinase inhibitors. J Clin Invest. 102:538–549. 1998.
ArticleIssa R., Zhou X., Constandinou CM., Fallowfield J., Millward-Sadler H., Gaca MD., Sands E., Suliman I., Trim N., Knorr A., Arthur MJ., Benyon RC., Iredale JP. Spontaneous recovery from micronodular cirrhosis: evidence for incomplete resolution associated with matrix cross-linking. Gastroenterology. 126:1795–1808. 2004.
ArticleKanzler S., Lohse AW., Keil A., Henninger J., Dienes HP., Schirmacher P., Rose-John S., zum Buschenfelde KH., Blessing M. TGF-beta1 in liver fibrosis: an inducible transgenic mouse model to study liver fibrogenesis. Am J Physiol. 276:G1059–1068. 1999.Knittel T., Mehde M., Kobold D., Saile B., Dinter C., Ramadori G. Expression patterns of matrix metalloproteinases and their inhibitors in parenchymal and non-parenchymal cells of rat liver: regulation by TNF-alpha and TGF-beta1. J Hepatol. 30:48–60. 1999.Ludtke JJ., Zhang G., Sebestyen MG., Wolff JA. A nuclear localization signal can enhance both the nuclear transport and expression of 1 kb DNA. J Cell Sci. 112:2033–2041. 1999.
ArticleMoon IJ., Choi K., Choi YK., Kim JE., Lee Y., Schreiber AD., Park JG. Potent growth inhibition of leukemic cells by novel ribbon-type antisense oligonucleotides to c-myb1. J Biol Chem. 275:4647–4653. 2000a.
ArticleMoon IJ., Kang H., Seu YB., Chang BC., Song DK., Park JG. Marked transfection enhancement by the DPL (DNA/peptide/lipid) complex. Int J Mol Med. 20:429–437. 2007.
ArticleMoon IJ., Lee Y., Kwak CS., Lee JH., Choi K., Schreiber AD., Park JG. Target site search and effective inhibition of leukaemic cell growth by a covalently closed multiple anti-sense oligonucleotide to c-myb. Biochem J. 346(Pt 2):295–303. 2000b.
ArticleNakamura T., Ueno T., Sakamoto M., Sakata R., Torimura T., Hashimoto O., Ueno H., Sata M. Suppression of transforming growth factor-beta results in upregulation of transcription of regeneration factors after chronic liver injury. J Hepatol. 41:974–982. 2004.Qi Z., Atsuchi N., Ooshima A., Takeshita A., Ueno H. Blockade of type beta transforming growth factor signaling prevents liver fibrosis and dysfunction in the rat. Proc Natl Acad Sci U S A. 96:2345–2349. 1999.Ramadori G., Saile B. Portal tract fibrogenesis in the liver. Lab Invest. 84:153–159. 2004.
ArticleSchwarze SR., Ho A., Vocero-Akbani A., Dowdy SF. In vivo protein transduction: delivery of a biologically active protein into the mouse. Science. 285:1569–1572. 1999.Sorgi FL., Bhattacharya S., Huang L. Protamine sulfate enhances lipid-mediated gene transfer. Gene Ther. 4:961–968. 1997.
ArticleTorchilin VP., Rammohan R., Weissig V., Levchenko TS. TAT peptide on the surface of liposomes affords their efficient intracellular delivery even at low temperature and in the presence of metabolic inhibitors. Proc Natl Acad Sci U S A. 98:8786–8791. 2001.
ArticleUeno H., Sakamoto T., Nakamura T., Qi Z., Astuchi N., Takeshita A., Shimizu K., Ohashi H. A soluble transforming growth factor beta receptor expressed in muscle prevents liver fibrogenesis and dysfunction in rats. Hum Gene Ther. 11:33–42. 2000.
ArticleVives E., Brodin P., Lebleu B. A truncated HIV-1 Tat protein basic domain rapidly translocates through the plasma membrane and accumulates in the cell nucleus. J Biol Chem. 272:16010–16017. 1997.Wagner RW., Matteucci MD., Lewis JG., Gutierrez AJ., Moulds C., Froehler BC. Antisense gene inhibition by oligonucleotides containing C-5 propyne pyrimidines. Science. 260:1510–1513. 1993.
ArticleZanta MA., Belguise-Valladier P., Behr JP. Gene delivery: a single nuclear localization signal peptide is sufficient to carry DNA to the cell nucleus. Proc Natl Acad Sci U S A. 96:91–96. 1999.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- The Effect of Ribbon-Type Antisense Oligodeoxynucleotides for Transforming Growth Factor-beta1 in Unilateral Ureteral Obstruction
- The Change of Transforming Growth Factor-beta1 Expression and the Effect of Vitamin E in the Acute Rat Liver Injury with Carbon Tetrachloride
- Effect of Antisense TGF-beta1 Oligodeoxynucleotides in Streptozotocin-Induced Diabetic Rat Kidney
- Downregulation of TGF-beta1 Expression in Penile Corpus Cavernosum by Antisense TGF-beta1 Oligonucleotides in Diabetic Rat
- Attenuation of the Expression of Fibrogenic Molecules by Transforming Growth Factor-beta1 Antisense in Cultured Rat Mesangial Cells