J Korean Med Sci.
2014 Sep;29(Suppl 2):S131-S138. 10.3346/jkms.2014.29.S2.S131.
Estimated Amount of 24-Hour Urine Sodium Excretion Is Positively Correlated with Stomach and Breast Cancer Prevalence in Korea
- Affiliations
-
- 1Department of Internal Medicine, Research Institute of Medical Science, Konkuk University School of Medicine, Seoul, Korea.
- 2Department of Internal Medicine, Armed Forces Capital Hospital, Seongnam, Korea.
- 3Department of Internal Medicine, Seoul Paik Hospital, Inje University College of Medicine, Seoul, Korea.
- 4Department of Internal Medicine, Ilsan Paik Hospital, Inje University College of Medicine, Goyang, Korea.
- 5Research Institute of Salt and Health, Seoul K-Clinic, Seoul, Korea.
- 6Department of Internal Medicine, Seoul National University Bundang Hospital, Seongnam, Korea. mednep@snubh.org
- 7Department of Internal Medicine, Seoul National University College of Medicine, Seoul, Korea.
- 8Department of Immunology, Seoul National University Postgraduate School, Seoul, Korea.
- 9Renal Institute, Seoul National University Medical Research Center, Seoul, Korea.
- KMID: 2069804
- DOI: http://doi.org/10.3346/jkms.2014.29.S2.S131
Abstract
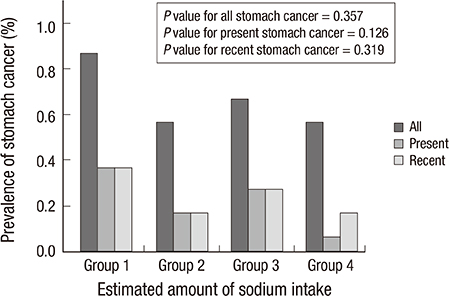
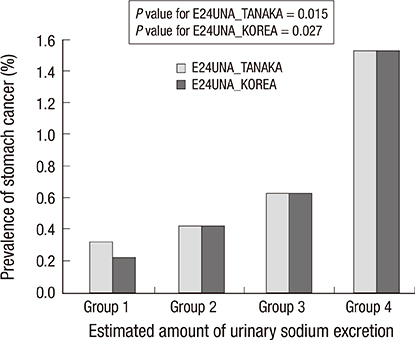
- Stomach cancer is one of the most common cancers in Korea. The aim of this study was to identify the association between the prevalence of cancer, particularly stomach cancer, and the amount of 24-hr urine sodium excretion estimated from spot urine specimens. The study included 19,083 subjects who took part in the Korean National Health and Nutritional Examination Survey between 2009 and 2011. The total amount of urine sodium excreted in a 24-hr period was estimated by using two equations based on the values for spot urine sodium and creatinine. In subjects who had an estimated 24-hr urine sodium excretion of more than two standard deviations above the mean (group 2), the prevalence of stomach cancer was higher than in subjects with lower 24-hr sodium excretion (group 1). By using the Tanaka equation to estimate it, the prevalence of stomach cancer was 0.6% (114/18,331) in group 1, whereas it was 1.6% (9/568) in group 2 (P=0.006). By using the Korean equation, the prevalence was 0.6% (115/18,392) in group 1, and 1.6% in group 2 (8/507) (P=0.010). By using the Tanaka equation, breast cancer in women is more prevalent in group 2 (1.9%, 6/324) than group 1 (0.8%, 78/9,985, P=0.039). Higher salt intake, as defined by the estimated amount of 24-hr urine sodium excretion, is positively correlated with a higher prevalence of stomach or breast cancer in the Korean population.
Keyword
MeSH Terms
Figure
Cited by 1 articles
-
Designing optimized food intake patterns for Korean adults using linear programming (II): adjustment of the optimized food intake pattern by establishing stepwise intake goals of sodium
Kana Asano, Hongsuk Yang, Youngmi Lee, Meeyoung Kim, Jihyun Yoon
J Nutr Health. 2019;52(4):342-353. doi: 10.4163/jnh.2019.52.4.342.
Reference
-
1. Jemal A, Bray F, Center MM, Ferlay J, Ward E, Forman D. Global cancer statistics. CA Cancer J Clin. 2011; 61:69–90.2. Jung KW, Won YJ, Kong HJ, Oh CM, Seo HG, Lee JS. Cancer statistics in Korea: incidence, mortality, survival and prevalence in 2010. Cancer Res Treat. 2013; 45:1–14.3. Kono S, Hirohata T. Nutrition and stomach cancer. Cancer Causes Control. 1996; 7:41–55.4. Honjo S, Kono S, Yamaguchi M. Salt and geographic variation in stomach cancer mortality in Japan. Cancer Causes Control. 1994; 5:285–286.5. Tsugane S, Tsuda M, Gey F, Watanabe S. Cross-sectional study with multiple measurements of biological markers for assessing stomach cancer risks at the population level. Environ Health Perspect. 1992; 98:207–210.6. Bates CJ, Thurnham DI, Bingham SA, Margetts BM, Nelson M. Biochemical markers of nutrient intake. In : Margetts BM, Nelson M, editors. Design concepts in nutritional epidemiology. Oxford, New York: Oxford University Press;1991. p. 170–240.7. Hunter D. Biochemical indicators of dietary intake. In : Willett W, editor. Nutritional epidemiology. New York: Oxford University Press;1998. p. 174–243.8. Levey AS, Coresh J, Greene T, Stevens LA, Zhang YL, Hendriksen S, Kusek JW, Van Lente F. Using standardized serum creatinine values in the modification of diet in renal disease study equation for estimating glomerular filtration rate. Ann Intern Med. 2006; 145:247–254.9. Coresh J, Astor BC, Greene T, Eknoyan G, Levey AS. Prevalence of chronic kidney disease and decreased kidney function in the adult US population: Third National Health and Nutrition Examination Survey. Am J Kidney Dis. 2003; 41:1–12.10. Tanaka T, Okamura T, Miura K, Kadowaki T, Ueshima H, Nakagawa H, Hashimoto T. A simple method to estimate populational 24-hr urinary sodium and potassium excretion using a casual urine specimen. J Hum Hypertens. 2002; 16:97–103.11. Sjodahl K, Jia C, Vatten L, Nilsen T, Hveem K, Lagergren J. Salt and gastric adenocarcinoma: a population-based cohort study in Norway. Cancer Epidemiol Biomarkers Prev. 2008; 17:1997–2001.12. Kneller RW, McLaughlin JK, Bjelke E, Schuman LM, Blot WJ, Wacholder S, Gridley G, CoChien HT, Fraumeni JF Jr. A cohort study of stomach cancer in a high-risk American population. Cancer. 1991; 68:672–678.13. Ngoan LT, Mizoue T, Fujino Y, Tokui N, Yoshimura T. Dietary factors and stomach cancer mortality. Br J Cancer. 2002; 87:37–42.14. Shikata K, Kiyohara Y, Kubo M, Yonemoto K, Ninomiya T, Shirota T, Tanizaki Y, Doi Y, Tanaka K, Oishi Y, et al. A prospective study of dietary salt intake and gastric cancer incidence in a defined Japanese population: the Hisayama study. Int J Cancer. 2006; 119:196–201.15. Tsugane S, Sasazuki S, Kobayashi M, Sasaki S. Salt and salted food intake and subsequent risk of gastric cancer among middle-aged Japanese men and women. Br J Cancer. 2004; 90:128–134.16. van den Brandt PA, Botterweck AA, Goldbohm RA. Salt intake, cured meat consumption, refrigerator use and stomach cancer incidence: a prospective cohort study (Netherlands). Cancer Causes Control. 2003; 14:427–438.17. Intersalt Cooperative Research Group. Intersalt: an international study of electrolyte excretion and blood pressure. Results for 24 hour urinary sodium and potassium excretion. BMJ. 1988; 297:319–328.18. Subramanian S, Teo BW, Toh QC, Koh YY, Li J, Sethi S, Lee EJ. Spot urine tests in predicting 24-hour urine sodium excretion in Asian patients. J Ren Nutr. 2013; 23:450–455.19. Karimi Z, Jessri M, Houshiar-Rad A, Mirzaei HR, Rashidkhani B. Dietary patterns and breast cancer risk among women. Public Health Nutr. 2014; 17:1098–1106.20. Kato S, Tsukamoto T, Mizoshita T, Tanaka H, Kumagai T, Ota H, Katsuyama T, Asaka M, Tatematsu M. High salt diets dose-dependently promote gastric chemical carcinogenesis in Helicobacter pylori-infected Mongolian gerbils associated with a shift in mucin production from glandular to surface mucous cells. Int J Cancer. 2006; 119:1558–1566.21. Takahashi M, Hasegawa R. Enhancing effects of dietary salt on both initiation and promotion stages of rat gastric carcinogenesis. Princess Takamatsu Symp. 1985; 16:169–182.22. Takahashi M, Kokubo T, Furukawa F, Kurokawa Y, Tatematsu M, Hayashi Y. Effect of high salt diet on rat gastric carcinogenesis induced by N-methyl-N'-nitro-N-nitrosoguanidine. Gan. 1983; 74:28–34.23. Furihata C, Ohta H, Katsuyama T. Cause and effect between concentration-dependent tissue damage and temporary cell proliferation in rat stomach mucosa by NaCl, a stomach tumor promoter. Carcinogenesis. 1996; 17:401–406.24. Charnley G, Tannenbaum SR. Flow cytometric analysis of the effect of sodium chloride on gastric cancer risk in the rat. Cancer Res. 1985; 45:5608–5616.25. Gancz H, Jones KR, Merrell DS. Sodium chloride affects Helicobacter pylori growth and gene expression. J Bacteriol. 2008; 190:4100–4105.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Evaluation of Random Urine Sodium and Potassium Compensated by Creatinine as Possible Alternative Markers for 24 Hours Urinary Sodium and Potassium Excretion
- Methodological issues in estimating sodium intake in the Korea National Health and Nutrition Examination Survey
- 24-hour urine sodium excretion among renal donors in India: a cross-sectional study
- High Sodium Intake in Women with Metabolic Syndrome
- Validity of Estimating Sodium Intake using a Mobile Phone Application of 24-hour Dietary Recall with Meal Photos