Cancer Res Treat.
2008 Dec;40(4):190-196.
Association of Single Nucleotide Polymorphisms in PIM-1 Gene with the Risk of Korean Lung Cancer
- Affiliations
-
- 1Department of Internal Medicine, Korea University College of Medicine, Seoul, Korea. yhk0215@korea.ac.kr
- 2Department of Internal Medicine and Division of Brain Korea 21 Project for Biomedical Science, Korea University Anam Hospital, Seoul, Korea.
- 3Genomic Research Center for Lung and Breast/Ovarian Cancers, Korea University Anam Hospital, Seoul, Korea.
- 4DNA Link Inc., Seoul, Korea.
- 5Department of Internal Medicine, Inha University College of Medicine, Incheon, Korea.
Abstract
-
PURPOSE: The expression of the PIM-1 gene, which is a proto-oncogene that encodes a serine/threonine kinase, is associated with multiple cellular functions such as proliferation, differentiation, apoptosis and tumorigenesis. In particular, several studies have reported that the PIM-1 gene is associated with the development of lymphoma, leukemia and prostate cancer. Therefore, this study was conducted to evaluate the association between the single nucleotide polymorphisms in the PIM-1 gene and the risk of lung cancer occurrence in the Korean population.
MATERIALS AND METHODS
To evaluate the role of the PIM-1 gene in the development of lung cancer, the genotypes of the PIM-1 gene were determined in 408 lung cancer patients and 410 normal subjects.
RESULTS
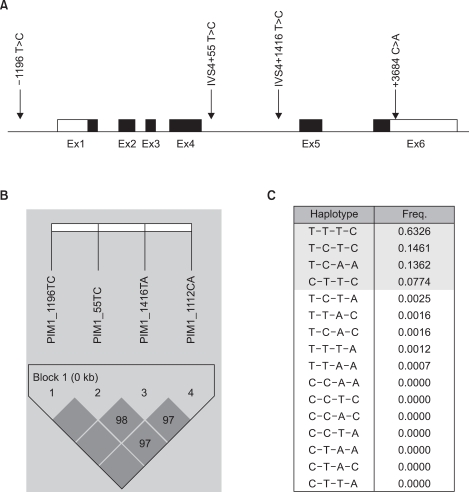
We found that the T-C-T-C haplotypes of the PIM-1 gene (-1196 T>C, IVS4 +55 T>C, IVS4 +1416 T>A and +3684 C>A) were associated with an increased risk of lung cancer [adjusted odds ratio (aOR): 3.98; 95% CI: 1.24~12.75, p-value: 0.020]. In particular, these haplotypes showed an increased risk of lung cancer in males (aOR: 5.67; 95% CI: 1.32~24.30, p-value: 0.019) and smokers (aOR: 7.82; 95% CI: 1.75~34.98, p-value: 0.007).
CONCLUSIONS
The present results suggest that the T-C-T-C haplotype of the PIM-1 gene could influence the risk of developing smoking-related lung cancer in the Korean population. Additional functional studies with an larger sample sized analysis are warranted to reconfirm our findings.
Keyword
MeSH Terms
Figure
Reference
-
1. Ionov Y, Le X, Tunquist BJ, Sweetenham J, Sachs T, Ryder J, et al. PIM-1 protein kinase is nuclear in Burkitt's lymphoma: nuclear localization is necessary for its biologic effects. Anticancer Res. 2003; 23:167–178. PMID: 12680209.2. Pasqualucci L, Neumeister P, Goossens T, Nanjangud G, Chaganti RS, Kuppers R, et al. Hypermutation of multiple proto-oncogenes in B-cell diffuse large-cell lymphomas. Nature. 2001; 412:341–346. PMID: 11460166.
Article3. Xu Y, Zhang T, Tang H, Zhang S, Liu M, Ren D, et al. Overexpression of PIM-1 is a potential biomarker in prostate carcinoma. J Surg Oncol. 2005; 92:326–330. PMID: 16299799.4. Oh Y, Herbst RS, Burris H, Cleverly A, Musib L, Lahn M, et al. Enzastaurin, an oral serine/threonine kinase inhibitor, as second- or third-line therapy of non-small-cell lung cancer. J Clin Oncol. 2008; 26:1135–1141. PMID: 18309949.
Article5. Shepherd FA, Rodrigues Pereira J, Ciuleanu T, Tan EH, Hirsh V, Thongprasert S, et al. Erlotinib in previously treated non-small-cell lung cancer. N Engl J Med. 2005; 353:123–132. PMID: 16014882.
Article6. Shin HR, Jung KW, Won YJ, Kong HJ, Yim SH, Sung JH, et al. National Cancer Incidence for the Year 2002 in Korea. Cancer Res Treat. 2007; 39:139–149.
Article7. Parkin DM, Bray F, Ferlay J, Pisani P. Global cancer statistics, 2002. CA Cancer J Clin. 2005; 55:74–108. PMID: 15761078.
Article8. Brambilla E, Travis WD, Colby TV, Corrin B, Shimosato Y. The new World Health Organization classification of lung tumours. Eur Respir J. 2001; 18:1059–1068. PMID: 11829087.
Article9. Matsuzaki H, Loi H, Dong S, Tsai YY, Fang J, Law J, et al. Parallel genotyping of over 10,000 SNPs using a one-primer assay on a high-density oligonucleotide array. Genome Res. 2004; 14:414–425. PMID: 14993208.
Article10. Kruglyak L, Nickerson DA. Variation is the spice of life. Nat Genet. 2001; 27:234–236. PMID: 11242096.
Article11. Barrett JC, Fry B, Maller J, Daly MJ. Haploview: analysis and visualization of LD and haplotype maps. Bioinformatics. 2005; 21:263–265. PMID: 15297300.
Article12. Khuder SA. Effect of cigarette smoking on major histological types of lung cancer: a meta-analysis. Lung Cancer. 2001; 31:139–148. PMID: 11165392.
Article13. Belinsky SA, Palmisano WA, Gilliland FD, Crooks LA, Divine KK, Winters SA, et al. Aberrant promoter methylation in bronchial epithelium and sputum from current and former smokers. Cancer Res. 2002; 62:2370–2377. PMID: 11956099.14. Kim DH, Kim JS, Ji YI, Shim YM, Kim H, Han J, et al. Hypermethylation of RASSF1A promoter is associated with the age at starting smoking and a poor prognosis in primary non-small cell lung cancer. Cancer Res. 2003; 63:3743–3746. PMID: 12839968.15. Agathanggelou A, Honorio S, Macartney DP, Martinez A, Dallol A, Rader J, et al. Methylation associated inactivation of RASSF1A from region 3p21.3 in lung, breast and ovarian tumours. Oncogene. 2001; 20:1509–1518. PMID: 11313894.
Article16. Shields PG, Harris CC. Cancer risk and low-penetrance susceptibility genes in gene-environment interactions. J Clin Oncol. 2000; 18:2309–2315. PMID: 10829052.
Article17. Sung JS, Han SG, Whang YM, Shin ES, Lee JW, Lee HJ, et al. Putative association of the single nucleotide polymorphisms in RASSF1A promoter with Korean lung cancer. Lung Cancer. 2008; 61:301–308. PMID: 18313166.
Article18. Bachmann M, Moroy T. The serine/threonine kinase PIM-1. Int J Biochem Cell Biol. 2005; 37:726–730. PMID: 15694833.19. Wang Z, Bhattacharya N, Weaver M, Petersen K, Meyer M, Gapter L, et al. PIM-1: a serine/threonine kinase with a role in cell survival, proliferation, differentiation and tumorigenesis. J Vet Sci. 2001; 2:167–179. PMID: 12441685.20. Mikkers H, Nawijn M, Allen J, Brouwers C, Verhoeven E, Jonkers J, et al. Mice deficient for all PIM kinases display reduced body size and impaired responses to hematopoietic growth factors. Mol Cell Biol. 2004; 24:6104–6115. PMID: 15199164.
Article21. Cuypers HT, Selten G, Quint W, Zijlstra M, Maandag ER, Boelens W, et al. Murine leukemia virus-induced T-cell lymphomagenesis: integration of proviruses in a distinct chromosomal region. Cell. 1984; 37:141–150. PMID: 6327049.
Article22. Cohen AM, Grinblat B, Bessler H, Kristt D, Kremer A, Schwartz A, et al. Increased expression of the hPim-2 gene in human chronic lymphocytic leukemia and non-Hodgkin lymphoma. Leuk Lymphoma. 2004; 45:951–955. PMID: 15291354.23. Fujii C, Nakamoto Y, Lu P, Tsuneyama K, Popivanova BK, Kaneko S, et al. Aberrant expression of serine/threonine kinase Pim-3 in hepatocellular carcinoma development and its role in the proliferation of human hepatoma cell lines. Int J Cancer. 2005; 114:209–218. PMID: 15540201.
Article24. Herman JG, Baylin SB. Gene silencing in cancer in association with promoter hypermethylation. N Engl J Med. 2003; 349:2042–2054. PMID: 14627790.
Article25. Thomas RK, Weir B, Meyerson M. Genomic approaches to lung cancer. Clin Cancer Res. 2006; 12:4384s–4391s. PMID: 16857815.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- No Association between Tumor Necrosis Factor-alpha Gene Polymorphisms and Lung Cancer Risk
- No Association between Promoter Polymorphism of STK11 Gene and Lung Cancer Risk in the Korean Population
- A Pilot Genome-Wide Association Study Identifies Novel Markers of Metabolic Syndrome in Patients with Psoriasis
- Application of Random Forests to Association Studies Using Mitochondrial Single Nucleotide Polymorphisms
- An Association Study of Polymorphisms in JAK3 Gene with Lung Cancer in the Korean Population